1,1-Difluoroethane: More Than Just a Refrigerant
Historical Development and Product Overview
Thinking about 1,1-difluoroethane, or HFC-152a as some in the refrigeration industry call it, brings to mind the constant push for safer, more efficient industrial chemicals. In the late twentieth century, as ozone-depleting chlorofluorocarbons started going out of fashion through legislative bans, industrial chemists started looking into alternatives that wouldn't leave the air punctured and breathing spaces compromised. 1,1-difluoroethane made its way into mainstream use mostly through refrigeration and aerosol propellants. Its story doesn't really stand alone though—it's a product defined by its place in the wider journey away from harmful chemicals and toward something a little less threatening.
Physical and Chemical Properties
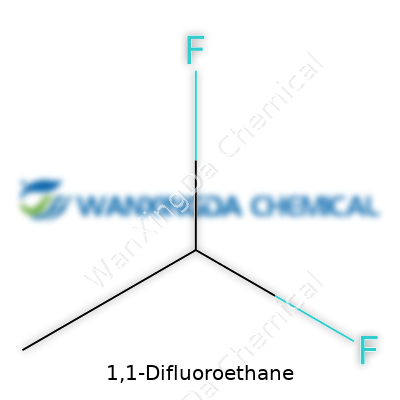
1,1-difluoroethane has a sharp, slightly sweet odor. At room temperature, this compound shows up as a colorless, flammable gas. People dealing with HVAC work or aerosol manufacturing recognize it thanks to its noticeably low boiling point, hovering around -25°C. Its density sits lower than many other common refrigerants and it dissolves decently well in alcohol or ether, but not so much in water. I've always found its molecular structure—two fluorine atoms replacing hydrogens on an ethane backbone—reminds me that even small atomic swaps can shift a substance's environmental and physical footprint.
Technical Specifications and Labeling
Labeling 1,1-difluoroethane pulls in strict international standards because its flammability creates risks beyond just chemical exposure. Storage cylinders and shipping containers bear standard gas hazard symbols, and there’s extensive documentation on pressure limits and handling instructions in safety data sheets. I find that anyone planning to use or transport this gas gets a crash course in its pressure-temperature relationships, often through hard-won experience with expansion valves or failed transfer lines during hot days.
Preparation Methods and Chemical Reactions
Production of 1,1-difluoroethane often starts with the reaction between hydrogen fluoride and acetylene or ethylene intermediates under controlled conditions. This process spins out byproducts that require careful management, since hydrofluoric acid could eat through skin, glass, and just about anything unprotected. In the lab, reactions with oxygen or chlorine shift the chemical to other fluorinated organics, though controlling conditions is key to keeping unwanted byproducts down. Sometimes, discussing reaction yields or purity in technical teams feels like playing chess—every move can affect the batch size and final safety profile.
Synonyms and Trade Names
Industrial chemicals usually carry a long list of synonyms, leading to confusion on warehouse floors or during imports. 1,1-difluoroethane has built up quite a collection—HFC-152a, Ethylidene difluoride, Freon 152a, or even R-152a for those deep in refrigeration dialogues. Differences in terminology crop up in safety data, and I’ve witnessed near mix-ups because someone used a trade name instead of the chemical designation. This highlights the need for clear, context-sensitive communication, especially in settings juggling multiple refrigerants.
Safety Measures and Operational Standards
Working with 1,1-difluoroethane means paying attention to the risks it carries. Its flammable nature turns minor leaks in tight spaces into clear hazards. Fire departments and factory safety managers rely on regular training drills with this gas because real-world slips leave little margin for error. That sharp, sweet smell isn't enough of an alert in a warehouse full of distractions. Industries adopt ventilation systems, spark-proof equipment, and multi-layer PPE based on costly lessons learned over decades. The push for transparency and regulation comes from a long trail of incidents, making every improvement in operational standards a hard-won advance.
Applications Across Sectors
Uses for 1,1-difluoroethane cut across several fields. Its mainstay as a refrigerant in low- and medium-temperature systems remains, especially for portable appliances and commercial cooling setups. Aerosol propellant applications—think spray paints or cosmetics—tapped into its low toxicity at low concentrations and ease of compression, making it a popular choice after the clampdown on CFCs. Some folks even found it useful for blowing clean electronic components, though safety remains debated since improper venting leads to quick vapor accumulation. Every sector using this chemical keeps an eye on the regulations because the stakes sit high for both health and compliance.
Research and Development Insights
Looking back at chemical research, the industry hasn't stopped at safe substitution. Current development rounds keep pressing further—lowering the global warming potential, minimizing toxicological footprints, and making recycling more effective. Some teams explore functionalizing difluoroethane molecules for specialized synthesis routes, while environmental researchers chase after breakdown pathways in the atmosphere. One thing stands out in my reading and experience: research rarely happens in isolation. Every insight feeds back into new engineering controls or informs international climate talks that steer what factories can responsibly produce.
Toxicity Research and Health Focus
Toxicologists keep a keen eye on 1,1-difluoroethane, especially since recreational abuse led to real harm in the form of cardiac arrhythmias and sudden sniffing deaths. These cases push studies on exposure thresholds—both for acute symptoms and for long-term organ effects. Emergency room data suggests regular occupational handling under strict limits doesn't seem to trigger chronic toxicity, but the margin narrows for accidental inhalation in unprotected spaces. Beyond human health, researchers map effects on wildlife and groundwater when container leaks or accidents spill the chemical outside regulated sites. These findings shape both public policy and real-life safety procedures.
Future Prospects
Fluorinated chemicals like 1,1-difluoroethane sit at a crossroads. Stricter climate targets mean industrial designers keep looking for replacements with even lower greenhouse potential. Some startups turn toward hydrofluoroolefins, making the future of 1,1-difluoroethane less certain as a mainstream refrigerant. That said, the know-how built around this molecule won't disappear overnight. Technological leaps in responsible handling, detection, and decomposition show strong potential to shape chemical stewardship for decades. Industry experts expect increasing regulatory scrutiny, but also better tools for training and detection, nudging the next generation of chemists and safety officers to take a more holistic view.
From Cleaning Keyboards to Fueling Lighters: Where 1,1-Difluoroethane Shows Up
1,1-Difluoroethane usually doesn’t come up in conversation unless you work with electronics or need to refill a cigarette lighter. Yet, it hides in plain sight, powering small but critical tasks behind the scenes. The most familiar place you’ll encounter it is inside those canned air dusters stacked up at the electronics store. After spending time repairing computers, I’ve watched these cans blow months of gunk out from keyboards and circuit boards, saving tech from sticky keys and overheating.
Some might picture canned air as filled with fresh oxygen, but that’s not the case. 1,1-Difluoroethane, a colorless and slightly sweet-smelling gas, often fills those cans because it’s non-flammable under normal use, evaporates quickly, and doesn’t damage sensitive electronics. That rapid evaporation helps dry out any moisture, so technicians trust it for cleaning. According to research from the U.S. National Institute for Occupational Safety and Health, it’s one of the more manageable options for workplace safety when handled with common sense.
The convenience doesn’t stop at cleaning. Grab a common butane lighter and there’s a good chance 1,1-Difluoroethane plays a part. Manufacturers use it either by itself or mixed with other gases as a propellant and fuel in lighters. Its pressure characteristics fit small containers, and it flows out steadily without much fuss. As a smoker in the past, I carried plenty of lighters filled with similar gases and never worried about leaks or unpredictable flames.
Aerosol Propellant that Makes Products Work
Companies turn to 1,1-Difluoroethane to spray more than air or fuel. The gas helps push cleaning agents, lubricants, and certain cosmetic sprays out of cans. If you’ve ever sprayed a cleaner on your old VCR or fiddled with an anti-static spray on your clothes, you’ve likely met this chemical. Its role as a propellant traces back decades—scientists and regulators sought options that wouldn't destroy ozone like CFCs. Statistics from the U.S. Environmental Protection Agency show less ozone depletion potential compared to older refrigerants, making it a go-to for companies wanting to avoid more heavily regulated substances.
Health and Environmental Responsibility
The story doesn’t end with convenience. Misuse looms as a real danger. People sometimes inhale products containing 1,1-Difluoroethane as a cheap high, risking serious health problems like heart arrhythmias, unconsciousness, or even sudden death. As a parent, hearing about kids or young adults experimenting with inhalants pushes home the need for clearer safety labels, robust warnings, and more education in schools. The facts don’t lie: emergency rooms report preventable injuries related to this kind of misuse every year.
Waste also stacks up. Empty duster cans often end up in the trash rather than recycling bins designed for pressurized containers. More public recycling bins and deposit programs could recover more material, cut down on landfill waste, and reduce risk to waste workers.
Looking at Safer and Sustainable Paths
Finding a balance between utility and safety takes work. Shift to refillable cleaning systems could lower single-use can dependence. Some companies focus on alternative propellants that don’t carry the same environmental baggage. Retailers play a part by keeping these products behind counters and reminding buyers about their proper use. Patchwork solutions from experience and evidence pave the way forward: smarter disposal, better warnings, and more choices lead to safer neighborhoods and less environmental stress.
Ask anyone working in industrial settings or even in DIY garages, and sooner or later, someone points to a can containing 1,1-difluoroethane. It pops up in products like air dusters and refrigerants. But only a handful of folks really weigh the flammable and explosive dangers it brings with it.
Dispelling Assumptions
There’s a dangerous trend where people tend to trust the product’s convenience instead of reading the fine print. The label on your average duster might say, “Use in a well-ventilated area. Keep away from heat, sparks, and open flame.” Somehow, these warnings become background noise. But here’s something undeniable: 1,1-difluoroethane catches fire easily. It does not take much—a spark from static electricity, an open lighter, or a simple faulty appliance can cause trouble.
Scientific reports and safety data sheets agree. The flash point of 1,1-difluoroethane sits low enough that vapors can ignite at room temperature. The stuff forms a combustible mix in air, and once ignited, it burns with a hot, almost invisible flame. Speed and intensity make it worse—so if something goes wrong, the fire doesn’t stay small.
It’s Not Just About Flames
Explosion comes next, right after fire risk. Confined vapors, like those inside a storage room or an unattended car, can lead to a buildup. If this vapor ignites—sometimes from as little as flipping a light switch—the explosion isn’t just a theory. Emergency room stories and fire marshal reports offer plenty of real-life examples.
It is easy to find incidents involving “dust-off” sprays and 1,1-difluoroethane. Improper disposal, use for cleaning electronics near a soldering iron, or even simple storage mistakes have ended in accidents big enough to wreck property or send folks to the hospital. The National Fire Protection Association ranks it as highly flammable. Inhaling the vapor out of curiosity, or for makeshift recreational highs, only adds risk—many cases have ended in fatal flash fire accidents.
Simple Habits Keep People Safe
Familiarity can turn into carelessness. People stash cans in cars, away from the reach of kids, but a summer afternoon heats up fast. Left in a trunk, a can may burst, shooting its contents into a small space. The smallest electrical arc, or even just spontaneous pressure from a hot day, can make the difference between a normal errand and a 911 call.
Engineering controls, like good ventilation and choosing proper storage spots, stop problems before they start. Regulators can help, but everybody who handles cans shares that responsibility. Awareness campaigns work best when they move beyond print manuals. Video tutorials, in-person training, and warnings in simple terms reach people better than dense material safety sheets that stay on the shelf.
Moving Toward Safer Choices
Companies begin to look for non-flammable substitutes, and more job sites set strict policies on propellant use. It isn’t just about ticking boxes for insurance anymore; a single fire or explosion brings consequences too big to ignore. Families and workers remember those accidents for years.
Paying attention isn’t complicated—it’s just not optional. Knowing the fire and explosion risks of 1,1-difluoroethane means no surprises. Spotting a can on a desk or in a maintenance closet is a reminder: ordinary products sometimes pack more power than we expect.
Clear Eyes on a Familiar Chemical
1,1-Difluoroethane shows up more often than people realize. You see it in canned air dusters, used by office workers to clean keyboards. Some HVAC folks run into it as a refrigerant. People trust these products because they hit store shelves and rarely cause trouble in daily routines. The truth is, this chemical demands more respect than it gets, especially when safety gets ignored for the sake of convenience or curiosity.
Health Risks Deserve Real Caution
I’ve spent enough time in shops and tech labs to see what carelessness looks like up close. Open a canister of the stuff indoors and you’ll quickly notice how easily the fumes hang around. These vapors can replace oxygen. One deep inhale in a small room leads to dizziness, a confused head, or, in extreme cases, unconsciousness. Some teenagers try to get high with it and wind up in the ER. The risk of cardiac arrhythmia—a sudden, life-threatening heart condition—skyrockets after inhalation abuse. There’s nothing harmless about breathing 1,1-difluoroethane.
Frostbite often gets overlooked. The compressed liquid blasts out cold enough to freeze skin on contact. Trust me, you’ll remember the pain of skin stuck to a frosty metal nozzle. Burn scars often follow.
Fire Hazards Go Farther Than People Assume
Commercials may not say it, but this stuff burns easier than most users imagine. Fluorinated gases often land in the non-flammable basket; that is not the case with 1,1-difluoroethane. Sparks from static electricity or a light switch can ignite concentrated vapors. I’ve seen the aftermath—a melted piece of equipment and an unmistakable chemical stink that lingers for hours. Fire marshals have plenty of stories about incidents in workshops where someone took shortcuts while handling these cans.
Solid Steps Everyone Should Take
Solid safety starts with fresh air. Keep windows open. Run fans to move the fume out. Spraying in a closed closet or an unventilated car turns routine cleaning into a hazard. At work, local exhaust and extraction arms help clear the air. Eye protection and gloves aren’t just for show. Chemically resistant gloves stop frostbite, especially on the fingertips where nerve endings scream the loudest after a slip-up. Protective goggles stand between sensitive eyes and a burst of aerosol or splashed liquid.
Static sparks sound like a detail, until they turn major. Workers using this chemical near electronics should make sure every surface gets grounded. Wires from a wrist strap to a grounded outlet do the trick. No hot surfaces, open flames, or smoking near the spray. Just a can and a heater on the same desk spells trouble, every time.
Storing and Disposing with a Little Foresight
Store 1,1-difluoroethane away from warmth or sunlight. Compressing it inside hot cars or next to radiators increases pressure and risks a leak or explosion. Tossing empties into regular trash cans ignores possible leftover pressure. Many recycling programs treat these canisters like paint thinners or propane. Take a little extra time to look up local hazardous waste rules or ask recycling centers nearby. Every community has a slightly different take but letting the experts handle it beats a can exploding at a landfill or on a trash truck.
Setting the Bar Higher for Routine Safety
I’ve watched enough friends and coworkers brush off warnings because they think the chemical is trivial. The risks demand respect, not fear. Training, clear instructions, and basic gear lower the odds of an accident. One slip leads to an ambulance or a fire. Sound habits take no extra time once they become second nature. Handling 1,1-difluoroethane safely means staying sharp, knowing the facts, and treating every can with a steady hand and a little humility.
Why 1,1-Difluoroethane Pops Up in Unexpected Places
I’ve seen the cans lining hardware store shelves—air dusters for keyboards, propellants in some spray cans, and even refrigeration systems use 1,1-difluoroethane. It’s a favorite for manufacturers, doesn’t catch fire as easily as other chemicals, and it clears out the gunk from tiny spaces. But many folks don’t realize what they’re getting exposed to beyond the dust-free computer screen.
What Happens in the Body After Exposure?
Take a breath around a freshly-sprayed keyboard, swipe an aerosol over your stovetop—most won’t feel anything at all. But inhaling 1,1-difluoroethane is a very different story than sniffing marker fumes as a kid. Direct exposure, especially from “huffing” as a recreational high, floods the lungs with a chemical that the body simply can’t deal with. People have blacked out, lost basic muscle control, and in severe cases, died from heart problems triggered by the chemical.
Medical literature over the past decade has recorded sudden cardiac arrest linked to 1,1-difluoroethane sniffing. The chemical scrambles signals in the heart. Even small doses can mess with the way heart cells communicate—so one unlucky dose could set off a deadly rhythm without warning. And it doesn’t take much; unlike some drugs where risks add up over time, a single exposure at the wrong moment can turn serious quickly.
Risks That Don’t Come With a Warning
The flashiest cases involve intentional misuse, but accidental inhalation matters, too. In poorly ventilated rooms, long workdays around aerosol propellants could bring nausea, lightheadedness, and trouble focusing. Some folks end up with frostbite if the expelled gas hits bare skin. Even if it seems like small-time exposure, there’s no guarantee that days or weeks of “just a little” will stack up safely.
The Environmental Protection Agency and the Centers for Disease Control have flagged 1,1-difluoroethane for causing central nervous system effects. Dizziness, confusion, or headaches after cleaning the office? That’s not just “having a long day”—it could be the chemical at work. Most people don’t suspect a can of spray could be behind mysterious symptoms, which keeps accidents under the radar.
Cleaners, Hobbyists, and Everyday Folks Deserve Better Information
Few products on the store shelf list health dangers in bold print. Warnings often hide in the fine print, and nobody hands out respirators with a can of electronics duster. That leaves workers, hobbyists, and even teens at risk. Prevention starts with honest labeling and training, especially for janitors, IT techs, and anyone cleaning offices, factories, or their own desks.
Public health policies haven’t caught up. Schools and workplaces need better safety talks—not just about the basics, but about the heart dangers and long-term risks. Basic steps, like using chemicals outdoors or in rooms with windows cracked open, go a long way. Proper storage and disposal keep the stuff out of the hands of kids eager for a cheap thrill.
Looking Ahead
Cities and businesses can’t afford to shrug off the hidden dangers of common chemicals. Regulators must demand clearer warnings—big, plain words on the front of every can. Doctors should know to ask about everyday product exposure when strange symptoms crop up. If enough people talk honestly about the risks, we can shrink the blind spot around 1,1-difluoroethane for good.
Storing Chemistry’s Wild Card
Most folks would never think about how to store 1,1-difluoroethane, but skip a few corners and this small oversight could set the stage for trouble. I once saw a forgotten canister leak in the back of a hot storeroom. Let’s just say no one enjoyed finding out it was both flammable and heavy on the lungs. Proper storage starts with simple, sturdy basics: Keep this gas away from heat, sparks, or anything ready to ignite. Steel cabinets marked for hazardous materials punch above their weight here, keeping the cylinders away from damage or tumbled piles of boxes. Ventilated, cool space keeps the pressure steady, and that reduces the risk of leakage or accidental discharge. A dry storage area also wards off corrosion on metal containers, which means no surprise leaks.
Labeling can feel tedious, but clear labeling prevents a small mistake from becoming a headline. Don’t set these containers anywhere near oxidizers or anything reactive. Let’s remember: 1,1-difluoroethane doesn’t just vanish once stored. It’s a volatile gas with a flashpoint below freezing, so even the smallest accident has the potential to escalate fast.
Health and Planet: The Stakes Run High
Plenty of folks use 1,1-difluoroethane. It’s found in air dusters and refrigerants. Inhaling this chemical for a quick high, “huffing,” shakes up lives with addiction or even heart failure. Stories from first responders regularly remind me how dangerous it can be in the wrong hands. Mishandling doesn’t just threaten users—improper storage and disposal can put entire neighborhoods at risk. Breathing the fumes can push out oxygen, leading to drowsiness or worse, and releasing it into the air chips away at the environment little by little.
Regulations help, but common sense packs an extra punch. Research out of the EPA and occupational health groups stresses that 1,1-difluoroethane is no friend to clean air, and big leaks could amp up greenhouse gas levels. Keeping storage tight is more than rule-following—it’s looking out for the planet and those who live on it.
Getting Rid of What’s Left
Too many times, I’ve seen cans tossed in the regular trash or out by the curb. What happens next stinks—literally and figuratively. Leftover gas escapes, wrapping landfill workers or pick-up crews with a cloud of risk. The right way involves local hazardous waste collection, where folks know how to drain, neutralize, and recycle. Many towns host regular take-back days for household chemicals. It takes a little planning, but the peace of mind is worth it.
Breaking open containers or burning them outside sounds easy, but it’s a shortcut to disaster. Open burning spreads chemicals into the air. A tossed can near a flame could mean a fireball nobody saw coming. At my last job, a colleague tried burying old refrigerant cans—a practice both illegal and surprisingly common in out-of-sight, out-of-mind operations. The truth is, land and water never forget. Groundwater contamination sticks around, and that’ll show up years later in a well or vegetable patch.
Looking Ahead
While safety training helps, people need real-world reminders. Keeping a clear inventory, separating chemicals, and always sending leftovers to the right place make the difference. It’s doing the work up front that keeps families, workers, and the climate out of harm’s way. That’s a commitment that matters as much for small garages as it does for big factories.