1,1-Difluoroacetone: A Closer Look at Its Scientific Journey and Societal Impact
Historical Development
It’s easy to overlook the significance of molecules like 1,1-difluoroacetone unless you’ve spent time in a research lab watching chemists pick apart carbon skeletons and test the influence of halogen atoms. The fluorination of organic compounds didn’t hit mainstream chemistry overnight. Back during the earlier half of the twentieth century, chemists got interested in the unique properties of carbon-fluorine bonds. They noticed fluorinated building blocks behaved quite differently from their hydrogenated cousins. Creating something as simple as 1,1-difluoroacetone meant not just tacking on a pair of fluorine atoms, but also wrangling with reactivity, scarcity, and cost. Today, it stands as another reminder that big shifts in applications can start with small tweaks to molecular structures. Documented syntheses and mentions of 1,1-difluoroacetone trace back to the decades when chemists started understanding the stability and biological behaviors induced by fluorine atoms, and since then its inclusion in research libraries has only grown.
Product Overview
Ask a working organic chemist about fluorinated ketones and they’ll probably mention volatility and reactivity before rambling about data sheets. 1,1-Difluoroacetone isn’t something you leave open on a benchtop. Its formula—C3H4F2O—packs two fluorines next to the carbonyl, adding both electronic and steric twists. As a colorless liquid, it draws attention because small molecules with highly electronegative atoms often spark creativity in synthetic routes. You typically won’t find it in your local hardware store or common chemical supplier, but in the right hands, it acts as an invaluable reagent, especially for folks pushing boundaries in fluorine chemistry.
Physical & Chemical Properties
This compound lives up to its expectations as a fluoro-organic ketone. It evaporates quickly, even when stored carefully, so containment matters. Its boiling point sits relatively low—a reminder of the volatility common to fluorinated species. The chemical structure delivers notable polarity; two electronegative fluorine atoms fight for dominance near a carbonyl, leading to considerable dipole moments. It’s these electronic effects that make it an attractive candidate for downstream reactions, providing both a reactive carbonyl and the strong electron-withdrawing influences of the fluorines. These properties make handling a challenge but also the benefit chemists chase during synthesis.
Technical Specifications & Labeling
In the context of laboratory and research settings, standard labeling incorporates its molecular formula, safety hazards, and storage instructions tailored to its volatility and potential for inhalation risk. Most reputable suppliers include guidance around temperature-controlled environments to minimize loss and accidental exposure. The need for clear identification on all storage vessels remains non-negotiable; careless handling invites unnecessary risk, not only from the parent compound but also from any breakdown products.
Preparation Method
Producing 1,1-difluoroacetone means working with sensitive and sometimes hazardous fluorinating agents. In labs, researchers have reported successful preparations through halogen exchange reactions, starting from more common haloacetones or via halogenation of suitable acetone derivatives. The process often requires carefully controlled conditions, since introducing fluorine to organic frameworks can lead to unwanted side reactions and significant yield losses. From my own experience prepping fluorinated intermediates, every step demands patience—sometimes fluorination refuses to go as planned, and the wastes generated carry handling concerns, as well.
Chemical Reactions & Modifications
The real value in 1,1-difluoroacetone lies in its willingness to serve as both a carbonyl-containing scaffold and a fluoroalkyl donor. It reacts with nucleophiles, undergoes condensation with various amines, and can serve as a precursor for larger, more complex fluorinated molecules. Each transformation owes much to the combined push of the carbonyl and the electronic pull of the fluorines. This dual characteristic opens the door for creative synthesis, whether you’re customizing pharmaceuticals or developing new agrochemicals. In hands familiar with handling fluoro-organic chemistry, it acts less like a challenge and more as a springboard for pushing into less-explored regions of molecular space.
Synonyms & Product Names
Depending on the literature or supplier, the same chemical gets referenced under different aliases. 1,1-Difluoro-2-propanone and methyl difluoromethyl ketone show up as synonyms, but the chemistry always comes back to the same volatile, challenging liquid that interests researchers. The importance of proper nomenclature seems tedious, but in multi-lingual and multi-disciplinary research, it keeps communication clear and accidents rare. Names serve more than just database entries—they help assure reagents meet expectations in international collaborations.
Safety & Operational Standards
Lab workforces always feel the shadow of chemical safety, especially with lower-molecular-weight, highly volatile organofluorine compounds. I’ve watched peers skip on gloves or dismiss the need for fume hoods, only to face headaches or worse. 1,1-Difluoroacetone calls for strict respect, as its volatility pairs with a moderate toxicity profile and likely irritant properties. Protective eyewear, gloves resistant to fluorinated solvents, and well-functioning local exhaust ventilation stand as basic requirements. Chemical safety training drills and real-time hazard labeling matter, particularly around new staff or visiting researchers. Regulatory standards done right create habits that go beyond standard operating procedures—exposing students and early-career researchers to consequences and correct responses before mistakes lead to harm.
Application Area
Researchers pull 1,1-difluoroacetone from shelves not out of convenience, but for the unique opportunities it creates in synthesis. Drug design and medicinal chemistry departments look for new ways to introduce fluorinated compounds, banking on the metabolic stability and biological activity tweaks that come from swapping in fluorine atoms. Fluorine rarely behaves like its periodic table neighbors, making it a critical design element in active pharmaceutical ingredients, enzyme inhibitors, and specialized agrochemicals. Materials science teams occasionally reach for it as well, testing its effects in polymer backbones or advanced performance coatings. The range remains niche, yet its influence on specialty chemical design can’t be denied.
Research & Development
The world of fluorinated intermediates doesn’t stand still. Chemists at universities and private companies alike chase the next big fluorine twist. Research on 1,1-difluoroacetone today leans toward using it as a substrate for creating more functionalized derivatives—longer-chain molecules, ring systems, and even highly fluorinated macromolecules. Studies get funded by pharmaceutical leads that want new binding motifs, along with materials scientists pushing for thermally stable polymers. Every new paper or patent offers a reminder that investment in safe, efficient, scalable synthetic methods can pay dividends across many sectors, sometimes in unexpected ways.
Toxicity Research
While some organofluorines go through toxicity studies early, others sneak into wider use before scientists fully understand the risks. With 1,1-difluoroacetone, available studies suggest it causes irritation of eyes, skin, and respiratory tract if mishandled. The volatility poses inhalation hazards in enclosed spaces. Longer-term data on chronic effects remains scarce, pointing to an ongoing challenge in the field: balancing innovation with measured, thorough investigation of health impacts. In my own lab experience, erring on the side of caution and treating all new volatile fluorinated compounds as potential hazards pays off. Training newcomers to recognize subtle symptoms, document all exposures, and report incidents adds another layer of community-driven protection.
Future Prospects
Fluorinated building blocks continue to draw attention because they promise benefits for drug discovery, materials science, and crop protection. Whether 1,1-difluoroacetone grows more important will come down to how researchers tackle two main hurdles: safe, economic synthesis and a fuller understanding of downstream biological impacts. Research teams look for ways to improve fluorination techniques, cut down on unwanted by-products, and reduce occupational hazards. Increased access to real-time toxicity assessment technologies may transform how quickly compounds like 1,1-difluoroacetone move from one-off research curiosities to mainstays in chemical supply. Everyone in the chain, from bench scientist to regulatory reviewer, must commit to both innovation and vigilance, ensuring that each leap in chemistry doesn’t burden society with unforeseen risks.
1,1-Difluoroacetone: Simple Chemistry, Broad Reach
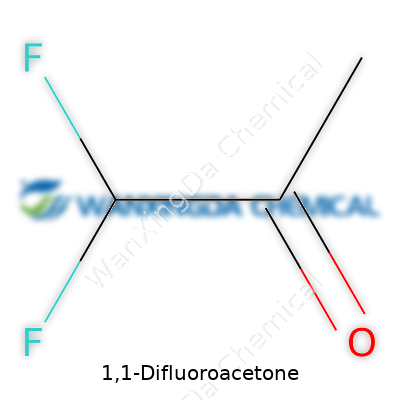
You don’t need a degree in organic chemistry to spot the patterns in molecules, but every so often, a name like 1,1-difluoroacetone shows up and has people scratching their heads. The chemical formula for 1,1-difluoroacetone is C3H4F2O. Breaking that down, it’s based on acetone, which many know as nail polish remover or a powerful solvent, but with two fluorine atoms swapped in for two of the hydrogens on one side. Add to that the oxygen double-bonded in the middle, and you have a structure that packs a punch both chemically and in terms of its uses.
Why This Molecule Stands Out
Fluorine isn’t your everyday atom. It’s one of the most electronegative elements, which means it grabs electrons with gusto. Replacing hydrogens with fluorines alters how acetone behaves. Gases, refrigerants, pharmaceuticals, and even some pesticides count on special properties from fluorine substitutions. These tweaks let chemists build compounds that resist breakdown, offer better metabolic stability, and often bring unique reactivity to the table.
From a personal standpoint, the appeal of 1,1-difluoroacetone rests in the fine balance between simplicity and possibility. Chemists love these kinds of molecular shifts because they can take a workhorse chemical and turn it into something with fresh purpose. Acetone alone isn’t rare in labs or garages. Add those two fluorines and suddenly it’s relevant for people designing new drugs or seeking safer solvents that won’t react too quickly or degrade in storage.
Practical Impact and Why Accuracy Matters
Working in research or chemical manufacturing means living with formulas and structures all day long. Mix up those details, and projects can miss the mark. Years ago, while working in a university lab, a simple mislabeling of a molecule led to a synthesis dead end—hours lost only because one atom had gone missing in a record-keeping mixup. Getting C3H4F2O right for 1,1-difluoroacetone isn’t just about trivia; it’s about safeguarding research and accurate communication.
Cells, tissues, and environmental systems treat a molecule with two fluorines much differently from one with none. Fluorinated compounds, for instance, can persist for long stretches in water or soil. That’s helpful—if you’re looking for stability. It can be a problem if the molecule gets into places it shouldn’t or resists breakdown longer than it should. Before using or disposing of chemicals like 1,1-difluoroacetone, companies need to have clear protocols and responsibility for their environmental impact.
Building Solutions from Knowledge
The best way forward starts with education. Teachers already see that small changes in a formula can lead to big changes in behavior. Industry and academia alike benefit from a focus on accuracy and open exchanges of research. Sharing insight and speaking plainly about molecules—what’s in them, what they do, and what risks they pose—keeps mistakes to a minimum and innovation on track.
As new molecules get tangled into everything from medication to electronics, it matters that people keep learning and questioning, double-checking the facts. One simple formula—C3H4F2O—underpins not just a laboratory oddity, but a springboard for new ideas and smarter solutions.
Why Chemists Pay Attention to 1,1-Difluoroacetone
Anyone who’s spent hours in a research lab knows the thrill of discovering a new molecule and figuring out how it opens doors in chemical synthesis. 1,1-Difluoroacetone isn’t the flashiest chemical, but it packs value for people trying to make things happen in organic chemistry. Working with it, you’ll notice how having two fluorine atoms next to a carbonyl group brings interesting reactivity — for better or worse.
Applications in Medicinal Chemistry
Drug discovery keeps marching forward with tools like 1,1-difluoroacetone. Medicinal chemists use it to bring fluorine atoms directly into carbon frameworks, which can boost metabolic stability or change a compound’s biological activity. Fluorine changes a molecule’s properties dramatically, sometimes leading to new pharmaceutical candidates that a non-fluorinated version couldn’t offer.
During my time exploring small-molecule synthesis in grad school, every time a fluorinated building block showed up in a project, there’d be real excitement about shifting a molecule’s behavior in the body. Fluorinated methyl ketones derived from 1,1-difluoroacetone have been explored in enzyme inhibitors and sometimes give drug candidates an edge by making them harder to break down in the liver. The compound often serves as a shortcut for introducing gem-difluoromethyl groups in lead optimization campaigns, which means medicinal chemists keep it on their radar.
Role in Agrochemical Research
Crops need protection from pests, and agrochemical researchers want to make plant treatments more effective and environmentally friendly. Fluorinated compounds tend to resist breakdown, sticking around long enough to do their job. Formulators often try out building blocks from compounds such as 1,1-difluoroacetone, aiming to tweak pest or herbicide molecules for slower degradation and selective targeting.
The push for better crop yields without excessive spraying means putting time into substances that can stand up to tough field conditions. The strength and persistence offered by difluoroacetone-based structures open up options for innovation, so the compound ends up featured in the development pipeline for new fungicides or herbicides, especially during the trial-and-error periods leading up to big breakthroughs.
Synthetic Pathways and Chemical Method Development
Anyone who develops new reactions or builds complex molecules has probably seen project notes referencing 1,1-difluoroacetone somewhere. Among all the acetone derivatives, this one lets you put difluoro groups on carbon atoms in a direct fashion. In building molecular scaffolds, adding specific fluorines is often tough, but difluoroacetone simplifies things. It enables coupling reactions or Michael additions, feeding into a library of novel compounds for testing.
Researchers don’t just want a supply of building blocks; they want to invent new transformations. The unique electron-withdrawing twist that two fluorine atoms bring gets method developers tinkering on how to use 1,1-difluoroacetone for cascade reactions and complex fragment coupling.
Looking Toward Safer and Smarter Use
While the potential of this compound in research and industry is clear, safety needs special attention. Fluorinated chemicals sometimes pose environmental persistence risks, so responsible use matters. Having seen both triumphs and headaches from handling strong-smelling, volatile chemicals in the lab—I can say respect for safe storage, handling, and disposal sets apart a good chemist from a careless one. Tools like 1,1-difluoroacetone keep pushing fields forward, but only if we use them with care and an eye on both discovery and safety.
1,1-Difluoroacetone doesn’t show up in every lab, but it asks for steady hands and clear-headed responsibility wherever it does. Those who’ve spent time with reactive organofluorine compounds can tell you: a busy day can turn into a bad day if simple safeguards take the back seat. Chemicals like these don’t hand out second chances.
Keeping Track of Hazards
Every chemist remembers at least one instance where an underestimated solvent or reagent demanded emergency attention. 1,1-Difluoroacetone can irritate the skin, eyes, and airways. Enough exposure and there’s risk to your internal organs. I’ve watched colleagues skip protective gear because it seemed overcautious, only to end up with a visit to the occupational health office. Gloves, goggles, and lab coats shouldn’t be negotiable. Working in a fume hood, with its strong ventilation, reduces exposure drastically. Real-world safety isn’t built from paperwork—it’s a reflex built on lessons learned.
Smart Storage Means Fewer Surprises
Organofluorines react with lots of stuff if left unchecked. They don’t mix well with bases, oxidizers, or open flames. Store 1,1-Difluoroacetone in tightly sealed containers made of materials known to resist fluorinated compounds. Glass works but only if the lid shuts tight and labeling stays legible. I’ve seen chemicals mislabeled and moved to the wrong shelf, leading to cleanup jobs nobody wanted and headaches for the safety officer.
Temperature makes a difference. A cool, dry, and stable environment beats a fluctuating one every time. If refrigeration is recommended, make sure that the fridge isn’t a shared unit where lunch containers mingle with dangerous reagents. Fluctuating temperatures don’t just change volatility—they speed up decomposition and sometimes cause container rupture. Keeping incompatible chemicals apart in storage has saved more than one facility from fire or toxic clouds.
Training Does More Than Manuals
No high-tech system replaces simple understanding. Chemistry students and seasoned techs alike benefit from hands-on training, not just printed instructions. Spills happen fast. Knowing how to stop a leak, neutralize a splash, or get help matters much more than just reading an MSD sheet. I’ve seen well-meaning new hires clean up a spill with the wrong absorbent, sending fumes through the lab for hours. Annual refreshers and drills aren’t busywork—they’re how those slow-motion emergency moments get replaced by confident, right moves.
Cultivating a Culture of Respect
Every chemistry department holds stories about close calls and preventable mishaps. Listening to the workers who’ve been through those moments changes attitudes far quicker than another list of recommendations. Clear walkways, uncluttered benchtops, and labeled shelves save time and stress. Simple routines—chemical inventories, frequent reviews, tidy storage—create a workplace where people look out for each other.
Storing and handling 1,1-Difluoroacetone isn’t just technical procedure—it draws a line between professional diligence and unnecessary risk. Chemicals like these reward patience, awareness, and solid training. Building that kind of environment earns the respect of everyone who shares the lab bench or the storeroom, and it’s worth every bit of discipline it takes.
Unlocking Clarity in Chemistry
Some topics barely make a splash outside the lab, but a choice compound like 1,1-Difluoroacetone deserves a closer look. Its boiling point and melting point capture the sort of information chemists chase down before uncapping a single bottle. Anyone who’s worked in a lab understands how a few degrees on a thermometer can separate success from disaster. In my own experience, I’ve seen curious shortcuts lead to messes—chemical bottles leaking in fridges, reactions boiling off before anyone could set up a condenser. So a clear sense of these key temperatures takes on more weight than the dry data tucked away in reference books.
Why These Numbers Pack Punch
1,1-Difluoroacetone, also called DFA, belongs to a group of small ketones treated with respect. Pull up its boiling point—roughly 53–54 °C—and you start to see why it doesn’t just sit quietly on the shelf. Above a warm spring day, DFA skips into vapor, filling the air every time the bottle opens. Its low melting point, sinking below -80 °C, means you’ll almost never see it as a solid unless working in ultra-cold freezers. That easy transition between liquid and gas brings practical risks. I remember once lifting a vial too close to a warm lamp, and having to step back as the sharp odor stung my nose.
Contact with air stands as a silent hazard. Experience teaches you to favor cold blocks and quick hands rather than classroom moves. Failure to respect volatility triggers spills, leaks, and accidental exposure—my own white coat has caught more than a whiff from hasty hands juggling similar reagents. There’s a lesson every time: an honest understanding of boiling and melting numbers doesn’t just keep things neat, it keeps people safe.
Chemical Behavior in Daily Work
Boiling and melting points never just stay numbers on a chart; they dictate how and where a compound fits into the chemistry puzzle. A boiling point in the low 50s Celsius means DFA enters the vapor phase with just a little warmth, making it thorny for those with poor ventilation. In glovebox work or chemical synthesis, you learn to pre-chill glassware and plan fast transfers. Low melting point, liquid even at freezer temperatures, signals danger for careless storage. There are stories in every lab of freezer shelves swimming with runaway liquids after a small leak.
Professional standards match personal care. Chemists use secondary containment and cold traps for DFA to minimize releases. Not just about keeping the workspace tidy—these steps cut down inhalation risk. As someone who values a sharp mind at the end of a shift, I appreciate how following methods that suit boiling and melting behavior protects both product and people.
A Route Toward Better Lab Practice
Working with DFA sheds light on broader industry gaps. I’ve noticed newcomers lack practical training about boiling and melting risks. Universities and research mentors should weave this kind of applied knowledge into sessions, making safe handling instinctive rather than afterthought. Visual cues—labels showing temperature ranges front and center—could reduce mishaps. More companies have moved to supply chemicals with improved caps and drip-resistant bottles, a nod to the needs of everyday users.
The numbers pinned to 1,1-Difluoroacetone seem basic, yet they carry impact across research, safety, and results. The next time hands reach for a flask or slide open a fridge, it’s worth remembering why chemists keep these values close: underestimating them turns labwork from learning to cleanup, sometimes in less than a minute.
What Is 1,1-Difluoroacetone and Why Should We Care?
1,1-Difluoroacetone has become a point of discussion in chemistry circles, mostly because of its reactivity and its usefulness in advanced synthesis. At first glance, it’s another tool in the chemist's arsenal. Yet anyone who’s ever dealt with fluorinated compounds knows they tend to bring a set of challenges you just can’t brush off. With 1,1-Difluoroacetone, you’d be wrong to assume otherwise.
Handling chemicals that release fumes or have reactive carbonyl groups can go sideways quickly. Take it from someone who has witnessed “routine” synthesis sessions end up with alarms blaring because a vapor escaped containment. 1,1-Difluoroacetone fits that bill. Its volatility and tendency to react with water make it more than a minor inconvenience. These traits come with health and environmental risks that deserve direct conversation—especially given the links established between organofluorine substances and toxicity in both lab workers and ecosystems.
The Real Hazards
It doesn’t take much: sniff just a whiff of a reactive fluorinated compound and your lungs protest. The effects range from simple irritation—burning nose, watery eyes—to more serious problems like persistent coughing and dizziness. The carbonyl group in the molecule spells trouble, too. It can interact with proteins and enzymes inside the body, which research suggests might compound its toxicity. Think of it like working with old-school formaldehyde, but with the added issue of fluorine’s tendency to stick around and bioaccumulate.
Many chemists end up learning the hard way that skin contact is no joke. I remember someone in my cohort brushed a drop onto their glove during a late-night experiment, only to find that their glove didn’t provide much protection. Swift rinsing helped, but not before the sensation of burning set in. It turned out that the chemical had permeated the material—proof that standard lab gloves are often not up to the task with small, reactive molecules.
Ventilation and Barriers Matter
Experience keeps teaching the same lesson: never trust fumes to stay put, even in a closed or “well-designed” bottle. Lab hoods save lives. Their fans and glass screens don’t just make for dramatic photos; they keep volatile fumes out of your lungs and the broader workspace free of contamination. This measure remains unbeatable.
Spills are rare, but never impossible. Here’s where habits kick in: always seal containers tightly, treat every potential spill as urgent, and keep neutralizing agents within reach. You can’t plan for every incident, but you can build routines that limit risks. After all, cleaning up after a poorly managed spill often means more exposure and extra waste that’s harder to dispose of safely.
Personal Protective Equipment Often Needs an Upgrade
Lab coats and disposable nitrile gloves form the bare minimum. Yet with especially reactive, low-molecular-weight compounds, thicker gloves—from butyl or laminated film—offer better protection. Eye protection with side shielding matters, too. Even a curious splash can find its way to exposed skin before you realize what happened. It gets old repeating, but nobody remembers their goggles until they’re needed.
Better Training, Safer Workplaces
Hazardous reagents aren’t going away anytime soon, so the smartest strategy involves ongoing education and regular safety briefings. It’s one thing to read about risks and quite another to see, hands-on, what a minor mishap can turn into. A well-informed team can make smarter decisions and move quickly if something goes wrong.
In short, 1,1-Difluoroacetone isn’t a walk in the park to handle. Yes, it serves important purposes in the lab. But anybody working with it owes themselves and those around them the diligence to learn its hazards, gear up accordingly, and make safety part of the routine—every time.