Reflections on 1-Bromo-1-Chloro-2,2,2-Trifluoroethane: Legacy, Chemistry, and Tomorrow’s Questions
Historical Development: Tracing the Path of Halogenated Ethers
The journey of 1-Bromo-1-Chloro-2,2,2-Trifluoroethane traces back over half a century, fitting squarely in the timeline of global discovery surrounding organohalide chemistry. In the twentieth century, scientists were chasing molecular features that could help solve problems in refrigeration and chemical synthesis. This type of halocarbon emerged from research schools determined to fine-tune vapor pressures, chemical stability, and low toxicity for industrial applications. Over the decades, the market for fluorinated and halogenated ethanes waxed and waned. Regulatory pressures on ozone-depleting substances and hazardous pollutants shaped its journey, reminding every chemist how discoveries live in the crossfire of innovation and regulation. Having worked beside engineers who remember the heyday of CFC and HCFC excitement, I see historical lessons in every regulatory update and renewed call for safer, more targeted molecules.
Product Overview: More Than Just a Name
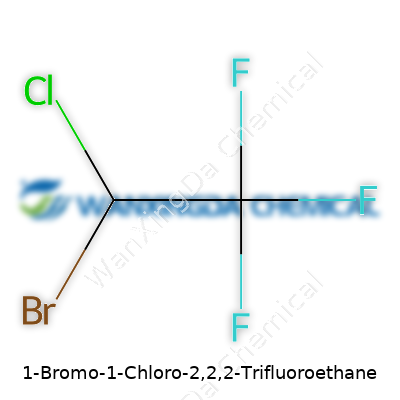
1-Bromo-1-Chloro-2,2,2-Trifluoroethane earns its technical reputation not just from its hefty name or CAS number, but from its unique blend of atom types that unlock industrial value. It’s more than a footnote in the catalog of halocarbons; it brings potent reactivity (or desired inertness) to fine chemical synthesis. Laboratories test its abilities as an intermediate because the three fluorines skew the electronic landscape, giving it stability where others might decompose. People in the field—whether organic researchers or product developers—quickly learn that a molecule’s quirks open doors for new reactions and, sometimes, spark fresh curiosity about safer, smarter replacements.
Physical & Chemical Properties: Fluorine’s Heavy Hand
Every time I’ve held a vial of a heavily halogenated compound, the weight and density surprise me. 1-Bromo-1-Chloro-2,2,2-Trifluoroethane feels substantial—halogen atoms bring bulk and a certain stubbornness against reactions. Chemically speaking, this kind of structure serves up low flammability, high volatility, but also invites nuanced safety risks. The trifluoro configuration drives up electronegativity, pulling electron clouds and producing a molecule that resists hydrolysis, oxidation, and casual breakdown. Such resistance, once prized for ticketing new refrigerants and solvents, now draws scrutiny because persistent molecules tend to linger in both the environment and the news headlines.
Technical Specifications & Labeling: What the Lab Demands
Nobody likes a surprise in the chemical storeroom or production line. That’s why people demand clear documentation—boiling points, reactivity notes, and purity standards. Labels list hazard codes and storage tips because safety depends on familiarity with the invisible dangers at play. Many a chemist has learned to read between the lines: warnings about halogen exchange, the taste of volatility on the air, and the hint of reactivity curled up in a simple structural diagram. This hands-on knowledge is not academic trivia; it serves as the backbone of responsible lab culture.
Preparation Method: Under the Hood With Synthesis
Building 1-Bromo-1-Chloro-2,2,2-Trifluoroethane demands careful orchestration. The key involves selecting the right starting materials—often fluoroethanes—and introducing bromine and chlorine with precision. The synthesis usually leans on halogen exchange reactions, where strict controls keep byproducts in check. Industrial-scale runs demand patience: each batch might require exact temperature ranges, pressure controls, and purification steps. In talking with process chemists, the refrain is the same—the trick lies less in textbook formulas and more in controlling the real-world quirks of exothermic reactions, trace impurities, and the constant battle against unwanted side products.
Chemical Reactions & Modifications: From Source to Suitability
Reactivity discussions about 1-Bromo-1-Chloro-2,2,2-Trifluoroethane fill up pages of organic synthesis journals. The molecule’s mix of halogens makes it a flexible building block—excellent for introducing bromine or chlorine into aromatic or aliphatic targets. Its trifluoro group offers up electron-withdrawing punch, often shifting selectivity and yield in substitution or elimination reactions. I’ve watched researchers tune reaction conditions, discovering that temperature, solvent, and catalyst selection all determine whether the product is a success story—or just another failed experiment. Efforts to tweak it for specialty compounds fuel a steady stream of patents and research, each chasing that elusive blend of performance, cost, and safety.
Synonyms & Product Names: Navigating an Alphabet Soup
A walk through chemical supply catalogs quickly teaches that 1-Bromo-1-Chloro-2,2,2-Trifluoroethane wears many hats. Whether someone calls it Halothane BCF3, or tags it by its IUPAC code, the underlying molecule brings clarity to those who follow the threads of global commerce and regulation. Chemists know that translation between synonym lists isn’t just academic—mixing up a halocarbon can shut down a lab or mire a project in paperwork. Those who spend years in the field develop sharp instincts for catching label mix-ups and cross-referencing numbers, not just names.
Safety & Operational Standards: Defending People and Planet
Hazardous materials demand discipline, not just at the bench but across the company. 1-Bromo-1-Chloro-2,2,2-Trifluoroethane brings the full suite of risks associated with heavy halogenation. Exposure limits, ventilation standards, and handling protocols fill thick safety binders and trusted online resources. The stories that stick in memory are always personal—a missed glove, a forgotten vent hood, a colleague’s cautionary tale. Forgetting safety is not an option; standards work best when they become habits, and the best labs make these routines second nature from the newest intern to the seasoned group leader.
Application Area: Not Every Molecule Joins the Mainstream
The value of 1-Bromo-1-Chloro-2,2,2-Trifluoroethane stretches across different industrial corners. Early on, researchers looked to it for refrigerant blends, custom solvents, and chemical intermediates that could handle tough reaction conditions. Its structure made it both an asset and a target as industries sought lower environmental burdens and higher performance. Academic labs and niche manufacturers sometimes use it in small batches—often as a specialty reagent, never on the scale of more famous halocarbons. In commercial settings, evolving regulations squeezed out some use cases, yet a select group of applications remain, tied to its unique blend of stability and reactivity.
Research & Development: The Relentless Search for Better Chemistry
R&D in the chemical world often means chasing the next big thing just as regulatory headwinds close the window on today’s solution. Research on 1-Bromo-1-Chloro-2,2,2-Trifluoroethane regularly appears at the edge of innovation and accountability. Analytical teams hunt for ways to break down persistent molecules, studying advanced oxidation or catalytic reduction that could render residues safer. Synthetic chemists look for derivatives or next-generation molecules that keep the benefits but leave behind the baggage. In my experience, the most exciting work rarely happens in isolation—it’s always a cycle of discoveries, setbacks, and partnerships between academic labs, regulators, and commercial teams.
Toxicity Research: Chasing Answers, Shifting Questions
With halogenated ethanes, the toxicity conversation never falls silent. Scientists call for robust data: animal studies, cellular assays, environmental fate models. Many of these compounds hang around in soils and air, sparking questions about bioaccumulation, long-term exposure, and unintentional outcomes. Consumer safety agencies and regulatory bodies keep standards moving as new findings surface—what was considered safe yesterday may face new restrictions tomorrow. I’ve seen firsthand how questions about one halocarbon ripple across the industry. Each toxicological study and regulatory update forces teams to rethink assumptions, balance risk, and weigh the consequences of using or retiring a longstanding workhorse molecule.
Future Prospects: Learning From the Past, Looking Forward
Every time a new environmental policy takes effect or a fresh scientific study lands, prospects for 1-Bromo-1-Chloro-2,2,2-Trifluoroethane get reevaluated. Molecules like this one face shrinking windows in conventional industries; the world wants alternatives that cut down on pollution, toxicity, and lifecycle impacts. Yet history shows that old chemistry sometimes sparks new ideas—breakthroughs can reinvent forgotten compounds as part of future green syntheses or closed-loop recycling technology. The most promising path forward demands honesty about limits, creative collaboration, and a steady hand for safety. It’s not easy, but the future of chemistry keeps pushing for molecules that outperform the past—without repeating its mistakes.
Understanding Its Place in Industry
Working in chemical research labs and talking with folks in refrigeration, the name 1-Bromo-1-Chloro-2,2,2-Trifluoroethane can make eyes glaze over. Most people outside the field wouldn’t recognize the formula, let alone feel comfortable working with it. But this specialty halogenated compound has a story tied closely to big changes in technology and environmental policy.
From Extinguishing Flames to the Next Big Thing
Decades ago, fire suppression demanded chemicals that could knock down flames fast, especially in places where water could cause more harm—think computer rooms, archival storage, or airplane hangars. Halons used to set the standard. Then science caught up with the environmental impact. CFCs (chlorofluorocarbons) and brominated compounds like Halon-1211, which shares similarities with our subject, ended up on the chopping block after research nailed down how they tear holes in the ozone layer. The Montreal Protocol forced entire industries to rethink flammability management.
1-Bromo-1-Chloro-2,2,2-Trifluoroethane (known by some as Halon 1211) carved out a niche because it packs a punch—knocking back flames quickly, interrupting the chain reactions that keep fires going. Firefighters in the aviation sector saw it as a go-to extinguishing agent. Its low electrical conductivity meant you could spray it on live electrical gear in a pinch.
The Push for Alternatives
The problem: ozone destruction. One kilogram of this halon can do way more ozone damage than the average car emits in a year. Once policymakers woke up to these risks, regulatory bans pushed much of the market toward alternatives. Reclaimed supplies and specialized exemptions kept it hanging around for some military, aviation, and certain critical industrial uses. Old fire suppression systems that can’t be easily swapped get grandfathered in and folks managing large buildings or airliners hope old stocks hold out.
Beyond fire protection, some chemists experimented with compounds like this one for limited roles in specialty synthesis or as solvents. But tighter controls and environmental reporting discouraged anyone from expanding those uses.
The Dilemma and a Path Forward
I’ve seen retired fire marshals wrestle with trade-offs: Use a less-effective, more equipment-damaging agent and risk slower response, or stick with a known chemical that carries severe environmental baggage. For people who handle legacy infrastructure or oversee critical communications rooms, the reality is more nuanced than simply switching a product. Swapping systems often means millions in upgrades or the logistical headache of finding trained service providers.
Research into “clean agent” systems gained speed, giving rise to alternatives like FM-200 or Novec 1230. These substitutes protect assets and lives without the same atmospheric fallout. Still, those working in older planes or with historic archives sometimes don’t have a simple plug-and-play replacement. The chemical’s history signals a broader debate: speed and convenience versus environmental stewardship.
Real progress depends on funding transitions, supporting alternative research, and making policy decisions that recognize practical hurdles for everyday users. It’s easy to point fingers at old technology, but without bridging the cost and training gap, calls for change won’t mean much to people left managing relics of a different era.
Why we should care about the stuff in our air
Chemicals like 1-Bromo-1-Chloro-2,2,2-Trifluoroethane do not roll off the tongue, but when these types of compounds show up in workplaces, warehouses, or even products at home, the risks deserve some attention. We sometimes trust that things on the market pose no immediate danger, but that trust rests on careful research and real-world vigilance. If you've heard of this chemical, you probably know it tends to pop up as a refrigerant or in specialty cleaning processes. People using it, or just working nearby, should ask tough questions about its safety.
What science tells us about toxicity
My years working around industrial sites taught me to watch out for anything unfamiliar in a drum. For 1-Bromo-1-Chloro-2,2,2-Trifluoroethane, animal studies point to dangers after exposure, especially if ventilation falls short or someone works without proper safety gear. Researchers and regulatory agencies classify this substance as hazardous, mostly thanks to its effects on the lungs and nervous system. When breathed in high concentrations, it can knock a person off their feet—drowsiness, confusion, even unconsciousness. Handling concentrated amounts indoors puts workers at real risk.
Skin contact does not get a free pass either. Irritation comes quickly if it lands on bare skin, and you do not want this liquid near your eyes. Like many fluorinated and halogenated compounds, break it down with enough heat and you get even nastier byproducts—think toxic, corrosive fumes.
Long-term exposure and environmental concerns
Short-term problems draw fire, but a bigger concern circles around what happens with chronic exposure. The lungs can bounce back after one bad day, but breathing low levels over months can chip away at health. Some sources flag worries about liver and kidney effects, but the science feels murky there, at least at ordinary workplace levels. No matter, it’s hard to shake the fact that repeated contact with chemicals like this cannot be healthy over the long haul.
People should also think past the workplace. Once this chemical escapes into the environment, it sticks around. Persistence means it does not break down easily in the air; it climbs into the upper atmosphere, where it may take part in ozone depletion. Decades of learning about refrigerants have taught me to look past the label and ask what unintended damage might turn up, years down the line.
How to reduce risk
From a practical perspective, the path forward starts with good ventilation and personal protective equipment—gloves, goggles, and reliable respirators. That sounds simple but I’ve lost track of how many times I’ve seen shortcuts taken. Training makes a difference; people do not always know what they’re handling. Substituting less hazardous alternatives should rank high if the job allows for it. Industry moves fastest toward safety when workers speak out about corners being cut.
Regulatory agencies keep tightening rules on substances like 1-Bromo-1-Chloro-2,2,2-Trifluoroethane, mostly because workers and advocacy groups keep raising the flag. Regular air monitoring, good storage practices, and common sense remain the best shield. This chemical shows that what we do not see can do real harm—paying attention is step one.
Paying Attention Before Accidents Happen
1-Bromo-1-Chloro-2,2,2-Trifluoroethane sounds like something only chemists worry about, but stories behind chemicals like this shape the real world far outside the lab. If you walk into any facility working with specialty refrigerants or cleaning agents, keeping the right conditions for chemicals matters every bit as much as the fancy equipment in the room. Put the wrong chemical in the wrong spot, and suddenly you've got fumes, ruined materials, or worse, someone hurt.
Real Risks, Real Responsibility
Anyone dealing with halogenated hydrocarbons—like 1-Bromo-1-Chloro-2,2,2-Trifluoroethane—faces a mix of hazard and responsibility. Leaks from poorly sealed bottles can lead to headaches at best and life-changing injuries at worst. Many years ago, I watched seasoned warehouse workers dismiss warnings about chemical vapors, only to evacuate their building later that year after a container failed. Preparation and respect for the material keep these stories from repeating.
The Details That Keep People Safe
So, what actually makes for safe storage? Start with a cool, dry, well-ventilated spot, with temperatures preferably below ordinary room temperature. Heat nudges chemicals to act up—vapors get released, pressure builds, and that turns sealed containers into potential hazards. Dry spaces matter just as much, since moisture can react with certain halogenated agents, ruining both the chemical and the container.
Storage away from sunlight and heat sources cuts the risk of pressure buildup and chemical change. I remember a facility where workers stacked refrigerant cylinders too close to heat lamps, thinking it wouldn’t make a difference. They got lucky—a leak set off alarms before things went further. That incident left everyone with a simple lesson: keep chemicals away from sources of heat and light, no matter how tough the container looks.
Containers: Not All Are Equal
Original containers do more than look tidy on the shelf; they provide a secure seal, chemical compatibility, and important information right on the label. Transfer to inappropriate bottles brings new danger. Materials like certain plastics or metals might react, corrode, or leak. Once a label is lost or switched, problems multiply—no one wants to play guessing games with hazardous substances.
Preventing Mix-Ups and Cross-Contamination
Mistakes rarely look dramatic at first. A careless label swap or keeping different chemicals crowded on the same shelf might not start a fire, but the unseen risks grow daily. Chemicals have personalities, and mixing them—even unintentionally—could spark reactions. Purpose-built, clearly marked cabinets help reduce the chance of grabbing the wrong container in a rush. Chemical storage cabinets with spill containment bases add another layer of safety for leaks or breakage.
Why Training and Respect Matter
Training looks boring until someone’s nose or lungs pay the price for forgetting it. Basic instructions about caps, seals, and ventilation keep the workspace safe for everyone—not just the chemists. From my experience in maintenance workshops, regular reminders and real-world stories make the risks feel close to home, leading to fewer shortcuts and mistakes.
Solutions That Actually Stick
Strong policies and clear routines make all the difference. Audits and reminders force everyone to look twice at old containers or sketchy storage spots. Investing in good storage cabinets and ventilation pushes the odds in your favor. Safe storage is no accident; it’s a habit built daily, long before anything goes wrong.
Pulling Back the Curtain on Specialty Chemicals
Most people walk right past the name 1-Bromo-1-Chloro-2,2,2-Trifluoroethane, but for those who keep tabs on specialty industrial chemicals, this stuff sticks out. Known to many as Halon 1211, it’s used mainly as a fire extinguishing agent—especially in spaces like aircraft, computer rooms, and museums where sensitive equipment or irreplaceable collections make traditional fire-fighting methods risky. That alone tells the story of why anyone started using this mixture in the first place: the balance between safety and risk.
Environmental Risks Under the Microscope
Cracking open the environmental track record of Halon 1211 doesn’t paint a rosy picture. The backbone of this chemical belongs to the halogenated hydrocarbons family. These compounds, which include old-school refrigerants and fire suppressants, have become infamous for their part in ozone depletion. There’s a very clear link between the release of halons and the thinning of the ozone layer. The Montreal Protocol wasn’t drafted without cause—it came about because scientists realized these substances do a number on the upper atmosphere, weakening our planet’s shield against ultraviolet radiation. Halon 1211 landed squarely on the list of chemicals to phase out because of this risk.
What surprises most is just how persistent these molecules can be. Once Halon 1211 escapes from a pressurized can, it doesn’t break down fast. The chemical floats up and, over several years, reaches the stratosphere. There, sunlight tears it apart. The resulting mess releases bromine and chlorine atoms, both of which actively break down ozone. Even in 2024, halons used decades ago are still doing damage.
Human Choices in a Balancing Act
Industry professionals—myself included—know there’s never an easy fix when protecting lives and heritage. Alternatives to Halon 1211 emerged over the years. The conversation among engineers and regulators shifted. Rather than sticking to the status quo, fire safety experts worked on swapping out halons where possible. Some switched to inert gas systems or new blends with less environmental baggage. These moves took guidance from hard evidence, not just wishful thinking. Living through these transitions taught me that adaptation is always possible if enough minds come together.
Ozone recovery isn’t instant. Even meticulous regulation and replacement schedules won’t erase years of emissions. What’s left for current and future users is a set of choices that mix practicality with responsibility. Organizations can start by stopping new releases, finding programs to capture and recycle the remaining stock, and only deploying halons under strict regulation where no immediate alternative measures up. International collaboration works best; the Montreal Protocol stands as proof.
Seeing the Forest, Not Just the Trees
Regulations can spell out the “what,” but the “why” lives in everyday decisions—what ends up in the air, not just what’s printed on a form. Learning from Halon 1211’s history reminds people that “effective” sometimes carries unspoken costs. I’ve met technicians who take extra care not to discharge fire systems during maintenance, knowing just how much it matters. These actions taken by individuals, not just policies, give hope that stewardship over the environment is alive.
Why Every Detail Matters with This Chemical
Spills of chemicals like 1-Bromo-1-Chloro-2,2,2-Trifluoroethane introduce real-world risks to those who spend time in labs, warehouses, or facilities that rely on these gases. I once spent a summer working maintenance around industrial refrigerants, and it taught me something quickly: the little details make a huge difference, for health and for the job.
Serious Health and Environmental Concerns
Most people won’t recognize this chemical by name, but it’s a popular refrigerant and cleaning solvent. It isn’t just unpleasant to inhale; science links it to headaches, dizziness, and possible heart trouble if exposure stretches out. These effects track with official agency warnings. Years ago, a coworker ignored the odd sweet smell after a hose coughed out a leak in a storage room. He walked it off at first but ended up needing fresh air and medical checks. That incident hammered home the lesson: chemical safety demands vigilance.
Workplace Habits That Make All the Difference
People who spend much time handling volatile chemicals pick up a few practical habits. Ventilation doesn’t get enough respect, yet it’s the first line of defense; even minor leaks call for clearing the space and getting air flowing. My experienced supervisors drilled a rule into me: use the right kind of mask, gloves, and goggles every time. No shortcuts.
A broom won’t cut it here. 1-Bromo-1-Chloro-2,2,2-Trifluoroethane spills often involve both liquid and vapor. Teams well-trained in hazards often keep absorbent pads close. Industrial absorbents do the heavy lifting, soaking up liquids without spreading residue or releasing extra vapors. After containment, material should go in sealed, marked drums for hazardous waste disposal.
Turning Spills Into Teachable Moments
These spills don’t just call for cleaning. They point out lapses — maybe someone skipped regular equipment checks or ignored cracks in hoses or storage valves. Every incident warrants a check-in. Who checked storage this month? Did we upgrade our leak detectors, or skip because budgets felt tight?
After an event, supervisors should walk everyone through what happened. I’ve been through post-incident briefings. The honest ones help keep everyone sharp and cut down on repeat mistakes.
Simple Solutions Stand the Test
Basic fixes go far: strong ventilation, reliable protective gear, and firm procedures. Facilities that stand out usually go the extra distance with training. Drills help shorten response time, and good signage lays out what people need to do if a spill hits. Local emergency numbers posted clearly have saved lives.
Choosing safer alternatives matters too. Newer refrigerants with lower toxicity now exist. Older, riskier chemicals fall by the wayside over time, and industry regulations reflect growing understanding of long-term harm.
Shared Responsibility Defines Safety
In my experience, the safest teams don’t just stick to the minimum. People look out for each other, speak up about broken gear, and report unusual odors or damage. Good managers create a culture where reporting a near-miss doesn’t cost anybody their job.
Handling a spill of 1-Bromo-1-Chloro-2,2,2-Trifluoroethane isn’t just about following a checklist. Good habits, open communication, and a willingness to learn each time set the floor for real safety. I’m reminded each time I step into a chemical storage area: nobody handles these risks alone, and each detail counts.