An Editorial Look at 1,1,1,3,3,3-Hexafluoro-2-Propanol: Opportunities and Realities
Historical Development: Bringing Fluorinated Alcohols Into the Lab
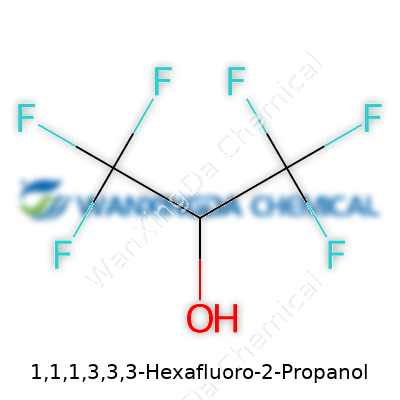
Chemists have always looked for ways to expand their toolkit. 1,1,1,3,3,3-Hexafluoro-2-propanol, better known among chemists as HFIP, represents a leap forward from the mid-20th century’s growing interest in incorporating fluorine into organic molecules. Back then, the novelty of fluorinated organics was less about performance and more about curiosity. Today, HFIP, first prepared in the context of military and industrial fluorochemicals, has become indispensable in labs worldwide. Its unique structure sets it apart: a six-fluorine substituent pattern gives it striking physical and chemical characteristics, pulling it away from more common alcohols like ethanol and methanol. This shift, from small-scale curiosity to a mainstay in synthesis labs, reflects both decades of collective effort and a shift in demand for specialty chemicals that deliver real advantages, particularly in complex organic transformations and materials science.
Product Overview: Offbeat, Yet Essential
To put it plainly, HFIP doesn’t turn heads until you use it. As a clear, heavy liquid with a distinctively pungent odor, it signals something different from the get-go. The high price tag and specialty status have kept it squarely in the hands of researchers and high-value manufacturers. Its distinctiveness isn’t superficial: the extreme electronegativity of six fluorine atoms means it behaves in often surprising ways. Anyone who’s worked with it for protein or polymer applications can recall just how rapidly it dissolves materials that other alcohols barely touch. Where water and simple alcohols fall flat, HFIP muscles through, quickly earning trust among those who need solutions—both literally and figuratively—that most solvents can’t provide.
Physical & Chemical Properties: Performance Shaped by Fluorine
Pouring HFIP feels like tipping mercury—there’s real heft behind the flow, thanks to its high density. The boiling point, sitting around 58°C, turns routine distillations into a careful balancing act, especially since it also evaporates easily and irritates mucous membranes. Its strong hydrogen-bonding capability stems from the interplay of a hydroxyl group and those fluorine neighbors, which withdraw electron density and make the alcohol proton even more acidic than you’d expect. This unique acidity sets the tone for everything else it does in the lab. HFIP dissolves polypeptides, stubborn resins, and synthetic polymers with a speed traditional alcohols never reach. Combining its low boiling point, chemical resistance, and remarkable solvation power, it lives up to its reputation as a workhorse for difficult tasks, though not without risks.
Technical Specifications & Labeling: Compliance and Clarity
No bottle of HFIP arrives without a gauntlet of warnings. Labels lay out its low flash point, volatility, and corrosive tendencies—critical information for any lab technician who values personal safety. Industry norms have standardized purity above 99 percent for most research applications, and suppliers keep water content low. Look past the technical jargon and you see practical advice: HFIP needs sealed, compatible containers, away from open flames and oxidizers, with clear reminders about required PPE. These rules do more than check boxes; they help safeguard every user who unlocks its potential in everything from unfolding proteins to prepping monomers for complex polymerizations.
Preparation Method: Fluorine Chemistry with Real-World Challenges
Making HFIP isn’t for amateurs. The traditional route begins with hexafluoroacetone, a material defined as much by its hazards as its chemistry. Reacting it with hydrogen sources, often through careful hydration or reduction, delivers the final product along with a stream of heat and highly toxic byproducts. Those who have toured bulk HFIP facilities often mention the extensive air handling precautions and specialist training these plants demand. The process underscores the broader point: specialty fluorinated chemicals draw high costs not just from rarity but the difficulty of manufacture and the need for world-class safety culture throughout the supply chain.
Chemical Reactions & Modifications: A Solvent that Doubles as a Reagent
HFIP is more than a bystander in reactions. Its acidity, paired with strong solvation power, means it often shapes outcomes simply through its presence, especially in acid-catalyzed organic transformations. Chemists lean on it for arylations, cyclizations, and late-stage modifications. Its ability to mediate peptide couplings or dissolve recalcitrant biomolecules adds a layer of strategy to protocol design. On rare occasions, its hydroxyl group will participate directly—one of those memorable lab moments when the solvent you count on quietly gets in on the action. As a modifier, it tunes properties of sheets, films, and fibers when blended in with polymers or used as a co-solvent in solution-based processing.
Synonyms & Product Names: Navigating the Language of Chemicals
HFIP sits on the roster under more than one name: Hexafluoroisopropanol, 1,1,1,3,3,3-hexafluoro-isopropanol, and in some texts, just “Hexafluoro-2-Propanol.” In patent literature, you’ll occasionally spot trade-specific codes that can confuse newcomers. Yet for most working chemists, “HFIP” remains the steady anchor, instantly recognizable across applications from university proteomics to high-tech fibers.
Safety & Operational Standards: Respect for the Hazards
There’s no shortcut when handling HFIP. Exposure brings acute respiratory, skin, and eye irritation, and the volatility means even brief encounters leave a mark. Most users have learned—often the hard way—that fume hoods and gloves aren’t optional but mandatory. Storage containers need compatibility checks, with regular inventories done to avoid deteriorated seals or unplanned reactions. Labs and plants follow standardized risk assessments and spill controls, elevated given its toxic and corrosive potential. Frequent refresher training and clear labeling are not overkill; they are the minimum standard anywhere HFIP is in regular use. The stories I’ve heard about accidental releases are never about harmless lessons, but costly, avoidable close calls that stick with a crew for years.
Application Area: Innovation Stemming from Necessity
HFIP carved itself a space in protein and peptide chemistry. Researchers rely on it to dissolve tough, hydrophobic sequences during mass spectrometry sample prep or for refolding denatured proteins in biochemistry. Polymers that resist dissolution elsewhere—think polyimides and high-strength aramids—go readily into solution under HFIP’s influence, opening the door for films and fibers with exceptional mechanical properties. The pharma sector taps into HFIP’s talent for stabilizing reactive intermediates, especially during polar or acid-catalyzed reactions. In materials science, I’ve seen it used to spin strong synthetic fibers or craft optically active polymers that resist ordinary solvents. These applications didn’t pop up by accident but through hard-fought trial and error, validated with real-world results and hundreds of peer-reviewed studies.
Research & Development: Ongoing Curiosity Drives Progress
HFIP’s track record as a solvent has inspired a steady trickle of research into next-generation fluorinated alcohols. Labs keep probing the edge, testing if tweaks in fluorine placement or backbone structure can yield even more specialized or sustainable alternatives. Analytical chemists compare HFIP to other exotic solvents for peptide sequencing, while materials scientists run head-to-heads on fiber toughness and thermal stability. The pace of publications suggests scientists see plenty of life left in HFIP—especially as industries push for finer molecular control in drug synthesis and high-performance polymers. Collaborative projects with biotechnology firms keep demonstrating not just what HFIP can do but how carefully designed variants might catch on next.
Toxicity Research: An Area of Real Concern
The conversation doesn’t end at effectiveness. Medical toxicologists warn that HFIP’s small size and volatility translate to rapid uptake in humans and animals. Acute exposures cause a mix of central nervous symptoms, respiratory distress, and organ stress—especially when concentrations rise in unventilated spaces. Chronic effects are harder to pin down, mostly due to limited long-term data, but early animal studies suggest liver and kidney risks from repeated contact. Industry keeps revisiting recommended exposure limits. Anyone who has worked with HFIP can attest that short-term discomfort is real and regulations lag behind the pace of innovation. Calls for more thorough, independently funded toxicity research echo across academic journals and regulatory forums alike, and for good reason. The promises of fluorinated solvents aren’t worth much if their long-term consequences catch up with us after widespread adoption.
Future Prospects: Weighing Performance Against Risk
HFIP’s future balances between opportunity and caution. Advances in green chemistry are pushing for safer, less persistent solvents, yet no substitute matches HFIP’s unique mix of acidity, volatility, and solvent power right now. Companies are investing in containment, recovery, and recycling methods to limit emissions. Researchers are chasing down “greener” analogs—either partially fluorinated alcohols or clever formulations that bring similar results with less hazard. Tighter regulations seem inevitable, given rising awareness and stricter workplace safety rules. Success, long-term, depends on deeper toxicology understanding, industry transparency, and an honest reckoning with both performance needs and social responsibility. Sustainability in chemical innovation never takes the easy route, but real progress owes nothing to shortcuts.
A Closer Look at the Unassuming Liquid
Anyone who’s spent time in a chemistry lab has seen glassware labeled "1,1,1,3,3,3-Hexafluoro-2-propanol"—often abbreviated as HFIP. Few bottles look so plain, yet pack so much punch for researchers and industries. The clear, sharp-smelling liquid barely catches an untrained eye, but it’s indispensable almost everywhere meticulous chemistry meets real-world need.
Dissecting Its Role in Science and Manufacturing
Let’s start in the research world. HFIP works as a powerful solvent—a go-to for dissolving stubborn biopolymers. People working with proteins and synthetic fibers hit bottlenecks with traditional solvents. HFIP cracks that nut. Dissolving proteins is incredibly tough; they're finicky, and require special handling to keep from tangling up or forming useless globs. HFIP helps coax those proteins into solution, making them available for analysis or even for spinning into synthetic fibers like polyvinylidene fluoride.
Researchers also reach for HFIP during peptide synthesis. Anyone who’s built a peptide chain knows that removing the final product from its resin support can be a chore. HFIP gets used in cleavage steps where harsh acids could ruin everything. In these cases, precision matters more than brute force, and the unique chemistry of HFIP makes it stand out from cheaper, traditional alcohols.
The Pharmaceuticals and Material Science Angle
Drug makers value HFIP for similar reasons as academic labs. Drug molecules often demand unusual solvents during discovery and testing phases. For tough-to-dissolve compounds, something as specialized as this fluorinated alcohol bridges the gap between "can’t dissolve" and "clean, workable solution." I remember a project screening anti-viral candidates, where only HFIP could dissolve the lead compound for analysis. These stories echo day-to-day across pharmaceutical research.
Material science labs push the envelope on polymers for membranes, electronics, and specialty coatings. They rely on HFIP to dissolve tricky plastics before spinning them into fibers or casting films. This comes up especially with "next-gen" materials like polyamides and polyesters designed for membranes and textiles. If the solvent balks, the material doesn’t hit target specs, and research grinds to a halt.
Why Its Chemistry Deserves Attention
Why not just use other solvents? Most solvents collapse in the face of dense or highly ordered molecules. The six fluorine atoms in HFIP give it a unique balance: it’s polar enough to dissolve both hydrophobic and hydrophilic chains, yet it doesn’t break apart fragile bonds as some more aggressive acids would. That versatility shapes real outcomes—whether it’s getting DNA ready for sequencing, tweaking a protein for study, or spinning out a uniform polymer fiber for filtration membranes.
The Worries and the Way Forward
Anyone in the lab appreciates the fast action of HFIP, but environmental and safety questions can’t be ignored. It’s toxic and requires careful handling. Chemists have to use it in fume hoods and follow disposal rules closely. The big question is whether less harmful, greener alternatives can step up. Some folks are hunting for options based on ionic liquids or new synthetic alcohols, but HFIP hasn’t been replaced in critical applications.
Progress in chemistry often rests on substances no one outside the lab talks about. HFIP doesn’t get headline stories, yet its impact surfaces quietly in medicines, materials, and research papers. Recognizing what’s inside that plain lab bottle gives a window into the pressures and choices facing both scientists and industry as they push technology forward, all while weighing safety and sustainability.
One Chemical, Many Hazards
Working with lab chemicals, and 1,1,1,3,3,3-Hexafluoro-2-Propanol in particular, has taught me that there’s no such thing as being too careful. This isn’t just another mild solvent—this stuff carries some real risks. It’s toxic, it evaporates fast, and breathing the vapors can hit lungs or nervous system hard. Exposure may also irritate the eyes and skin, so splashing or even standing around an open bottle demands a vigilant approach.
Personal Protective Gear Saves More Than Clothes
If you rush through a task without gloves or goggles, you’re betting your health for the sake of a few minutes. I’ve seen colleagues break out in a rash from just a small drop on their wrist, and one inhalation incident hospitalized a coworker who ignored the chemical fume hood. Nothing beats simple nitrile gloves, a lab coat, and safety goggles for barriers against this compound.
Avoiding skin contact really takes a bit of habit. Always check gloves for holes and make it a non-negotiable rule to wear goggles—nobody expects a splash, but accidents happen. If your lab provides a face shield for distilling or pouring, grab it every time.
Ventilation: An Unseen Lifesaver
The odor from 1,1,1,3,3,3-Hexafluoro-2-Propanol carries more than just sharp fumes; it can signal overexposure. My experience tells me: if you can smell it, move straight to the fume hood or boost the airflow. Modern labs get this right with properly maintained fume extraction, but people working in crowded maker spaces or less formal environments sometimes take shortcuts. Good ventilation isn’t expensive, but skipping it can cost you days off or worse. If you ever feel dizzy or lightheaded, step away fast and get some fresh air.
Storage and Handling: Small Steps Matter
This chemical likes sealed containers, away from anything open to the air. It can chew up certain plastics, so make sure you know what kind of bottles you’re using—stick to tested glass or recommended materials. Once, I made the mistake of reusing an old plastic bottle, only to watch it deform, and I’ll never risk it again. Any workspace holding this solvent should have spill kits—preferably those designed for fluorinated compounds—and absorbent material ready. A quick cleanup beats a slip-and-fall in the middle of an emergency.
Training and Respect Go Hand in Hand
It’s easy to underestimate chemicals you work with every day. I learned early that safety briefings aren’t just for new staff: everyone benefits from regular refreshers and drills. Real stories about incidents help these precautions stick, much more than memorizing a list ever could. If you ever have to advise a novice, walk them through a dry run with water first. People remember what they do.
Better Alerts, Safer Days
Smart labels with bold hazard warnings make a difference. Real-world experience proves people stop and think before grabbing something marked with clear warnings about toxicity or flammability. Digital logbooks where users sign out the container help track use, making sure no one leaves a cap loose or forgets proper disposal.
Good safety around 1,1,1,3,3,3-Hexafluoro-2-Propanol isn’t just about rules or risk charts. It's built on shared responsibility, honest communication, and habits reinforced day in and day out. Mistakes don’t get do-overs; respect the danger, and chemical work can stay productive without costing anyone their well-being.
Understanding the Substance
Anyone working in labs has come across chemicals that command respect. 1,1,1,3,3,3-Hexafluoro-2-Propanol is no exception. Thanks to its volatile nature and ability to break down some plastics and rubbers, letting your guard down with this one can cost more than just equipment. Most researchers recognize those sharp, pungent fumes and know this isn’t something you casually toss onto a shelf.
Why Storage Matters Here
I’ve watched colleagues scramble to deal with cracked vials and ruined gloves after a careless storage decision. This solvent goes through materials many expect to withstand chemical attack. Glass works, but stashing it in plastic—even “chemical-resistant” plastic—has quietly eaten holes in more than a few safety records. The vigilance for correct materials pays off, both for people and the environment.
1,1,1,3,3,3-Hexafluoro-2-Propanol evaporates quickly, filling the air with harsh fumes. Long-term inhalation can impact the nervous system, sometimes even at low exposure levels. I remember a postdoc who underestimated just one minor spill; her headache and fogginess lingered for hours. Fume hoods aren’t just a good idea—they’re non-negotiable.
Key Storage Practices That Protect People and Research
Proper storage starts with well-sealed glass containers. Tightly closed caps protect everyone from leaking vapors. Working at room temperature in a cool, well-ventilated space keeps the risk of vapor build-up low.
This chemical doesn’t play nicely with open shelves above eye level. Spills land squarely on shoes or skin, and this solvent’s bite is worse than its bark. Storing it at bench height—never overhead—prevents avoidable accidents. Flammability remains a risk factor too. It seems counterintuitive since it’s usually water-clear and unassuming, but it can catch fire around open flames or sparks. Always choose flame-resistant storage cabinets for flammable solvents, even if mixing it with less hazardous stuff seems tempting.
Moisture causes quality problems. Humidity invites hydrolysis, which means the solvent could react or degrade, leading to unpredictable contamination. Dry cabinets and desiccators become good friends in any lab that values reliability, especially with a material this sensitive. I’ve seen research projects take a beating when solvents pick up water from a damp basement shelf—months of work lost, just from water sneaking into a bottle.
Thinking Beyond the Individual vial
Training others to treat this chemical with healthy caution carries real weight. I recall a safety seminar where a younger scientist admitted never hearing about its skin or inhalation dangers. After that, our team started routine refreshers, and near-misses dropped off.
Staying transparent about risks protects the youngest and most experienced in a lab. Posting signs, using labels with strong warnings, and writing protocols that remind everyone how quickly things can change add layers of protection. Real safety comes from respect—not just for the rules but for the lived experience behind them. The safety steps we take each day build habits that last far longer than an inspection checklist.
Realistic, Simple Safeguards
Storing 1,1,1,3,3,3-Hexafluoro-2-Propanol in the right spot, in the right container, says something about who you work with and how much you value clean science. It comes down to glass containers, tight caps, fireproof cabinets, dry storage, and clear labeling. Building these practices isn’t just about compliance, it’s about making sure everyone gets home healthy—and the science stays trustworthy and strong.
1,1,1,3,3,3-Hexafluoro-2-Propanol: More Than Just a Solvent
Walk into any synthetic organic chemistry lab and you’re bound to find bottles labeled “HFIP” stacked on shelves. Chemistry students joke about how quickly a faint whiff can fill the room. It’s not just another clear liquid hiding behind a complicated name—this stuff matters. In research years back, I ran into it while trying to break through with a tough peptide coupling. Bringing HFIP into the mix felt a bit like bringing jumper cables to a dead battery. Suddenly, reactions that wouldn’t start chugged along.
What Makes HFIP Stand Out?
Dive into its structure and the secret becomes clear: those six fluorine atoms don’t just look impressive on paper. They make HFIP more acidic than your everyday alcohols like isopropanol. Put simply, the molecule gives up a proton easier. Acidity around pKa 9.3 might not wow folks outside the lab, but it’s just right when working with delicate reactions or breaking stubborn hydrogen bonds. For scientists who want control over their reactions, having a solvent like HFIP opens new doors.
Another point: HFIP can dissolve things that usually resist solution, including certain proteins and polymers. There’s a reason biochemists reach for it when trying to untangle gnarly protein aggregates or disrupting amyloid fibrils. I’ve shared benches with folks who relied on HFIP to prepare peptide samples, swearing nothing else seemed to work. Its solubilizing power deserves respect, though its volatility and strong odor keep everyone on their toes.
Physical Traits, Personal Experiences
HFIP’s boiling point hovers low, just around 58°C. You set a bottle on the bench and before long, that sharp, almost surgical smell lingers. It lets chemists distill it off gently, but also means lab gloves and goggles become non-negotiable. This is not just about comfort—with repeated use, I learned firsthand that exposure to the vapors brings headaches and irritation fast. The liquid mixes easily with water and common organics, which can be a blessing and a curse. Trickling HFIP into water triggers heat. Mishandle this, and a small experiment unexpectedly grows into a brief panic.
Its density—higher than water—often surprises newcomers. I saw a graduate student spill a few milliliters once and watch it sink straight through aqueous waste. Knowing these quirks makes a difference between a well-run synthesis and a rough lab day.
Environmental and Safety Impact
The story doesn’t end with efficiency. Like most fluorinated organics, HFIP resists breakdown, staying persistent in the environment. If small spills get into drains, the consequences don’t appear right away, but accumulation can become a worry for local wildlife and water systems. I’ve seen labs adopt stricter handling protocols, including fume hood-only use and hazardous waste disposal to cut down the risk. With organizations tightening standards for lab emissions and solvent handling, there’s growing pressure to balance innovation against exposure and ecological impact.
Smart Use, Safer Future
Scientists continue exploring greener alternatives, but for now, mastery over HFIP means respecting what it brings to the table. It’s a powerful tool for chemists and biochemists willing to put in the time learning the ropes. The secret lies not just in its chemistry, but in the habits built around its use—protective gear, good ventilation, responsible waste collection, and an eye on what comes next for the environment. That’s a lesson books won’t always spell out, but one I wish every new researcher heard on day one.
Understanding the Realities of Mixing Chemicals in the Lab
Working in chemistry labs long enough, you start to respect the rules of chemical compatibility. Small mistakes can trigger headaches, wasted time, or dangerous incidents. Take 1,1,1,3,3,3-Hexafluoro-2-Propanol. Here’s a liquid that’s earned a place on many shelves for good reason. It dissolves peptides, stands up as a reagent for coupling reactions, and helps researchers handle tough-to-dissolve biological samples. Yet, every time I see someone reaching for it with that ‘all-purpose solvent’ attitude, I remember the stories of ruined experiments and scorched glassware. Compatibility—real, tested compatibility—matters.
What Sets 1,1,1,3,3,3-Hexafluoro-2-Propanol Apart
This isn’t your ordinary alcohol. The six fluorine atoms make it polar and batch it into a category of solvents that act as both strong hydrogen-bond donors and acceptors. Its high volatility lets people dry samples easily, but its aggressive nature also trips up those who overlook its quirks. It attacks some plastics, chews up rubber, and doesn’t play nicely with certain strong bases or acids. If you store solutions in containers not rated for it, you end up with sticky messes and lost inventory.
Factoring in Compatibility Before Mixing
Blending this solvent into aqueous buffers, you get the benefit of dissolving proteins, yet, at higher concentrations, it denatures those same proteins—the solution becomes a double-edged sword. In organic synthesis, folks appreciate its unique solvent power, but mix it with reactive metals or alkali metals, and you risk hazardous byproducts, even fire. I’ve lost count how often someone assumes because it’s an alcohol, it ‘should’ mix without incident. Chemistry rarely rewards assumptions. The Material Safety Data Sheet calls out potential dangers: contact with oxidizers raises explosion risks, and combining with strong bases or reducing agents sometimes leads to heat, unwanted products, or material degradation.
Where Safety Meets Pragmatism
Lab safety walks are often treated like paperwork, but I’ve seen near-misses that make you rethink every habit. That small bottle of hexafluoro-2-propanol has a low flash point and vapor pressure high enough to surprise even seasoned folks. In a poorly ventilated closet, one careless mix-up can become everybody’s problem. Gloves and goggles aren’t optional; neatness counts for more than you expect. Glass, PTFE, or stainless steel containers usually offer a safe bet, since plastics and soft metals can deteriorate or allow slow leaks.
Thinking Ahead—A Culture of Testing, Not Guessing
A single compatibility mishap can damage months of work or put colleagues at risk. Even with years of experience, I don’t rely on memory—I check chemical compatibility charts and cross-reference with up-to-date literature, since chemical suppliers sometimes change formulations. New composites or specialty containers appear all the time, but nothing replaces a careful controlled test of mixing small quantities first. Institutions do well to keep updated lists of chemicals in shared spaces that don’t react safely together. Training new staff to read safety sheets, look up product information from reputable sources like NIOSH or PubChem, and ask questions before combining anything unfamiliar goes a long way toward keeping the lab productive and safe.
The Lasting Value of Healthy Skepticism
Every chemical, no matter how useful, comes with its own set of limitations. The best labs I’ve worked in foster a culture proud of asking questions, double-checking the basics, and sharing stories of what went right and what nearly went wrong. 1,1,1,3,3,3-Hexafluoro-2-Propanol’s quirks make it valuable and risky in equal measure. Compatibility isn’t about memorizing a chart, but about respecting the unique reality of each experiment, each reagent, and each person in the room.