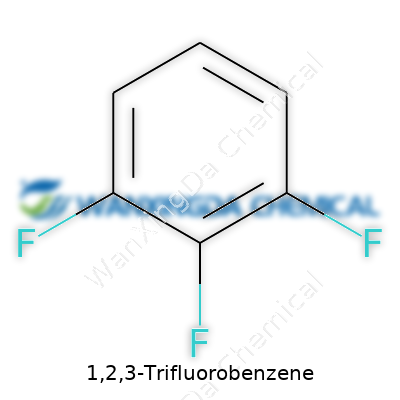
1,2,3-Trifluorobenzene: A Deep Dive into Its Place in Modern Chemistry
Historical Development of 1,2,3-Trifluorobenzene
Few molecules tell the story of changing chemical priorities like 1,2,3-trifluorobenzene. Decades ago, research focused on simpler aromatic compounds; then the mid-20th century brought a surge of interest in fluorinated aromatics. Synthetic chemists wanted greater chemical stability, metabolic resistance, and electronic effects for new pharmaceuticals and polymers. This particular trifluorinated benzene appeared on the lab bench as fluorination chemistry grew more precise. Labs in North America and Western Europe raced to produce specific trifluorobenzenes for use as molecular building blocks in dyes, agrochemicals, and drug intermediates. Today, it’s rare to find synthetic or medicinal chemists unfamiliar with these versatile molecules.
Product Overview: The Role in Industry
1,2,3-Trifluorobenzene often serves as a benchmark compound. Those three fluorine atoms fundamentally shift the electronic nature of the aromatic ring, which leads to unusual reactivity. In the hands of an experienced chemist, it can give rise to tailored molecules with improved pharmacokinetics or industrial robustness. Chemical companies look to such trifluorinated benzenes as key intermediates for the manufacture of advanced materials or as probes to study enzyme mechanisms. Specialty chemical suppliers tend to focus on high-purity batches since trace contaminants can cause major headaches in pharmaceutical research.
Physical and Chemical Properties
Pouring 1,2,3-trifluorobenzene into a flask, you’ll notice its clear, colorless appearance and characteristic sharp odor. The presence of three fluorine atoms in adjacent positions on the aromatic ring increases volatility relative to many other substituted benzenes. Its boiling point sits just above room temperature, making it manageable yet prone to evaporation without proper care. Chemical inertness stands out, driven by strong C-F bonds that resist most forms of attack. The fluorines draw electron density away, making electrophilic aromatic substitution reactions sluggish, but nucleophilic aromatic substitutions gain an edge thanks to the activated leaving positions.
Technical Specifications and Labeling
For anyone handling this compound, the label often confirms trifluorination at the ortho and meta positions, and you might spot references to precise GC purity percentages. The labeling sometimes includes batch numbers to trace back any impurity issues. In my own lab, we look for IR and NMR spectra confirming all three fluorines have landed where intended, avoiding isomer confusion that can derail downstream chemistry. Safe storage demands airtight containers, away from direct sunlight or heat, to prevent slow loss through volatilization.
Preparation Method
Building 1,2,3-trifluorobenzene involves strategic thinking and good process control. One route starts with benzene and leverages selective electrophilic fluorination using reagents like Selectfluor or elemental fluorine under controlled conditions. Direct fluorination rarely delivers only the 1,2,3-isomer, so chemists lean on multi-step synthetic routes—introducing fluorine atoms one at a time, often via diazonium salt intermediates. Another approach uses nucleophilic fluorination, swapping out halide groups on suitably substituted benzenes for fluoride. In commercial applications, yield, selectivity, and waste minimization steer route selection.
Chemical Reactions and Modifications
This compound offers a launching pad for creating more complex molecules. The fluorines subtly direct further substitutions on the aromatic ring, supporting fine-tuned synthesis. Nucleophilic aromatic substitution enables the introduction of amino, alkoxy, or thiol groups by replacing one of the fluorines under the right conditions. Chemists have explored hydrogenation, halogenation, and cross-coupling strategies, broadening the synthetic utility of the molecule. The interplay of electron-withdrawing effects allows for regioselective functionalization rare in less-modified benzenes, which is a core reason why it remains a favorite in medicinal chemistry circles.
Synonyms and Product Names
1,2,3-Trifluorobenzene sometimes goes by different names in catalogs and literature. Common synonyms include m-Trifluorobenzene or benzene, 1,2,3-trifluoro-. Some research papers use non-systematic terms, but the CAS number often provides clarity in the midst of naming confusion. This helps sharpen communication when handling regulatory documents or ordering from suppliers.
Safety and Operational Standards
I treat 1,2,3-trifluorobenzene as I would any low-boiling organic liquid: gloves, goggles, fume hood at all times. The compound’s volatility and mild toxicity require careful handling to avoid inhalation or skin contact. Long-term exposure data remain limited, but fluorinated benzenes generally carry risk of respiratory irritation. Disposal aligns with hazardous organic waste protocols, since standard drains or trash cannot neutralize organofluorine content. The GHS labeling flags flammability and acute toxicity, so planning procedures around emergency showers and spill containment remains good practice. Cross-training all lab personnel on safe handling and emergency response holds as much importance as the MSDS itself.
Application Area
Applications for 1,2,3-trifluorobenzene span pharmaceuticals, agrochemicals, material science, and electronics. Medicinal chemists value its use as a core structure in drug leads, leveraging the metabolic stability imparted by fluorines. In crop protection research, it serves as a template for more effective agrochemicals, as fluorination often improves potency and environmental fate. The electronics field uses it for specialty polymers due to unique dielectric properties, and certain liquid crystal formulations also incorporate trifluorinated aromatics. Each of these areas demands consistent quality, tight supply chain controls, and transparent communication from suppliers to end users.
Research and Development
The research landscape for 1,2,3-trifluorobenzene reflects a broader quest for selective functionalization of fluorinated aromatics. Friedel-Crafts reactions rarely work on such electron-poor rings, which has driven scientists to innovate using metal-catalyzed cross-couplings or photochemical techniques. I’ve followed recent studies exploring these newer methods, which promise greener processes and better atom economy. High-throughput screening tools now accelerate the discovery of novel derivatives, expanding this compound’s impact far beyond classic applications.
Toxicity Research
Reliable toxicity data for 1,2,3-trifluorobenzene remain somewhat limited compared to more commonly used aromatic chemicals. Animal studies so far suggest low acute toxicity, but the risk grows with dose, and chronic exposure data don’t tell a complete story. What stands out in fluorinated organics is their persistence—once released, they rarely break down quickly in the environment. Researchers need to keep pushing for long-term and ecosystem-level toxicity data, especially since demand for fluorinated compounds continues to rise. Industry and academia have opportunities to collaborate, investing in better models and safer design principles early in the research pipeline.
Future Prospects
Looking ahead, demand for trifluorinated benzenes like 1,2,3-trifluorobenzene shows no sign of slowing. As designers of pharmaceuticals and advanced materials search for molecules with improved performance and stability, the value of well-understood aromatic scaffolds grows. Efforts to improve green synthetic routes, reduce hazardous byproducts, and close the loop on solvent recovery will only get more intense. Regulatory scrutiny for all things fluorinated already shapes research priorities, pushing chemists to weigh performance against sustainability and safety. In this space, chemists with a deep understanding of trifluorinated arenes will continue to shape the next wave of functional molecules—always balancing creativity with caution.
Not many folks outside chemistry circles chat about 1,2,3-trifluorobenzene, but those who work with advanced materials or pharmaceuticals have seen the impact of this small molecule. Three fluorine atoms attached to a benzene ring create more than just a fun name. They unlock a set of properties that bring real benefits to the table.
Fueling Innovation in Pharmaceuticals
Chemists gravitate toward fluorinated compounds for a reason—these molecules stand up surprisingly well under tough conditions. In drug discovery, a single tweak on a molecule can make or break a medicine. Introducing fluorine atoms can change how a candidate drug behaves in the body, affecting everything from how quickly it gets absorbed to how long it lingers in the bloodstream.
From what I’ve seen in published research and from speaking with chemists, trifluorinated benzenes offer a way to boost stability and tune biological activity. Think of 1,2,3-trifluorobenzene as a building block. Medicinal chemists use it while piecing together more complicated molecules. It can slip into a larger molecular scaffold and deliver those sought-after tweaks in drug metabolism or target binding. There’s documented use of this compound in developing new antivirals and in cancer research pipelines.
Advanced Materials: Beyond the Lab Bench
Ever handled a phone or laptop? High-performance plastics and liquid-crystal displays owe thanks to specialty chemicals like this. The trifluorinated ring isn’t there for show—it delivers resistance to heat and chemicals in ways plain benzene can’t. The push for electronics that last longer and stay reliable in harsh settings keeps turning up the value of specialized aromatic compounds like 1,2,3-trifluorobenzene.
Electronics manufacturers tap into these molecules as part of the complex mixtures for display technologies and dielectric materials. The fluorine atoms help increase stability and prevent breakdown from light or heat. That reliability matters when devices travel all over the world and face everything from tropical humidity to cold winters. Every chemist working on a new generation of flexible screens or tougher circuit boards keeps an eye out for materials that deliver both toughness and precision, and that's where this trifluorinated compound stands out.
Agrochemicals: Helping Crops Grow Stronger
Fluorination brings big changes to agricultural products as well. Pesticides and herbicides often benefit from enhanced persistence and unique ways of interacting with unwanted pests. 1,2,3-Trifluorobenzene helps formulate new crop-protection compounds that plants tolerate but bugs and weeds don’t. Farmers get more selective action and better yields, while companies dig into ongoing studies to address concerns about environmental buildup.
What Comes Next?
The chemistry isn’t cheap, and there are questions about making and disposing of fluorinated compounds responsibly. Wealth of studies point out the need for better recycling and waste management. Some industry groups and researchers now chase greener pathways to these molecules—everything from milder reaction conditions to the search for bio-based feedstocks. It’s a tough road, but every move toward safer, more sustainable production clears the way for chemists to use powerful compounds like 1,2,3-trifluorobenzene with fewer tradeoffs.
My own experience with industry-university partnerships makes one thing clear. Progress doesn’t just come from discovering new reactions; it comes from responsible use of these tools, open communication, and a shared drive to keep science moving forward, but not at the expense of environmental stewardship. This is how chemistry builds trust—and secures its place in shaping the future.
Looking Beyond the Formula
Growing up around a family business that handled dyes and pharmaceuticals, I learned early that chemistry isn’t some distant subject best left to folks in lab coats. Ask any synthetic chemist about 1,2,3-Trifluorobenzene, and you’ll see the spark of recognition. Its chemical formula—C6H3F3—makes it a classic example where subtle molecular tweaks create serious impact. The molecular weight clocks in at 146.09 g/mol, compact but potent. Some people hear these numbers and shrug. Let me tell you, they matter more than you think.
Why This Molecule Matters
Chemical structure shapes everything. The trifluoro arrangement in this benzene ring transforms its reactivity and behavior. Fluorine, which ranks among the most electronegative elements on the periodic table, doesn’t just sit there. It pulls electrons, alters ring dynamics, and resists many forms of breakdown. As a result, 1,2,3-Trifluorobenzene becomes useful in making pharmaceuticals and specialty materials. The market for high-performance organic compounds has boomed in the past two decades, with companies trying to pack more punch into smaller, more controllable molecules. Fluorinated aromatics—like this one—frequently serve as key intermediates or building blocks.
From Synthesis Lab to Industry
Behind every clean bottle of reactant in a lab, there's a backstory. Industry chemists pursue efficiency, safety, and environmental sense. Making trifluorinated benzenes isn’t trivial. Old-school processes loaded benzene derivatives with hazardous reagents and generated piles of toxic byproducts. Newer routes include catalytic, selective fluorination or clever use of less aggressive agents. There's been a real push for sustainability because waste is costly—financially and environmentally. The industry has grown wiser, favoring atom economy and recyclable solvents. My uncle, a process engineer, once said, “What you throw away today could bankrupt you tomorrow.” He wasn’t wrong.
The Role of Fluorinated Compounds in Progress
Most breakthroughs in drugs for hard-to-treat diseases come from fine-tuning molecules to control how they interact with the body. A few fluorines in exactly the right place can boost drug stability, absorption, and effectiveness. That’s not hype; that’s been proven by the explosion of new fluorine-containing therapies. Electronics also benefit from these molecules. As devices shrink, durability and reliability take priority. Polymers, lubricants, and specialized coatings drawn from precursors like 1,2,3-Trifluorobenzene keep manufacturing on the cutting edge.
Addressing Challenges and Moving Forward
Balancing chemical innovation with environmental and human health remains critical. Safety data on many fluorinated organics is still developing. Companies face pressure to track long-term impacts and prevent bioaccumulation risks. For every neat chemical that makes life easier, someone has to think about accidental releases and responsible disposal. One solution is open collaboration—sharing data, best practices, and process improvements instead of keeping everything behind closed doors.
Modern chemistry asks us all to pay attention. A molecule’s formula and weight are only the beginning. Behind these numbers lies a world of discovery, risk, and potential that stretches from the lab bench to your everyday life. The story of 1,2,3-Trifluorobenzene offers a glimpse into how small changes ripple through industries and shape tomorrow’s solutions.
Why Storage Details Matter More Than You Think
1,2,3-Trifluorobenzene might sound like just another item in a catalog, but mishandling even small bottles can turn routine lab work into a mess—or worse, a health risk. Anyone working with chemicals learns early that ignoring basic safety can be costly. This compound isn’t as dramatic as hydrofluoric acid, but its storage and use deserve real attention.
Room Temperature Isn’t Always “Safe Enough”
Labs and facilities often keep many chemicals on shelves, and it’s tempting to treat them all the same. 1,2,3-Trifluorobenzene is volatile. Its fumes don’t just vanish harmlessly. Without tight lids and good ventilation, those vapors build up. Over the years, I’ve seen sealed bottles bulge and leak because staff overlooked proper closures. This liquid wants a spot away from heat, sparks, and busy walkways. Not every storeroom meets those needs. Chemical-resistant cabinets, away from sunlight and ignition sources, make a basic difference.
Personal Protection Isn't Optional
Shortcuts add up, especially in shared spaces. Gloves and eye protection should come out the moment this liquid appears at the bench. I’ve handled organics a thousand times, but the moment you forget, skin exposure or accidental splashes remind you why PPE is a must. Any spill on the hands can cause irritation, and those fumes aren’t friendly to the lungs. Fume hoods matter. Even if a task looks simple, open bottles frees up enough vapor to impact air quality.
Segregation: No Guesswork with Chemical Neighbors
Not all chemicals play nicely. 1,2,3-Trifluorobenzene should never share a shelf with oxidizers or strong acids. Mixing can start a chain reaction. I’ve witnessed poorly organized storage cause scares and even fire department calls. Years of practice make it second nature to check labels and chemical compatibility charts. Some labs lay out colored stickers for quick checks. Every bottle’s neighbor counts.
Containers: Not Every Bottle Makes the Cut
Glass tends to work best for storing aromatic compounds like this. Metal containers react over time, and leaky plastic can turn minor leaks into headaches. Reliable seals matter most. I’ve seen entire shelves cleared over a single crack in a cap. Never decant unless new containers stand up to harsh liquids. Misjudged transfers lead to losses and exposure.
Disposal Closes the Safety Loop
Disposal often gets rushed. Pouring leftovers down the drain is illegal and pollutes water supplies. Most places want organic waste collected in marked bottles, kept away from acids. Teams must know where disposal lanes run, who empties them, and which vendor removes hazardous waste. Incineration or approved chemical breakdown is the safest exit route.
Better Practices for Fewer Incidents
Good habits save money, product, and time. Training matters as much as gear. Regular reviews, safety drills, and labeling audits catch problems before they become violations. I’ve seen attentive staff spot cracked containers before leaks start. Even one thorough walkthrough a month can prevent a lot of pain. Chemical safety always rewards those who don’t cut corners.
Everyday Lab Hazards Hide in Plain Sight
Chemistry promises excitement, but it doesn’t always hand you a warning label you can spot from across the lab. 1,2,3-Trifluorobenzene doesn’t carry the drama or danger of more notorious chemicals, but taking it lightly proves risky. A chemical doesn’t have to come with a skull and crossbones to deserve your respect. Decades around organic labs have taught me that even substances that seem straightforward often bring hidden dangers. This compound, with its three fluorine atoms, illustrates why you don’t put on safety gear for show—you do it for your future health.
Don’t Skip the Gloves, Don’t Skip the Mask
Handling 1,2,3-trifluorobenzene usually means exposure as a colorless liquid. You might catch a whiff of its sharp, sweet odor, but trust me: you don’t want to breathe it in. Benzene rings have a way of sneaking up on human biology. Prolonged inhalation or skin contact can irritate your eyes, lungs, or cause headaches. The real problem comes with routine—someone adjusts a hood too late or leaves a vial open out of habit. After years around solvents, I learned not to let my guard down just because a compound isn’t labeled “toxic.” Acute exposure may not seem like much, but I’ve seen enough irritant rashes and raw throats to last a lifetime.
Environmental Safety Isn’t Optional
Lots of people forget about the waste. Fluorinated aromatics don’t break down easily. Labs that pour used 1,2,3-trifluorobenzene down the drain are not just breaking rules—they’re passing today’s risks into tomorrow’s water and food. In some countries, strict laws control fluorochemical disposal for good reason. Trace organofluorines in the environment may affect aquatic life or show up in groundwater. Fact: proper disposal means marked, sealed waste containers, not wishing the problem away with running water.
What I’d Like to See in the Lab
No one expects a novice to memorize every hazard. It’s easy to forget the basics in a rush. We should place laminated safety instruction cards near chemical shelves—that’s a solution that stops the hurried shortcut. Mandatory training on the real-life consequences of solvent exposure would help, not just another online quiz. I’d encourage student chemists to work in pairs when handling organofluorines—at least until confidence meets competence. We should keep NIOSH-approved respirators handy in addition to the usual gloves and goggles, since organic vapors stay in the air, long after a spill. Whenever we buy new solvents, the safety data sheet needs to be on permanent display—not filed away in an unread binder.
The Stakes Go Beyond the Lab Door
Safe handling isn’t just about ticking boxes or following orders. Responsible chemistry keeps people healthy today and protects the environment for the long haul. I’ve met seasoned researchers with diminished lung capacity from basic negligence back in college, and seen environmental data showing traces of persistent fluorinated organics in streams near research campuses. That ends when we treat every bottle with the care we use for the most dangerous vial. Gloves, goggles, lab coats, local exhaust ventilation, airtight storage, and waste discipline—those habits protect everyone, not just the current team. Once you make safety second nature, you cut away the risk—quietly, reliably, every single workday.
Everyone who’s ever spent time working in a lab, managed chemicals on a shop floor, or even dabbled in DIY projects with industrial materials knows the routine: check the label, glance at the hazard symbols, and hope the paperwork tells you what you’re really up against. Now, with 1,2,3-Trifluorobenzene, things aren't so different, but the stakes still add up. Chemical safety doesn’t run on trust or guesswork. It demands reliable data, shared openly and clearly.
Trustworthy Sources Still Do the Heavy Lifting
Safety data sheets, or SDS, aren’t just formalities. For chemists and safety officers, seeing the standard sixteen sections of an SDS means answers: what temperature sparks a fire risk, how to treat a spill, what personal protective equipment you really need. Any time that information is missing, mistakes get made — sometimes quickly, sometimes quietly, but always with consequences.
In my years rounding up chemicals for research and teaching, a handful of sources always delivered. Reaching out straight to the supplier – big names like Sigma-Aldrich, TCI, or Alfa Aesar – got results. If you’re buying 1,2,3-Trifluorobenzene from an established vendor, their website should carry a downloadable SDS. No chemistry background necessary; a search bar, the right chemical name or CAS number (for this compound: 2836-89-5), and you’ll usually find the file ready to go. Legislation in the US and EU requires this level of transparency.
For those outside the standard supply routes – maybe a small start-up, a teaching lab on a shoestring, or someone inheriting legacy chemicals – things get trickier. This is when I’ve turned to public chemical safety databases. Both Chemical Safety Library and PubChem include hazard sections and, sometimes, downloadable data sheets. These resources draw from public records and regulatory filings. They tend to lag behind supplier databases on the latest revisions but cover most common uses and exposures.
The Gaps Still Deserve Attention
An honest look reveals gaps in SDS access. Dealers in certain regions play fast and loose with regulations. Smaller start-ups popping up on global e-commerce platforms may not provide proper safety paperwork, leaving buyers unprotected. Even local suppliers sometimes cut corners and send high-quality product without a word about safe handling. In these cases, science educators like me got used to chasing down safety information from public regulators or calling up industry colleagues for help.
Legal access to SDS isn’t a “nice-to-have”—it’s mandated by law in most countries. OSHA in the United States and REACH in the European Union both enforce it strictly. If businesses or buyers choose to look the other way, they risk regulatory penalties, workplace accidents, or worse. I’ve seen well-meaning technicians handle chemicals with outdated or missing data sheets and land in the emergency room. We all owe it to each other to keep safety up to standard.
Solutions Lie in Honesty and Shared Responsibility
Sourcing and sharing SDS for chemicals like 1,2,3-Trifluorobenzene boils down to responsibility. Big suppliers already set the benchmark with transparent, online access. Small operators and new e-commerce dealers should meet the same bar. End users can push back — request the SDS before purchase and refuse to work with vendors who won’t provide it. Most importantly, sharing accurate safety data builds a culture where labs, plants, and classrooms stay safe. That culture starts with asking for — and expecting — clear answers, not hiding behind paperwork.