Editorial Commentary: The Story and Future of 1,3-Difluoroacetone
Historical Development
Just a few decades ago, the fluorinated building blocks that drive modern synthetic chemistry were either scarce or painfully expensive. Organic chemists sought out ways to add fluorine atoms to familiar carbon skeletons, looking for harder-hitting pharmaceuticals, enhanced agrochemicals, and materials with sharper properties. During that search, 1,3-difluoroacetone slowly entered the scene. Its development didn’t arise from one landmark paper, but from a series of incremental steps as research groups recognized the power of introducing multiple fluorine atoms into small, reactive molecules. Over time, this compound moved from obscure curiosity to a structure frequently found in reaction schemes published in chemical literature. The rise in demand for specialized fluorinated reagents drove larger batch synthesis, improved purification methods, and deeper investigation into its reactivity profile.
Product Overview
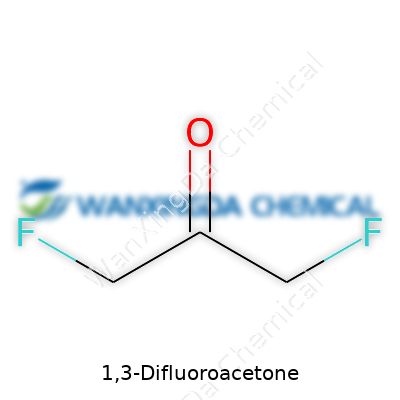
1,3-Difluoroacetone tells a clear story through its structure. Two fluorine atoms, starkly different from hydrogen or other halogens, transform this simple ketone into something robust yet reactive. It sits among a class of difluorinated carbonyl compounds that bring unique electronic twists to reaction pathways, letting chemists build more complex molecules with distinct electronic features. Anyone who studies modern organic synthesis quickly bumps into molecules like this—compact, potent, and broadly useful. In the market of specialty chemicals, it stands apart for both its volatility and its ability to introduce difluoromethylene groups into everything from candidate drug molecules to specialty polymers.
Physical & Chemical Properties
Unlike conventional acetone, swapping hydrogens for fluorines fundamentally alters the landscape. 1,3-Difluoroacetone typically presents as a clear liquid, with an acrid scent and a boiling point notably higher than its parent molecule. Those fluorines not only stiffen the molecular backbone but also pull electron density away, which means this chemical resists both oxidation and reduction more than many non-fluorinated ketones. Its solubility patterns change as well, often limiting water miscibility but showing a knack for dissolving in organic solvents where fluorinated chains thrive. The high electronegativity of fluorine atoms modifies not just the reactivity, but almost every aspect of handling and storage, forcing chemists to think twice about compatibility and containment.
Technical Specifications & Labeling
Commercial bottles carry clear hazard warnings, with proper regulatory labeling for flammability and toxicity. Density and purity matter greatly; minor impurities—especially those affecting the difluoro motif—can skew experimental outcomes in pharmaceutical discovery. Most suppliers describe the chemical as being over 98% pure, but anyone running a synthesis knows a GC-MS analysis is worth more than a thousand assurances. Labels rarely exaggerate: respect for volatility, a tight cap, and cold storage make for good practice.
Preparation Method
Bringing 1,3-difluoroacetone to life in the lab takes more than a simple fluorination step. Early methods relied on harsh reagents like anhydrous hydrogen fluoride, but advances have softened the route, often using milder fluorinating agents such as diethylaminosulfur trifluoride or specialized fluoride sources. The typical approach starts with a 1,3-dicarbonyl skeleton and methodically swaps out hydrogens for fluorines under strongly controlled conditions. Yields climb with steady patience, careful temperature control, and vigorous stirring. Even today, scale-up brings surprises, as heat buildup and byproduct formation can easily derail large-scale runs, so process chemists keep a close eye on reaction profiles.
Chemical Reactions & Modifications
Any chemist seeking to modify a molecule quickly learns that fluorines make the carbon chain more stubborn and selective in reactions. The presence of two fluorine atoms locks in a specific geometry and pushes most nucleophilic attacks toward the central carbonyl. This selectivity enhances its use in enolate chemistry, where difluoroacetone reacts cleanly with a variety of nucleophiles. It tolerates Grignard reagents, acid-catalyzed condensations, and even undergoes subtle reductions without devolving into a mess of side products. In the hands of a skilled researcher, it becomes the launching point for a spectrum of difluoromethylated heterocycles and intermediates. Such reactivity supports not just academic curiosity, but industry’s hunger for patented molecules with small but critical fluorine tweaks.
Synonyms & Product Names
In scientific circles, calling things by their IUPAC names may be technically correct, but everyday conversation usually leans toward the familiar. Here, “1,3-difluoro-2-propanone” often pairs with its shorter trade name or catalog number. Literature occasionally lists variants like “difluoroacetone” or “DFA,” subtle reminders that shorthand grows as fast as research spreads.
Safety & Operational Standards
Handling this chemical takes experience and common sense. Its vapors irritate eyes, nose, and throat, so even a modest spill orders a retreat to fresh air. Work behind a ventilated fume hood is standard. Nitrile or even fluoroelastomer gloves help, since fluorinated ketones can dissolve right through lesser materials. Laboratories that routinely deal with such chemicals post clear signage, keep spill kits at hand, and lay out standard operating procedures that leave little to chance. Waste collection follows local and international rules for halogenated solvents, reflecting both toxicity and environmental persistence. Standard training on emergency eye wash and shower use gets plenty of emphasis for new team members.
Application Area
1,3-Difluoroacetone’s strongest pull for research and manufacturing lies in its role as a building block for advanced molecules. Medicinal chemists prize its difluoromethylene group, often leveraging it to improve metabolic stability in candidate drugs, since fluorine atoms tend to slow down oxidation inside the body. Agrochemical developers follow suit for similar reasons, aiming for longer-lasting field performance. Material scientists use this compound to give polymers low surface energy, resulting in water repellent coatings, high-performance lubricants, and even medical device coatings that shed proteins. More companies now invest in cataloging new derivatives, driven by market demand for specialty reagents and novel scaffolds.
Research & Development
Across the industry, teams keep turning over new stones in the quest to push 1,3-difluoroacetone’s reactivity further. Focused research now targets safer and greener synthesis pathways—both a nod to growing regulatory pressure and to the real dangers of older, harsher fluorinating agents. Computational studies help to predict new reaction pathways, providing chemists with digital insight before committing costly batches of material to the bench. Cross-disciplinary teams often join forces: synthetic chemists work hand-in-hand with pharmacologists or polymer engineers to explore untapped potential. Across scientific journals, there’s a growing stream of literature picking apart every nuance: reactivity, byproducts, even alternative uses in catalysis and imaging.
Toxicity Research
Safety officers and environmental scientists keep close tabs on toxicity, both acute and chronic. Inhalation poses clear respiratory risks, but chronic exposure brings more complex concerns, especially given the stubbornness of fluorinated compounds in living systems. Some in vivo studies indicate potential for organ damage at high doses, though full toxicokinetic profiles remain an area of active research. Regulatory agencies watch for new findings that may push revisions in workplace exposure limits. Operators learn how to balance productivity and caution, always assuming the least exposure is the safest target. This area underscores the necessity for continual review, as each generation of data builds a stronger case for best practices.
Future Prospects
The road forward for 1,3-difluoroacetone points not just toward more innovation, but toward more responsible stewardship. Demand for fluorinated building blocks keeps climbing, thanks to pharmaceutical and materials breakthroughs that depend on unique properties like metabolic resistance and thermal stability. Practical improvements to synthesis—lower energy requirements, less hazardous reagents—look set to lower both production costs and environmental harm. Researchers keep searching for biodegradable analogs that can perform similar chemical tricks without adding to persistent pollutant burdens. In the hands of a new generation of chemists, this compound may soon find fresh uses in fields like imaging, green chemistry, and even electronics, if current trends continue. The future depends not only on what the molecule can do, but also how responsibly we handle its potential and risks.
A Tool for Modern Chemistry
1,3-Difluoroacetone doesn’t show up in splashy headlines, but in lab settings and industry, it quietly gets a lot done. Chemists respect its reactivity. I’ve seen people chase after the latest catalysts or green solvents, but when you want to add some kick to a synthesis involving fluorine, this compound deserves a spot on the shelf. Its structure, with two fluorine atoms flanking a carbonyl, gives it unique properties that keep it in the running for real chemistry work.
Pharmaceuticals: Creating New Paths to Molecules
Pharmaceutical labs often turn to 1,3-difluoroacetone for a reason. Fluorine can dramatically shift a molecule’s performance in the body. Adding this element sometimes means tweaking a drug’s absorption, stability, or resistance to being broken down by enzymes. Instead of lengthy multistep routes, chemists with access to 1,3-difluoroacetone can knock out challenging syntheses with fewer headaches. They use it as a building block to craft various heterocycles—think oxazoles, imidazoles, and pyrroles—found in antibiotics, cancer drugs, or antiviral agents. One study showed that swapping in a fluorine often boosts a compound’s oral bioavailability, proving small changes can make big differences.
Materials Science: Laying the Groundwork for Better Polymers
Out in the world of materials, 1,3-difluoroacetone serves as a link to the polymers of tomorrow. Polymer chemists eye it for specialty plastics and coatings because fluorinated materials tend to have super resistance to chemicals, water, or heat. The drive for nonstick or low-friction surfaces starts at the molecular level, and this compound helps stitch those properties right in. While most people only see the outcome—weatherproof cables or membranes in fuel cells—the chemistry starts with something as humble as this molecule. It’s about turning reactivity into practicality.
Agrochemicals: Precision in Pest Control
Modern pesticides and herbicides increasingly rely on smart molecular design. Farmers and field specialists want results but also demand products that break down safely. 1,3-difluoroacetone plays a role by providing fluorinated intermediates that stray less in the environment or linger just long enough to finish their job. A few major agrochemical classes, including some triazoles and pyrazoles, use similar building blocks during synthesis. The goal is safe, predictable action that balances environmental safety with robust performance.
Challenges and the Road Ahead
Every time you introduce something new into a chemical process, you weigh risks. Fluorinated molecules need careful handling. Waste from their production sometimes doesn’t break down as quickly as non-fluorinated analogs. That’s where industry has to step up with greener byproducts, tighter waste management, and better process design. Some research groups are already chipping away at these obstacles, insisting on low-impact reactions and recycling wherever possible.
Why It Matters
The real story with 1,3-difluoroacetone is about access. Bringing unique reactivity to chemists’ fingertips means more options to solve tough problems in medicine, farming, and materials. Picking the right tools—and using them responsibly—makes innovation possible without losing sight of health, safety, or common sense. For anyone who’s ever watched lab work go much smoother because the right building block was stocked up, the worth of a compound like this isn’t theoretical.
Why Chemistry Details Still Matter
Chemistry doesn’t always win the prize for most exciting conversation starter, but knowing what lies behind a name like 1,3-Difluoroacetone can make a big difference. Experience has shown me that when you really dive into the details of a compound—its formula or molecular weight—you get more than trivia. You start to see the connections that drive both lab work and real-world applications.
The Formula: Small Adjustments, Big Outcomes
Roll up your sleeves, and let’s look closely at the formula. 1,3-Difluoroacetone packs a punch with just a handful of atoms: its formula is C3H4F2O. A molecule this small brings fluoro groups—two, to be exact—onto opposite ends of an acetone skeleton. Each group changes the way the molecule behaves, reacts, and even how it can cause trouble or bring about new possibilities in the lab.
In my own work with organofluorine compounds, minor tweaks in formula have thrown off an entire week’s worth of synthesis. Chemistry turns on a dime, especially with halogens like fluorine in play. They may be less flashy than their cousins like chlorine or iodine, but these two fluorines give 1,3-Difluoroacetone its own identity.
A Weight with Meaning
Let’s talk numbers: the molecular weight of 1,3-Difluoroacetone clocks in at 94.06 g/mol. That number unlocks all sorts of calculations, from simple stoichiometry to the more detailed planning that comes up in the middle of a long experimental series. It’s not just digits after a decimal point—it guides ordering, storage, and the precise measuring that keeps experiments repeatable.
I’ve seen chemists spend hours recalculating yields because someone overlooked a decimal point. Memorizing weights and formulas might sound tedious, but it builds a foundation for real trust in data, and makes collaboration across labs a lot smoother. Missing one atom or miscalculating atomic mass doesn’t just throw off a day. It can tank whole batches, send budgets spiraling, or put important health and safety protocols at risk.
Trust, Training, and Better Practices
Every inch of progress in chemical research depends on reliable, up-to-date information. The facts about something like 1,3-Difluoroacetone—right down to its formula and weight—are not just academic trivia. They shape how we handle everything from storage and disposal to reaction planning. If you’ve worked with chemicals long enough, you know the small numbers and letters have the power to steer costly or even dangerous mistakes out of your path.
Access to trustworthy databases and peer-reviewed references makes an obvious difference. I’ve had times when pulling an outdated manual meant starting over. Regulatory standards push for accuracy and transparency, but so does the shared sense of responsibility in any good lab. Better education around the meaning and importance of these basics pays off. Apprentice chemists who start by digging into formulas don’t just sit on the sidelines; they’re safer, more curious, and their troubleshooting instincts get sharper.
Moving Ahead
Chemistry has always leaned on the backbone of clear, consistent documentation. From my experience, the real value in knowing something like the formula and molecular weight of 1,3-Difluoroacetone isn’t just about being “correct”—it’s about making better choices. As science asks more from every compound and every experiment, a small detail brings a big return.
Why We Need Respect for Chemical Safety
Anyone who spends time in a lab or on a production site learns something quickly: mishandling chemicals isn’t just a paperwork problem, it’s a life-and-safety problem. 1,3-Difluoroacetone isn’t a household name, but its hazards deserve just as much respect as any other potent industrial reagent. This compound is volatile and reactive, so getting its storage and handling wrong can invite big trouble.
Practical Storage: No Room for Complacency
With 1,3-Difluoroacetone, there’s no margin for error. The stuff’s volatility means it can turn from a colorless liquid to hazardous vapor before you know it. Leaving it in a warm workspace, a flimsy container, or even under regular room lights can turn a routine task into a safety incident. I remember working late in a university lab where someone left a similar acetone derivative on a countertop. Half an hour later, the room reeked, and alarms blared—reminding all of us why strict storage matters. A proper system keeps chemicals like this in cool, dedicated chemical storage spaces, away from sunlight, heat, and, ideally, anything with an ignition source.
Tightly sealed, chemically compatible containers sit at the foundation of good practice. Materials like glass or seasoned fluoropolymer plastics give better reliability than cheap plastics, which may soften or degrade. Every label needs to stay legible—no reusing old solvent bottles. Good labeling can seem like a chore, but it saves mistakes and sometimes lives.
Handling: More Than Just Gloves and Goggles
Personal experience has taught me never to underestimate fumes from volatile organics. Even one whiff can tell you that regular ventilation doesn’t always cut it. 1,3-Difluoroacetone belongs in a fume hood during any transfer or open operation. Basic gloves and goggles form the first line of defense, but real preparation includes splash-proof goggles, lab coats, and solid training. I’ve seen smart teams throw out gloves after spills, clean glassware twice, and post clear waste disposal instructions—little steps that add up to fewer accidents.
Spill management always matters with hazardous chemicals. Nobody wants to scramble during an accidental knock-over—so it pays to keep spill kits within arm’s reach, absorbent pads ready, and a plan in place before anything leaves the bottle. It helps to walk through potential scenarios as a team: what if the power goes out, what if someone drops a bottle, what’s the fastest exit route? Teams that rehearse these drills respond faster and with less panic.
Why It Matters Beyond the Fine Print
Strict storage and handling stop small mistakes from ballooning. A cracked bottle, a misread label, a ventless room—each seems minor alone, but together they spell disaster. Chemical injuries, costly fire cleanups, and lost research hours all trace back to preventable lapses.
There’s value in building a culture of safety, not just cataloging hazards. Training for new staff, regular audits, and quick access to reliable MSDS resources mean everyone from research interns to experienced chemists keeps safety front-of-mind. The best solutions come from sharing stories, not just checklists—lessons learned the hard way, passed down so the next person doesn’t repeat the same mistake.
1,3-Difluoroacetone isn’t the world’s most notorious chemical, but the attention it demands is non-negotiable. Respect for practical, consistent safety routines gives peace of mind and protects everyone in the line of fire.
Understanding the Chemical’s Presence and Impact
1,3-Difluoroacetone sounds like a name only a chemist would love, but it can find its way into conversations far outside the lab. Synthetic chemicals like this hold real-world consequences. Some sit quietly on shelves, while others demand respect due to their effects on health and the environment. Having worked in a research lab, I’ve learned that unfamiliar chemicals often invite trouble when people take their risks lightly. I remember a time when a forgotten solvent, much less exotic than 1,3-difluoroacetone, caused respiratory irritation for several colleagues. The lesson? Every new compound brings its own baggage.
Toxicity Isn’t Always Obvious
Talking about toxicity, folks often picture skull-and-crossbones labels or thick chemical odors. In reality, some substances do their worst damage invisibly. 1,3-Difluoroacetone fits into that tricky category. Current safety data places it with other difluorinated organic compounds, which can be harmful if inhaled, ingested, or splashed on the skin. Small molecules like this can sneak past natural defenses, sometimes vaporizing readily and entering the body before anyone realizes there’s a problem.
Published research on similar fluorinated ketones points to a range of hazards. Many act as irritants, affecting mucous membranes and deeper tissues through rapid absorption. Handling without gloves or eye protection opens the door to problems. No one wants a preventable accident, especially in a field where basic precautions usually keep people safe. My own routine—checking glove integrity, double-checking container seals—feels like common sense after years of practice, but it continues to pay off.
Environmental and Long-Term Concerns
Fluorinated chemicals stick around in the environment longer than most people expect. Their carbon-fluorine bonds resist breakdown by sunlight, water, and microbes. Substances in this family don’t just fade away; they can persist in water, soil, and living organisms. Areas near chemical plants sometimes turn up traces years after supposed cleanup. That kind of persistence deserves close scrutiny—not just for workers, but for the communities nearby and the wildlife sharing the local ecosystem.
The science is clear about the need for careful management, even when regulatory bodies haven’t drawn strict lines yet. We’ve all seen the headlines about “forever chemicals.” While 1,3-difluoroacetone hasn’t landed on everyone’s radar, its structure hints at similar resilience. As knowledge expands, so does responsibility. Protecting against accidental releases and investigating potential residues belongs at the top of the list, especially since long-term health effects often take years to reveal themselves.
Building a Safer Framework
Companies and researchers can do better by digging into real-world exposure scenarios. Inventories, spill drills, and proper disposal channels make up a sensible toolkit. Workers benefit from ongoing education that evolves with the science, something I try to reinforce anytime I mentor new lab staff. People outside the field also gain when experts share results of environmental testing and risk assessments. Candor, not secrecy, builds trust.
Some professionals push labs and manufacturers to swap out risky chemicals for safer alternatives whenever possible. Progress moves slowly, but the right incentives help tip the balance. Paying attention and redesigning old habits goes a lot farther than hoping a hazard never causes trouble. In the end, respect—honest, methodical, everyday respect—keeps people and nature safer around chemicals like 1,3-difluoroacetone.
Curiosity and Controversy Around 1,3-Difluoroacetone
Interest in 1,3-difluoroacetone often comes from labs working on niche chemical syntheses or research projects. Students and researchers come to me asking where to track it down, maybe because it’s not something you’ll see on the shelf of the corner hardware store. Tracking down this specialty chemical involves more than knowing catalog numbers or supplier names—it brings up real questions about access, safety, and responsibility. People want to know not just where to buy it, but why it matters who buys it and in what quantities.
The Sales Channels: More Gatekeepers Than Gateways
Finding 1,3-difluoroacetone doesn’t follow the pattern of getting, say, acetone or sulfuric acid. Most suppliers that stock it work with certified chemical distributors catering to researchers, universities, or industrial clients. I’ve seen purchases pass through established chemical supply houses, and the process usually kicks off with submitting a formal request, backed by documentation outlining the intended use and safety protocols. I’ve noticed these steps become stricter—especially for chemicals with potential regulatory concerns—since a lack of oversight can lead to trouble.
Online chemical suppliers sometimes list 1,3-difluoroacetone, but access usually stops at demanding credentials. Whether you’re trying to buy from Sigma-Aldrich, TCI, or similar players, they ask for institutional verification and proof that you run a real research or production lab. These measures spring up out of necessity, not corporate inertia. Legal guidelines get stricter every year, and suppliers stand to lose much more than a sale if chemicals fall into the wrong hands. From the customer side, the process ensures anyone handling these things knows what they’re doing.
Packaging Sizes—No Bulk Bargains Here
Unlike common solvents that arrive by the drum, 1,3-difluoroacetone gets packaged on a much smaller scale. My own lab orders typically range from 1 gram to 25 grams, rarely more than 100 grams. Manufacturers favor glass ampoules or smaller-sealed bottles, often triple-packed to prevent leaks or contamination during transit. Packaging reflects more than just volume; it’s about safeguarding shipping, lab handling, and end-user safety. I’ve never come across a reputable supplier selling this compound by the kilogram or liter, likely because demand remains low, and the chemical itself presents hazards that make bulk handling risky.
Anyone who wants to work with 1,3-difluoroacetone quickly realizes there’s a reason for these small containers. Smaller quantities minimize the fallout from spills or accidents. Suppliers, conscious of their own liabilities, avoid shipping anything that could escalate into a disaster if mishandled. Regulations—the kind you can’t ignore—sometimes enforce these packaging restrictions outright.
Why Access Remains Limited—and Why That Matters
For legitimate researchers, these restrictions may seem like a headache. I’ve spent hours filling out forms and verifying credentials just to acquire a few grams. But past incidents have taught the industry tough lessons. Careless distribution or poorly regulated sales let hazardous materials slip into the wrong hands. Nobody wants to turn on the news and see a familiar compound headline a safety incident. The buying process stretches out because it must. Supply companies, regulatory agencies, and researchers all share a piece of the responsibility to keep rare or hazardous chemicals out of trouble.
For those running into roadblocks, collaboration often works best. Labs can pool resources with university procurement offices or partner with approved institutional vendors. Pushing for clearer communication between researchers and suppliers helps speed approvals and ensures responsible use. Ultimately, the journey to secure 1,3-difluoroacetone spotlights real-world lessons: careful access and well-considered limits serve everyone in science, safety, and society.