Reflections on 1,4-Bis(Methoxymethyl)-Benzene: Science, Use, and Future
The Story Behind a Versatile Aromatic Compound
Chemistry often stirs curiosity because it gives us building blocks that shape hundreds of finished goods. 1,4-Bis(Methoxymethyl)-Benzene caught my attention early during my laboratory days for its curious blend of stability and flexibility. The journey for this compound, which chemists sometimes call p-xylylene dimethyl ether or 1,4-dimethoxymethylbenzene, isn’t one you find in history books, yet it deserves a place as it bridges classic benzene chemistry and the world of tailored specialty molecules. In earlier decades, scientists chasing better ways to functionalize aromatics looked for controlled methods. The methoxymethyl groups on this benzene ring bring both protective qualities and reactive opportunities—a rare combination. From my own experience, the careful crafting of such ethers really reflected the drive to push boundaries in organic transformations, even before they became common reagents in some advanced syntheses.
Understanding the Product Beneath the Lab Coats
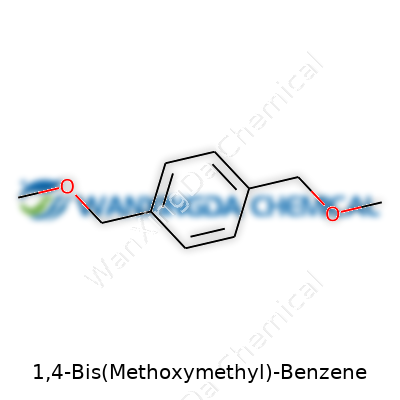
At its core, 1,4-Bis(Methoxymethyl)-Benzene rests on a benzene ring, carrying two methoxymethyl groups at the para positions. This structure gives it a mild, organic scent and a pale appearance, sometimes as a solid, other times as an oil depending on lab conditions. In handling the substance directly, you’d find it doesn't evaporate easily under standard conditions. What sticks out is its low solubility in water but much higher solubility in most organic solvents—useful in multi-step syntheses and often helping to keep things simple when separating products. Empirical formula C10H14O2, melting around 42-44°C, and boiling well past 250°C, tell you that it stands up to moderate heat without much fuss. Lab work hinted that this molecule behaves with enough resilience to survive in a flask with slightly rough handling, yet the oxygen-containing side groups make it ready to react if chemists push it in the right direction.
Technical Specs and Real-World Labeling Insights
Labels on scientific containers signal trust and accountability; over the years, I’ve learned to look beyond just the CAS number (which for this compound is 5529-99-3) and consider purity reporting, recommended storage, and precautionary notes. Reliable supplies often hit above 98% purity, and in research settings, this standard gives confidence for even sensitive downstream reactions. Quality assurance always asks more than just a chemical formula; one seeks batch analysis, residual solvent content, and handling recommendations. I remember working through outdated inventories and feeling nervous if purity or date tested were unclear. Modern best practice means storing away from strong oxidants, in amber bottles, away from direct sunlight, at room temperature as moisture and air lead to slow degradation.
The Science of Making and Transforming
Preparation methods earned attention not just for what they make, but for the cleverness chemists inject. In practice, 1,4-Bis(Methoxymethyl)-Benzene usually stems from a Friedel-Crafts alkylation or methylation route. Starting with p-xylene or its dibrominated sibling, synthetic chemists coax the needed methoxymethyl groups onto the ring, using formaldehyde derivatives with acid catalysts. It’s not a simple “mix and stir;” moderate heat, slow addition, and careful workup yield the cleanest product. Later, I saw this compound pressed into service as a protecting group source in multi-step total syntheses; it’s strong enough not to break during tough reactions, but susceptible enough to remove under milder acidic conditions. Researchers also lean on it as a precursor for aromatic polyethers, cross-linked polymers, or specialized ligands. I recall frustration when pushing too hard on reaction conditions, only to lose some of that precious ether to decomposition—a humbling reminder chemistry isn’t just theoretical, but always subject to the messy realities of the bench.
Naming and the Value of Clarity in Synonyms
The more chemistry classes I taught and research papers I read, the more I realized how synonyms help or hinder. For 1,4-Bis(Methoxymethyl)-Benzene, p-xylylene dimethyl ether flashes by in older European texts; dimethoxymethylbenzene pops up in certain patents. Such diversity in naming sometimes leads to order miscommunications. It’s vital, in my experience, that protocols standardize how they reference both the common and systematic names, making sure results cross borders and generation gaps without confusion. Chemical nomenclature doesn’t just serve pedantry—it works as a bridge across research teams who might otherwise double up effort or, worse, miss critical safety points.
Practical Safety Mindset and Handling Rules
The ordinary laboratory sometimes feels safer than it is; an aromatic ether like this seems harmless at first glance, but experience teaches respect. This molecule doesn’t come across as volatile poison, yet repeated exposure without gloves or proper ventilation can cause real irritation. Lab notes from past years recall headaches after hours in poorly ventilated rooms. Regulatory bodies rarely list this compound as highly flammable or explosive, but they don’t eliminate risk. Good practice guides recommend nitrile gloves, splash goggles, and using chemical hoods during synthesis or transfer—overkill only until that one moment something spills or splashes. Waste management needs the same routine care; collecting any spent or excess chemicals for incineration or specialized disposal, never the drain.
Where Ideas and Application Meet: Making a Difference in Research
1,4-Bis(Methoxymethyl)-Benzene finds its way into more than just obscure reaction schemes. Modern organic synthesis turns to protecting groups as both shield and key; this molecule fits both roles in synthesizing pharmaceuticals, dyes, and complex polymers. Some researchers build on its foundation to generate new materials, including extended aromatic frameworks or advanced polymers prized for thermal stability. The electron-donating nature of its side groups tweaks reactivity patterns, allowing creative approaches to functionalize benzene derivatives in ways simple methyl groups never could. I’ve seen colleagues in polymer research tweak backbone flexibility using this building block, opening up new materials for electronics or coatings.
In the Shadow of Discovery: Research, Risks, and Development
Even small molecules draw massive effort from researchers worldwide. Part of the appeal of 1,4-Bis(Methoxymethyl)-Benzene in labs is the balance of commercial availability and the simplicity of in-house preparation. Analytical chemists probe its behavior using NMR, IR, and MS, mapping every resonance and shift to learn more about its stability and breakdown paths. Newer work stretches into using this benzene as a launching pad for functionalized dendrimers, surface-linked catalysts, or as a core for host-guest chemistry in supramolecular assemblies. These discoveries hinge on real experience, sharing mistakes, reporting impurities, and significant collaboration.
Exploring Toxicity: Learning Before the Crisis
Toxicity remains elusive for specialty aromatics. Publicly available data lags behind what chemists wish they had. Small-scale reports hint at mild skin and eye irritation, and long exposure during manual handling leaves dry hairline cracks in the skin—personal reminders from seasons gone by. No one I know reported acute poisoning, but chronic effects remain essentially undocumented outside limited in-house industry studies. These gaps signal how much more work the research and regulatory community could devote to gathering data, not just trusting that a “lack of evidence of harm” means complete safety. Knowledge from analogous compounds supports cautious optimism for low environmental persistence, yet that doesn’t excuse slack management of wastes or disregard for chronic exposure limits in workspaces.
The Road Ahead: Prospects and Unfinished Questions
Research constantly seeks tools that combine reliability and adaptability; 1,4-Bis(Methoxymethyl)-Benzene brings both. Its future in chemical manufacturing depends on downstream demand for specialty aromatics capable of protecting, activating, or bridging two diverse functional realms. Technological progress around green chemistry spurs creative ways to prepare such molecules with smaller waste streams and milder reagents—goals I’ve seen move from ambitious to necessary in only a decade. Industry and academia both weigh environmental impacts now as fiercely as reactivity or yield. In coming years, more data from high-throughput screening, predictive toxicology, and pilot-scale applications will decide if the potential for safer, more selective benzene derivatives really changes the way we build pharmaceuticals, advanced materials, and industrial chemicals. Every day in the lab, facing real mess and incremental success, reminds me the story of a simple aromatic ether never finishes—it adapts as science and society need it.
Getting Down to the Details
Let’s clear the fog around 1,4-Bis(Methoxymethyl)-Benzene. Picture a benzene ring first—it’s a simple, flat hexagon made up of six carbon atoms with alternating double bonds. To that familiar structure, take slots 1 and 4 across from each other on the ring and attach a methoxymethyl group to each one. In plain terms, a methoxymethyl group is a –CH2OCH3 chain. The full structure looks like a symmetric benzene, dressing up with these two extended arms on opposite sides.
I’ve always found symmetry in chemical structures satisfying—something about knowing exactly where those groups sit helps bring order to the lab bench. That’s what you get here. Chemists often refer to this pattern as “para-” substitution, because the two groups face off at the furthest points from each other on the ring. It’s more than an academic detail; it shapes how the molecule behaves, reacts, and fits into the world of materials and manufacturing.
Why This Structure Matters Beyond the Lab
Understanding chemical structures isn’t just for textbook fun. Take my time working with aromatic compounds in the lab—one thing I learned quickly was how a tiny change in structure could flip a process on its head. With 1,4-Bis(Methoxymethyl)-Benzene, the methoxymethyl arms change the way this compound interacts. Those extensions shield the benzene core and tweak its solubility, making it more willing to mix with organic solvents. That opens up possibilities in making specialty polymers, coatings, or even as a building block for more intricate molecules.
The practical angle kicks in when research groups use this backbone to craft dendrimers or custom resins. Each methoxymethyl group isn’t just decoration—the way it hands off electrons or interacts with other reagents shifts the outcome of a whole reaction. That means this compound could help chemists dial up or down certain properties for advanced materials, something industries keep searching for.
Roots in Everyday Chemistry
As I see it, 1,4-Bis(Methoxymethyl)-Benzene is a small cog in the bigger machinery of synthesis. A painter doesn’t need to know how paint was made, but for anyone cooking up the next adhesive, coating, or even pharma intermediate, knowing the structure at this level matters. The para-positioning often brings stability and reactivity to the table, letting chemists predict how a reaction will unfold. It isn’t just luck—folks spent decades mapping out how every flavor of substitution changes the game. This groundwork makes modern chemistry faster, safer, and less wasteful.
What Could Make a Difference Going Forward
Looking ahead, the lesson for chemists and product developers: dig deeper into structures like this one. A textbook image only tells half the story. By experimenting with these substitution patterns and arm lengths, new properties can be unlocked—cleaner reactions, greener synthesis methods, or just better performing end products. I’d push for more shared databases and open collaboration across borders, so discoveries around molecules like 1,4-Bis(Methoxymethyl)-Benzene don’t just sit on lab shelves but help spark the next breakthrough, wherever it’s needed.
Looking Beyond the Lab Bench
Chemicals like 1,4-Bis(Methoxymethyl)-Benzene rarely draw the spotlight, yet these small players drive some of the most important progress in science and manufacturing. Known among chemists for its stability and unique structure, this compound brings its own set of skills to several key fields.
Synthesizing New Possibilities
Most people won't ever see a bottle labeled with this compound on a hardware store shelf. Yet, its true value appears in research labs, where it helps shape the frontiers of organic synthesis. From my own time working with complex aromatic compounds, it’s clear that few molecules play such a reliable supporting role as this one. It often serves as a building block—imagine Lego bricks, but on a molecular scale. Its symmetrical benzene core and methoxymethyl arms make it flexible for scientists trying to add or adjust different functional groups in a new molecule.
Patents and academic journals document its frequent starring role in pharmaceutical research, too. Drug developers sometimes choose 1,4-Bis(Methoxymethyl)-Benzene to create safer reaction paths and cleaner products. This cuts waste and cost, which matters a lot once a drug scales up for hospitals and clinics.
A Link in Advanced Material Chains
Creating stronger, lighter, or smarter plastics and resins often begins with small molecules that bring unique features to the mix. The structure of 1,4-Bis(Methoxymethyl)-Benzene lets chemists fine-tune crosslinking in specialty polymers. In my experience, tweaking the backbone of a polymer with such compounds directly impacts the final material’s durability and resistance to heat or stress. Electronics and automotive parts owe some of their reliability to careful chemistry like this far upstream in the process.
Its use also stretches into coatings and specialty adhesives. These coatings must stand up to harsh industrial environments, so engineers rely on compounds that handle both strong acids and temperature swings. Here, the stability in its structure keeps things in balance, especially in high-stress conditions where weaker molecules might degrade.
A Quiet Ally for Clean Science
Green chemistry isn’t just a trend; it’s a must. Safer processes and better yields cut both pollution and production costs. Having worked with alternative solvents and safer reagents, I know the value of a dependable compound that doesn’t produce a pile of nasty byproducts. 1,4-Bis(Methoxymethyl)-Benzene often fits with these goals, giving chemists an option with less risk in both handling and disposal.
Room for Innovation
Looking forward, the search for new battery materials, recyclable plastics, and efficient solar cells puts pressure on chemists to find or invent better ingredients. By providing a stable, adaptable base, this compound gives researchers a leg up as they design tomorrow’s materials. Its continued use signals more potential breakthroughs, as chemists build on what we already know.
Facing Real Issues and Solutions
No chemical comes without its challenges. Handling, cost, scaling, and sourcing all present hurdles as demand changes. Solutions often spring from investment in safer manufacturing and greener sourcing, while open collaboration between academic and industrial labs helps spread new ideas about its uses and impacts. Regulatory oversight based on rigorous evidence builds trust and encourages best practices, making sure advancements don’t trade safety for speed.
Experience and Lessons from the Chemical Bench
Over the years, few things have taught me more respect than handling aromatic ethers in the lab. Tidy benchwork and careful storage habits can save a lot of headaches, both for the user and for others down the line. Among the compounds that cross lab benches, 1,4-Bis(Methoxymethyl)-Benzene stands out, not because it's common in everyone's garage, but because it needs just the right dose of respect in daily use.
Why Conditions Matter for Stability
If you’ve worked with solid organics before, you know not everything likes to play nice in the open. Exposure to moisture or light can turn even the most stable-looking powder into a sticky or degraded mess. For 1,4-Bis(Methoxymethyl)-Benzene, the story goes much the same. Moist conditions trigger slow hydrolysis, which damages both the purity and the usefulness of the material. Direct sunlight can bring unwanted chemical changes too, leading sometimes to yellowing or breakdown products that might surprise a user months later.
The best practice I’ve seen in labs: a tightly sealed glass container, stored away from sunlight, at a spot where temperature swings are rare. Most modern storage rooms rely on cool, dry shelving, often with desiccators around if the season runs humid. Cold storage isn’t usually essential for this compound, but room temperature—away from any sources of humidity—minimizes risk. In my experience, skipping these steps tends to show up as batch-to-batch inconsistency, something no one enjoys troubleshooting.
Practical Safety: Avoiding Fumes and Skin Contact
Chemical industry guidelines and safety data sheets agree on the basics: gloves, goggles, and a good habit of not sniffing directly above your sample. This compound doesn’t always have a strong odor, but skin exposure can cause irritation. I have seen new lab workers skip gloves—once. One case of mild redness or itch, and they never made that mistake again.
Proper ventilation can’t be ignored, either. Even solids can release vapors over time, especially if ambient temperatures rise or the seal on a container fails. Local exhaust fans and well-maintained hoods do more than keep smells at bay—they help prevent chronic exposure. There’s enough research connecting aromatic compound fumes to long-term health drawbacks that no one should cut corners.
Sensible Planning: Labeling and Logistics
Clear labeling often gets overlooked, but I remember cleaning out a store cupboard where similar-looking jars had lost labels and picked up a sticky residue. It took half a morning to sort out what was what. Using sturdy labels that won’t peel or fade lets everyone know exactly what’s in front of them, and how to deal with it.
Organizing chemical storage isn’t just about tidiness. Segregating compounds by compatibility means accidental mix-ups or reactions almost never happen. Simple logic: don’t store reactive chemicals near acids or oxidizers. If disaster strikes and something spills or breaks, the risk of a dangerous chain reaction drops way down.
Waste and Spill Handling: A Slice of Reality
There’s a strong temptation to shrug off a small spill, especially late in the day. Don’t give in. Even trace amounts of 1,4-Bis(Methoxymethyl)-Benzene can make a surface slippery or leave behind irritating residues. Absorbent pads and simple soap-and-water cleaning do the trick for most work surfaces. For larger spills, chemical waste containers—marked and handled with respect—avoid cross-contamination and trouble with disposal authorities.
Experience shows a good system beats luck every time. Good handling and storage aren’t just for peace of mind or ticking regulatory boxes. They mean cleaner results and safer work, day after day.
What This Chemical Is All About
1,4-Bis(Methoxymethyl)-Benzene doesn’t exactly roll off the tongue. In a typical chemistry lab, you’ll find it quietly tucked away—one of many organic compounds with a niche use. Most folks outside pharmaceuticals, plastics, or materials science won’t bump into it. Still, for those working around the stuff, questions about its safety naturally come up.
Is Danger Lurking Beneath the Surface?
Chemical names often spark concern. There’s real reason for caution, as stories about mishaps in labs or factories remind us. Curiosity about toxicity isn’t paranoia; it’s a necessity, especially when handling anything with a scientific name as long as your arm.
Digging through trusted sources—peer-reviewed journals, chemical safety databases, and official government repositories—hard toxicology data on 1,4-Bis(Methoxymethyl)-Benzene appears scarce. That’s not a green light for recklessness. The lack of data doesn’t mean it’s harmless, only that it sits below the regulatory radar. A lot of specialty chemicals end up this way, especially those used on a small scale or in controlled academic settings.
The chemical structure hints at its nature. Aromatic rings with side chains containing methoxymethyl groups suggest that it might act as an irritant. Similar compounds sometimes cause skin, eye, or respiratory irritation if handled carelessly. I’ve watched seasoned lab veterans treat even “safe” organic compounds with gloves and goggles, because risk can hide where you least expect it.
Hazard Awareness in the Real World
On the job, nobody wants preventable accidents. I’ve handled a parade of suspicious chemicals over the years and can say that hazard comes in many forms. Sometimes the danger lies in acute toxicity—exposure that brings on sudden symptoms like headaches or nausea. Other times, it lurks in the long game, building up in the body, or latching on to DNA in ways that take decades to show up in statistics.
Without concrete studies proving long-term harm or safety, the default setting should lean toward care. Even for chemicals without a stack of health warnings, smart practice means keeping ventilation humming, wearing gloves, and working behind a shield if there’s splashing or vapor.
Why Broader Attention Matters
Environmental persistence weighs on my mind, too. Aromatic rings can stick around long after they leave the lab bench. Disposal protocols matter. Pouring leftovers down the drain might feel routine, but you could send that chemical on a journey through the water system, where its effects remain unknown. We’ve watched enough unregulated compounds turn up in odd places to know this deserves respect.
Regulators simply can’t review every substance landing in our lives. There’s always a lag between introduction and careful scrutiny. Grassroots safety fills in the gaps. If a compound lacks a clear safety profile, it makes sense to treat it much like its better-known cousins—don’t inhale, don’t touch, don’t release it carelessly.
What Steps Can Make the Biggest Difference?
Every industry and lab can take simple actions. Set up clear labeling and storage. Share hazard updates as science catches up. Build disposal routines that keep new pollutants out of drains and air. Personal experience says that a strong culture of respect for chemicals, no matter how obscure, keeps everyone out of the danger zone.
In the face of uncertainty about substances like 1,4-Bis(Methoxymethyl)-Benzene, practical caution shapes real safety. Asking questions, demanding transparency, and relying on common sense and good habits—this is how workers, researchers, and the wider public can keep risk contained and avoid the lessons learned the hard way.
Purity: More Than Numbers on a Page
In any lab, bottling up organic intermediates like 1,4-Bis(Methoxymethyl)-Benzene means chasing after a level of purity that balances workload, cost, and safety. From handling the white powder form at my own bench to prepping for a chromatographic run, a single point crops up over and over—what’s actually in the bottle? Purity specs seem boring at first, but lives and millions in project spending quietly ride on this one fine detail.
Major suppliers lean on purity values over 98%. Some squeeze standards tighter, hitting 99% or better depending on intended use. It’s not about splitting hairs. A synthetic sequence can collapse if a trace contaminant trips up a catalyst. Process-scale hiccups can flood a batch with by-products, leaving more waste and spiraling costs. Even if you’re just in research, leftover solvents or unreacted intermediates from an impure starting material can muddy up NMR readings, mess with yields, and trigger days lost on finding what went wrong.
Facts Behind the Bench: What’s Actually at Stake?
Nobody likes to talk about costs until the project goes over budget, but let’s keep it real: higher purity drives up price, since it pulls extra steps out of synthesis or purification. I remember hunting through suppliers, hoping for a reliable COA showing HPLC, NMR, maybe melting point—anything to pin down what’s in the vial. At the higher end, you’ll see products clearing 99% purity as verified by several analytical techniques. This extra certainty matters most for pharmaceutical and electronics work, where a single unknown can send an FDA audit off the rails, or short a chip in a prototype.
That said, not every project needs the highest number. Sometimes a quick in-house crystallization or distillation puts the material where you want it. Who hasn’t tried to stretch a tight budget by buying the technical-grade stuff and doing a cleanup? But, let’s not kid ourselves—certain impurities don’t come out so easily, especially aromatic by-products or heavy-metal residues.
The Impact on Safety and Sustainability
Many overlook how purity links to safety. Unexpected impurities mean side reactions, sometimes exotherms, which can generate hazardous conditions during scale-up. For labs juggling greener chemistry or minimizing hazardous waste, every impurity is a potential source of downstream contamination or disposal issues. Regulatory agencies also have their eyes on these problems, and so do insurers. The more trustworthy the source and documentation, the less likely the experiment goes sideways or that you’ll pay for clean-up later.
Solving for Purity in a Real-World Lab
Nobody loves extra steps, but sometimes, in-house testing is the best move. Running your own NMR, MS, or checking for water by Karl Fischer gives some peace of mind, especially on sensitive or scaled-up syntheses. Vetting suppliers for lot-to-lot consistency stops headaches before they start. Over time, building relationships with suppliers who take analysis seriously—sharing spectra, not just numbers—beats chasing refunds after a failed batch.
Labs and companies can also push the conversation beyond just “what percent purity?” by asking about residual solvents, trace metals, and specific side-products. Don’t just assume 98% means “good enough” for every project. Understanding what’s in that other two percent can save a week of troubleshooting, or worse, a regulatory disaster.
As the market keeps tightening standards, and end-users face new regulatory and technical hurdles, purity won’t just be about numbers. It’ll be about trust, about documented evidence, and about the real, practical cost of that peace of mind.