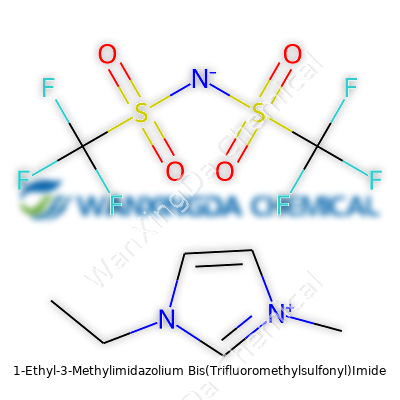
Rethinking Chemistry: A Closer Look at 1-Ethyl-3-Methylimidazolium Bis(Trifluoromethylsulfonyl)Imide
Historical Development
Curiosity has always driven progress in chemistry. The emergence of so-called ionic liquids shook off the usual limits of solvents—flammability, volatility, narrow temperature use. In the late 1990s and early 2000s, researchers sought stable, easy-to-handle solvents for tricky syntheses and green chemistry. 1-Ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide—often called [EMIM][Tf2N]—stood out. Early papers highlighted its thermal stability and almost total lack of measurable vapor pressure. This opened doors in academia and industry. Adoption grew not because the compound was cheap or easy, but because it solved headaches in labs, from electrochemistry to difficult organic transformations. My own work in an electrochemical lab in that era meant handling noxious, smelly chemicals or glassware crusted with dried salt. Using [EMIM][Tf2N], things just felt less like trench warfare, more like progress.
Product Overview
Despite sounding like a mouthful, [EMIM][Tf2N] has a simple architecture at heart: a positively charged imidazolium ring, tacked to a short ethyl and methyl group for stability and solubility, paired with a bulky, highly fluorinated sulfonyl imide anion. This recipe grants the salt its liquid nature at room temperature, and the structure resists easy decomposition. It struck a balance between handling costs and outright performance. Over time, it earned many earning names, especially as the landscape filled with similar chemicals: EMI-TFSI, 1-ethyl-3-methylimidazolium TFSI, or just "EMIM bis(triflylimide)" in some circles.
Physical & Chemical Properties
One feature jumps out: [EMIM][Tf2N] doesn’t boil away in open air. It holds up under heat, tolerating temperatures usually out of reach for classic solvents like acetone or acetonitrile. Because it lacks measurable vapor pressure, spillages don’t turn into safety drills. The chemical sits as a clear, syrupy liquid, heavier than water, with moderate viscosity—sticky compared to ethanol, but nowhere near honey. Because the ion pair distributes its charge, it doesn’t short out electronics or trigger runaway reactions. And, thanks to the fluorinated anion, harsh exposure to air or moisture rarely spoils a batch. This tough, flexible nature supports deep dives into new reaction spaces, battery tech, separation processes, and even protein work.
Technical Specifications & Labeling
Labs across the world depend on accuracy. Tracking assay, purity, presence of water, and residual halides isn’t just a regulatory issue; these factors directly affect experimental results. Out of the bottle, pure [EMIM][Tf2N] offers ion purity above 99 percent. Water represents the enemy. Even a few tenths of a percent take a toll on sensitive reactions, distorting yields. Professional suppliers always provide tight specs, usually indicating water content by Karl Fischer titration and the absence of common contaminants. Safe labeling warns about skin and eye contact hazards, flags the chemical for fluoride content, but generally underscores its low volatility: no need for explosion-proof cabinets. Over the years, better purification steps reduced trace halides and organic byproducts. I remember running conductivity tests, finding that poor-quality batches could skew energy storage results by orders of magnitude—one more reason that labeling isn’t just bureaucracy, but sound science.
Preparation Method
The best route to [EMIM][Tf2N] starts from a simple imidazole base. Chemistry students often learn it’s a two-step game: alkylate the imidazole ring to attach methyl and ethyl chains under controlled conditions, then swap out the old anion (usually a halide) with the bis(trifluoromethanesulfonyl)imide anion. The devil lives in the details—the purity of initial reactants, temperature control during alkylation, and especially the handling of toxic byproducts. Industrial-scale prep optimizes every step, aiming for maximum yield and minimum waste. Most homemade routes never reach this level of efficiency, but careful, clean glassware and frequent checks on reaction progress kept more than one graduate student out of trouble. The challenge comes when scaling up, as the cost of specialty reagents and safe disposal starts to matter.
Chemical Reactions & Modifications
The ionic character of [EMIM][Tf2N] lets it play referee and catalyst across a wide variety of chemical contests. In battery labs, the stability of the ions lets cells run for hundreds of cycles without breaking down, something unthinkable for water-based electrolytes. Skilled chemists tweak the imidazolium ring, swapping out longer alkyl chains or altering the anion for subtle shifts in viscosity, conductivity, hydrophobicity. The compound resists strong acids and bases, takes up organic molecules like a sponge, and can mediate tough couplings and separations. Over time, its frameworks have expanded to tune properties for specialized work, yet the backbone remains the same. From green solvents to reaction media for enzymes, its adaptability stands out. In my experience, curious researchers stretch its function beyond obvious fields, linking it with nanoparticles, or trapping pharmaceuticals for slow release.
Synonyms & Product Names
Acronyms rule the chemical world. [EMIM][Tf2N] alone hosts a crowd of near-synonyms: EMIM TFSI, EMI-TFSI, 1-ethyl-3-methylimidazolium NTf2. Each name highlights a slightly different tradition or focus—some prefer the IUPAC flavor, some stick to lab slang. Catalogs reflect this, and a hasty order from a vendor in a rush can end in the wrong bottle on your desk. I learned the hard way: a mismatched anion once set my whole reaction off-kilter. Confusion eased as the community pushed for standardized naming, although cross-checking remains second nature for experienced researchers.
Safety & Operational Standards
Working with [EMIM][Tf2N] doesn't mean unchecked risk, but it still calls for respect. It isn’t volatile, so inhalation risk stays low, but prolonged skin contact leads to irritation. The compound’s fluorine content carries its own concerns—thermal breakdown produces trace toxic gases, especially in fires. Laboratory standards lean on personal protection: gloves, eye shields, good ventilation. Disposal of spills still matters, since persistence in the environment could endanger water supplies or soil. Regulatory groups classify it in line with similar industrial chemicals, without draconian restrictions. Still, a culture of caution remains vital, especially as students and early-career chemists join the field.
Application Area
The reach of [EMIM][Tf2N] stretches far beyond routine synthesis. Battery developers lean on its conductivity, stable electrochemical window, and tolerance of demanding cycles. In separation technology, its affinity for organic molecules drives new liquid–liquid extraction pathways, particularly where water-based systems fail. Biotechnologists put it to work unfolding proteins or stabilizing enzymes, despite initial fears that ionic liquids might outright kill delicate biomolecules. In lubrication and surface modification, industry profits from its ability to cut friction and withstand harsh loads without gumming up moving parts. From carbon capture pilot plants, to electrode design, to oddball projects in spectroscopy and nanotechnology, the versatility of this single compound never seems to stop. I saw journal after journal fill with creative attempts, some wild, a handful world-changing.
Research & Development
Chemists and engineers never stop pushing. [EMIM][Tf2N] is the standard-bearer for an entire family of ionic liquids. Ongoing research explores property tuning: lengthening alkyl chains, cycling through new anions, and exploring hybrid systems with deep eutectic solvents. Every year, a new crop of students tries to force it into battery chemistries, test it in recycling, or study its effects on cellulose and lignin. Startups tinker with formulations for advanced lubricants or antistatic coatings. Green chemistry advocates hold it up as a replacement for volatile organics in specialty synthesis, although cost and purification headaches slow industry uptake. The dynamism here reflects wider social and economic interests as well: clean energy, safer chemicals, lower emissions, and recyclable processes.
Toxicity Research
As with any new staple, long-term effects remain a concern. Years of research put [EMIM][Tf2N] under scrutiny: cell cultures, soil tests, even fish larvae in some papers. Acute toxicity appears lower than many halogenated organics, but certain byproducts and metabolites prompt more careful investigation. Persistence in the environment raises eyebrows, with studies highlighting slow biodegradation, especially under cold or anaerobic conditions. The ability of the compound to solubilize both hydrophobic and hydrophilic substances leads some critics to warn about unforeseen ecological risks—impacts on aquatic life, bioaccumulation potential, or changes in microbial soil populations. Testing requirements keep growing, mirroring concerns about PFAS compounds and other fluorinated chemicals. I recall adding simple bench protocols—waste segregation, dedicated cleaning—long before any official policy landed.
Future Prospects
Looking ahead, [EMIM][Tf2N] stands at a crossroads. The science suggests promise: a platform for cleaner reactions, safer manufacturing, and new electronic components. Battery makers crave stable, high-voltage-ready solvents. Bioplastics and advanced materials could grow if costs drop. Yet, the path forward demands honest answers about environmental impact, affordability, and end-of-life scenarios. Researchers keep digging for next-generation variants—compounds offering the benefits without the baggage of persistence or high price. My experience says the inertia of decades-old technology can only be broken with clear advantages and lower risks. The world of specialty chemicals moves slowly, but the future still holds room for game-changers, and [EMIM][Tf2N] will continue to fuel debate, surprising results, and maybe greener processes for years to come.
Pushing Past Traditional Limits in Labs
You probably haven’t heard much about 1-Ethyl-3-Methylimidazolium Bis(Trifluoromethylsulfonyl)Imide unless you spend your days in a lab. This ionic liquid, often called simply EMIM-TFSI, isn’t just another chemical on a dusty shelf. People working in research and industry have found that it changes the game for a lot of chemical tasks that used to rely on more toxic, less stable solvents.
Transforming Energy Storage and Electronics
I remember reading about early lithium-ion batteries, where scientists struggled to balance safety with energy capacity. This ionic liquid stepped in with some answers. In batteries and supercapacitors, it doesn’t catch fire easily and can handle high voltages. Electrolytes based on EMIM-TFSI improve lifespan and resilience for the batteries in our phones and electric cars. If we want greener transportation and longer-lasting gadgets, updating what’s inside the battery counts as much as bigger design changes on the outside.
Researchers also explore how EMIM-TFSI helps in advanced electronics. It helps manage charge in transistors and sensors. Its chemical stability means devices last longer, and its low vapour pressure makes leaks and emissions far less of a worry in manufacturing settings.
Making Chemistry Less Harmful
Many traditional solvents give off dangerous fumes or pollute water, but EMIM-TFSI barely evaporates and works at a wide range of temperatures. This means you can run tough reactions or extractions while lowering risks to workers and the environment. Chemists keep searching for solvents that cut down on hazardous waste, and this ionic liquid is a real player in that shift.
Growing Use in Industrial Processing
Beyond the lab, I’ve watched more companies turn to EMIM-TFSI to extract metals from ores and recycle electronic waste. Gold, cobalt, and rare earths can be tricky to pull out cleanly, yet using this ionic liquid instead of harsh acids and bases means less contamination for communities downstream from mines and smelters. Processes become safer for workers, and companies face less penalty from new environmental standards.
Bumps in the Road: Cost and Disposal
Of course, EMIM-TFSI isn’t a cure-all. Manufacturing it remains expensive, and if not managed well, it still poses its own environmental headaches—a problem with many specialized chemicals. I see great promise in research that reuses or recycles the liquid after use, cutting both costs and waste. Strong transparency about life cycle impacts also helps build trust as new chemicals move from lab shelves into factories.
What Comes Next?
People who work with EMIM-TFSI continue to hunt for ways to make the liquid more affordable and easier to recover after use. Investing in green chemistry, stricter guidelines for recovery after industrial processes, and public data on safety and recycling all push progress. As a result, this ionic liquid has become part of a wider shift toward safer, more responsible chemistry. In the end, updating what we use on the molecular level directly shapes how sustainable modern technology can really become.
What Stands Out About Ionic Liquids
My first encounter with an ionic liquid caught me off guard. Instead of the solid crystals I’d expected, here was a glass-clear oil that didn’t smell much and didn’t evaporate in open air. Imagine pouring it out and seeing those droplets linger, barely wanting to move—no cloud of vapor escaping, no sting in the nose. This is what sets these substances apart from the acids and salts most of us know.
In practical work, a common ionic liquid like 1-Butyl-3-methylimidazolium hexafluorophosphate brings to the table very low volatility. Leave it on a bench all day, and it won’t disappear the way ether or acetone will. In labs, folks appreciate this; there’s less fire hazard, less air pollution, and more control. The high thermal stability also surprises people. These liquids can soak up heat—some close to or above 200°C—before breaking down.
Understanding Their Structure and Behavior
At the root, ions make up these liquids. The positive and negative charges stack close together, but the structure is unbalanced just enough to stay in a liquid state at room temperature. This arrangement explains why ionic liquids don’t conduct electricity as quickly as water with dissolved salts, but still do a job. You’ll see researchers testing their conductivity for newer batteries or find them used to dissolve metals during rare-earth extraction.
The density and viscosity of ionic liquids often move closer to syrup than to water—let a drop slide down a pipette and it creeps along, refusing to rush. Handling them reminds me of the first time using heavy mineral oil in a reaction: everything slows down just a notch, and you need patience mixing them with other chemicals.
Why Ionic Liquids Matter for Clean Chemistry
Ionic liquids offer a shot at greener chemistry. Most don’t release heady fumes, which cuts down on air contamination. With organic solvents, I remember fume hoods always roaring to pull vapors away, but here, the risk drops. This property plays a role in safer workspaces and less environmental release—big factors for people thinking long term about sustainability.
Another powerful trait rests with their chemical “tunability.” By tweaking the ions, chemists control solubility, polarity, or even the ability to coordinate with metals. This tailor-made capability opens doors for selective catalysts or recyclable solvents. Companies exploring plastic recycling or pharmaceutical production have tested ionic liquids for dissolution or as reaction media.
Cautions and the Road Ahead
While ionic liquids answer some problems, others remain. The environmental impact isn’t zero—certain varieties break down slowly, and some let go of toxic byproducts. In my opinion, transparent reporting on environmental fate and toxicity should keep pace with new product launches. Peer-reviewed studies already help, flagging risks before these liquids get dumped into rivers.
Real progress would mean two changes. First, more labs should share results about breakdown pathways: what happens when a liquid spills loose in soil or water? Second, chemical suppliers and universities ought to push for liquids built from biodegradable ion pairs. Early versions already show less persistence in test environments, signaling a step in the right direction.
Final Thoughts
Ionic liquids don’t answer every problem, but their unusual blend of non-volatility, chemical flexibility, and thermal stability brings real promise. Success in the coming years will ride on keeping innovation rooted in responsibility, with honest tests and practical limits. Chemists, industries, and regulators all play a part in making sure the next steps for ionic liquids lead to solutions—not headaches.
People often raise questions about handling chemicals that sound as intimidating as 1-Ethyl-3-Methylimidazolium Bis(Trifluoromethylsulfonyl)Imide. The tongue-twisting name signals that it’s no kitchen ingredient, and its growing popularity in research and industry has put it under the microscope for safety.
Why This Chemical Pops Up in Labs
This ionic liquid gets a lot of attention for its ability to dissolve salts and unique stability. More labs use it for battery development, electrochemistry, and some creative organic reactions. On the bench, its near non-volatile nature feels like a bonus—no strong smell, no obvious fumes climbing out of a beaker. That sometimes tricks folks into thinking it’s harmless. The reality is less cut-and-dried.
Assessing the Real Hazards
Looking through published toxicity studies, you won’t find evidence suggesting this chemical is wildly hazardous in trace exposures. There are no dramatic explosive reactions or toxic clouds like the ones we see in classic lab horror stories. Even so, any comfort from its low volatility feels misplaced. The risk doesn’t vanish just because you can’t smell it.
I’ve handled this chemical plenty, mostly in glove boxes and under hoods. The texture fools you: slippery, no sting to the skin, no sharp odor. Lots of ionic liquids fall in this tricky category—just because it behaves doesn’t mean it’s friendly. Some research suggests potential concerns about it soaking through the skin over time or sticking to gloves and getting inadvertently spread around workspaces. Sometimes, the chemical can cause mild irritations. No well-documented cases of severe acute toxicity exist from casual lab contact, but long-term effects remain a question mark. The fluorinated bits in its structure also raise eyebrows among people worried about chemical persistence in the environment.
PPE and Handling – Lessons Learned
No shortcuts replace good lab hygiene. Gloves made from nitrile hold up well, and eye protection shields from accidental splashes, especially during transfers. I always keep my workspace organized and label waste strictly. Open bottles under a hood, not on open benches, and keep the chemical away from food, drink, and skin. Don’t get too casual—things slip through the cracks when attention wanders, and that’s when accidents happen.
Colleagues have told stories about discovering faint residue stains days later, reminding everyone that invisible risks—like residue on gloves transferred to other equipment—can slip past notice. A spill kit and good training help the most. Dumping leftover liquids in the sink seems unthinkable, both for local safety and wider environmental reasons.
The Bigger Picture: Safety Outpaces Comfort
Just because this ionic liquid doesn’t attack senses right away doesn’t mean it’s time to drop the caution. From my own habits and what I see echoed in scientific guidance, treating it with respect pays off. Use PPE, work thoughtfully, and never let routine lull you into carelessness. Regulations and best practices are shaped by real stories—mistakes, bad habits, near-misses. My advice? Stay a little skeptical of anything that seems easy, especially in the lab.
If your team brings this chemical into regular use, build it into your safety training, ask questions, read up on emerging research, and watch for updates. Protect yourself, your coworkers, and the environment—and don’t let the chemical’s mild manners fool you.
Understanding Solubility Beyond the Textbook
Solubility usually crops up as a confusing chapter in school chemistry. It used to trip me up, too, with all those rules about which compounds dissolve in what. In real life, though, what dissolves in water or some fancy organic solvent still shapes a lot of what we encounter—from medicine to cleaning products and even technology. That isn’t chemistry for ivory towers. It’s the chemistry inside the cup of coffee you just sweetened or the aspirin you took for your headache.
Why Water Matters: The Universal Solvent
Everybody calls water the universal solvent for a reason. It dissolves lots of things thanks to its polar structure, but that doesn’t mean it dissolves everything. Toss sodium chloride—plain old table salt—into water, and it vanishes. Drop in a chunk of wax, and it sits there stubbornly unchanged. That’s polarity and molecular structure at work. Polar molecules like water bond well with other polar molecules or anything that can ionize. Charged or polar substances break apart and go their separate ways in water, letting daily life tick along—think of electrolytes mixing into bodily fluids, or sugar disappearing into your morning tea.
Moving Past Water: Organic Solvents and Their Role
Sometimes, water just doesn’t cut it. Organic solvents—like ethanol, acetone, or hexane—show up where water falls short. If something’s nonpolar, oil-based or waxy, it shrugs off water and snuggles right into these solvents. Oil paint won’t budge with water, for example, but turpentine melts it away with no trouble. Getting the right match makes all the difference, whether you’re making painkillers, coating electronics, or cleaning machinery. Organic chemists spend a lot of energy picking solvents that work with the stuff they’re handling, balancing solubility against health and safety concerns.
Why Solubility Impacts Health, Environment, and Industry
Solubility isn’t just technical trivia for lab coats. It decides how medicine enters our bloodstream. If a new drug doesn’t dissolve, it might not reach the cells it’s supposed to help. That means pharmaceutical companies chase down ways to improve how their active ingredients break down in the body, sometimes through clever chemical tweaks or by pairing drugs with “helper” molecules. Cleaning up pollution also leans hard on solubility. Harmful chemicals that dissolve easily in water slip downstream and affect drinking water supplies. Other chemicals cling to organic matter and need special tactics for removal.
Looking Toward Solutions—Responsible Choices and Safer Chemistry
Safer solvents and greener chemistry are no longer fringe ideas. Choosing the right solvent means more than boosting yield—it’s about keeping people and ecosystems healthy. Water often stands out as the preferred pick because it’s safe and easy to handle. Still, many useful compounds demand organic options. Chemists now experiment with plant-derived solvents, ionic liquids, or other creative answers that sidestep hazards linked to petroleum-based solvents.
Getting solubility right can save energy, cut spills, and make recycling possible. Safer choices in industrial settings also protect workers and neighbors, a lesson learned the hard way in places hit by chemical accidents. Even at home, looking for water-based products and reading ingredient labels gives everybody a little more power to keep themselves and the environment safer.
Solubility turns out to be one of those unsung heroes of science. Ignore it, and things stop working—medicines become useless, stains refuse to come out, pollutants linger. Pay attention, and the solutions start to reveal themselves.
Demand, Safety, and Why It Matters
If you ever tangle with ionic liquids, you probably know the name 1-ethyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide. It’s a mouthful and yet, in labs and industries alike, everybody uses the shorthand [EMIM][Tf2N]—a versatile liquid salt that won’t freeze up in the cold or boil away quickly. It handles temperature swings with ease, pushes green chemistry further, and solves some sticky problems in battery and separation projects. In recent years, demand soared as researchers started looking for safer solvents. You can find dozens of studies on the topic: chemists trying to avoid volatile organics are shifting to ionic liquids, and [EMIM][Tf2N] stands out.
Looking for a Supplier
Sourcing this compound boils down to picking the right supplier. You won’t pick it up at the neighborhood hardware store. This isn’t simple acetone. Major chemical distributors like Sigma-Aldrich, Alfa Aesar, and Tokyo Chemical Industry sell it. They ask for details—like a business tax ID or a confirmation you’re working in research—because [EMIM][Tf2N] is a specialty chemical, not something for hobby work. Most of the time, these suppliers will not sell to individuals. Some folks try peer-to-peer buying through chemical networks or academic connections, but the risk of questionable quality or expired stock jumps in those cases.
Purity: The Biggest Decision in Ordering
Purity levels drive cost, and also shape how well the compound works in your setup. If you’re aiming for routine battery testing, 98 percent purity shows up as a standard choice. For electrochemistry, trace metals mess with results, so some labs push for >99% or even >99.5% purity, and the supplier might list "for synthesis" or "electronic grade." Any impurity—moisture, chloride, even a fraction of leftover starting material—can throw off an experiment. The purity you pick should match your work’s demands.
Request a Certificate of Analysis with your batch. Every reputable supplier will provide it, and it’s your only true assurance that what you paid for is what you get. Sweating the small stuff like this matters because some lower-cost sources cut corners and you only catch that mistake after your whole project is on the line. Laboratory-grade [EMIM][Tf2N], which often lands at 98-99 percent, covers most research, while those working in electronics or with demanding sensors spring for the higher, more expensive, ultrapure product.
Impact and The Road Ahead
I’ve noticed more universities encourage students to ask about the supply chain before buying reagents. It saves money and sharpens safety awareness. Part of good science means asking if the product contains what the label claims. A few years back, I saw a group waste weeks because their solvent was contaminated. Results made no sense until chromatography pointed at faulty raw material. That lesson sticks: never skip the authentication step.
We need smarter distribution channels and more transparency about product stewardship—especially as more advanced materials go into consumer products. For now, sticking to trusted distributors and keeping one eye on documentation will keep labs and tech development on the right track.