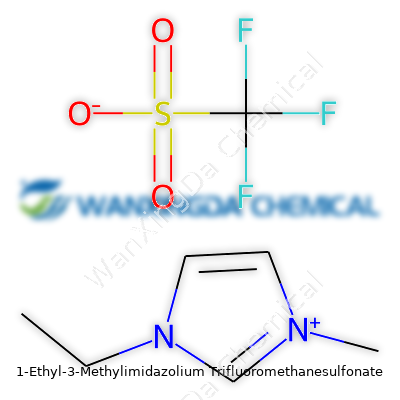
1-Ethyl-3-Methylimidazolium Trifluoromethanesulfonate: More Than Just a Chemical
Historical Development
Looking back at the emergence of room-temperature ionic liquids, a push towards greener chemistry set the stage for 1-ethyl-3-methylimidazolium trifluoromethanesulfonate to appear. Chemists kept running into limits with volatile organic solvents. They needed something less prone to evaporation, offering strong solvation without many downsides. The imidazolium-based ionic liquids, appearing in scientific literature throughout the late 20th century, stood out. By the early 2000s, researchers realized that swapping the anion with triflate (trifluoromethanesulfonate) created a compound pairing high thermal stability with fascinating solvent properties. Since those early papers, this ionic liquid has landed in countless labs, always attached to that deeper promise of cleaner processes and reliable performance.
Product Overview
1-Ethyl-3-methylimidazolium trifluoromethanesulfonate often gets discussed more in academic circles than in everyday conversations, but its influence stretches out in ways that surprise most newcomers. This salt remains liquid at room temperature and rarely gives off a distinct smell. You’ll cross paths with it packaged in airtight containers to keep moisture out—a subtle nod to its hygroscopic character. The product usually takes the form of a clear or pale yellow viscous liquid, a visual cue of its high purity and strong ionic character. People interested in future-oriented solvents or electrolytes should take note; a lot of intellectual energy has gone into making this substance accessible and consistent on the shelf.
Physical & Chemical Properties
The science behind this chemical gets impressive quickly. The melting point drops well below zero on the Celsius scale, while thermal decomposition waits until over 300°C, a span that hints at considerable versatility in the lab. Solubility shines brightest in organic synthesis circles, as this ionic liquid blends nicely with both polar and nonpolar compounds. Low vapor pressure keeps it in place even at elevated temperatures. Its dielectric constant allows it to dissolve a range of substances, acting as a kind of middleman between water and oils when other solvents would fail. People who care about conductivity get drawn in, too, because ions move easily, letting the liquid shine in electrochemistry and batteries. Unlike water or classic organic solvents, 1-ethyl-3-methylimidazolium trifluoromethanesulfonate stands firm against oxidation and reduction within a wide potential window, which matters a lot for high-stress reactions.
Technical Specifications & Labeling
Anyone purchasing this ionic liquid should pay attention to supporting documents that confirm purity, moisture content, and the absence of halide impurities. Appearance tells some of the story, but reputable producers include trace analysis by NMR or ion chromatography to ensure chemical identity. The CAS number and IUPAC name help connect researchers across continents. It gets labeled with transport recommendations since, while not explosive or highly toxic, it is reactive with strong bases and oxidizing agents. The supply chain relies on airtight drums and smaller glass bottles to shield contents from water vapor and air, a simple act with serious payoff for consistency and safety. Shelf life runs long if stored right, but keeping an eye out for cloudiness or changing color is standard practice for those who rely on it daily.
Preparation Method
Synthesizing this chemical starts with imidazole chemistry, no question. Chemists begin by alkylating imidazole with ethyl and methyl halides, then pairing the resulting cation with triflate anion, often from trifluoromethanesulfonic acid or its silver salt. Operational know-how means drying starting materials and solvents to limit water content, followed by rigorous purification—frequently with silica gel chromatography or recrystallization steps. Strict temperature control and air exclusion become non-negotiable as mishaps can ruin an otherwise pure batch. The growing interest in greener, scalable syntheses mirrors trends across specialty chemicals, nudging new research into more sustainable paths for making these ionic liquids.
Chemical Reactions & Modifications
One of the real joys comes from the reactivity of this liquid in both solvent and participant roles. As a solvent, it dissolves organometallics and transition metal complexes, making it an elite pick for catalytic reactions such as Suzuki couplings or Heck reactions. It can stabilize charged intermediates, sometimes driving faster and cleaner product formation. The triflate anion offers some chemical inertness, limiting interference with delicate transformations, which sets it apart from halide-based ionic liquids. Modifications to the cation—including switching alkyl groups—keep popping up in journals, as researchers aim to tailor viscosity, hydrophobicity, and electrochemical windows for specific tasks. This ongoing push for customization keeps the chemistry community engaged, always looking for the next improvement.
Synonyms & Product Names
Nomenclature gets complicated with so many similar-sounding names in the ionic liquid family. On paperwork and barrels, the compound might appear as [EMIM][OTf], 1-ethyl-3-methylimidazolium triflate, or even EMIM triflate. Some catalogs use the IUPAC name: 1-ethyl-3-methylimidazol-1-ium trifluoromethanesulfonate. Cross-checking these names matters, since different sources occasionally use older shorthands. Consistent naming not only avoids confusion in research but also helps connect experimental outcomes across disparate groups and industries.
Safety & Operational Standards
In the lab, handling safety doesn’t come from a single line on a safety sheet; it’s woven into the way people set up glassware, select gloves, and work with ventilation. While this ionic liquid avoids the acute hazards linked to many volatile organics, it demands respect as a powerful solvent. Skin contact should be avoided, and any spills need quick cleanup using absorbent materials. Where chemistry gets scaled up, special attention must turn toward preventing long exposures by controlling vapor and ensuring proper storage. Regular protocol reviews help prevent lapses in PPE use. Eye protection never gets skipped; some people find repeated contact leads to discomfort or mild irritation, so simple cautions go a long way. Incompatibilities with strong acids or oxidizers belong at the top of mental checklists, not buried in a binder.
Application Area
Versatility lands this ionic liquid in a remarkable range of fields. Electrochemistry labs treat it as a secret weapon for lithium-ion battery research, as its wide electrochemical window and non-flammability offer safety advantages unavailable in traditional carbonate solvents. Pharmaceutical chemists test it in drug synthesis, exploiting its ability to dissolve tricky substrates and stabilize sensitive catalysts. Materials science relies on its unique properties to template nanomaterials or synthesize advanced polymers with narrow dispersity. Extraction processes and separations have also seen improvement, pulling rare earth metals or organic compounds out of stubborn mixtures with less waste and lower toxicity. One field where I’ve witnessed substantial gains: green processing, as regulatory pressure builds against volatile organics, and ionic liquids like this one help industrial chemists meet new standards without sacrificing performance.
Research & Development
Journal databases reflect a steady climb in citations and patents focused on 1-ethyl-3-methylimidazolium trifluoromethanesulfonate, which speaks to broad scientific curiosity. Some research targets the fine details of its solvation effects—why certain catalytic cycles run better here than elsewhere. Others dive into optimizing purification or recycling methods after use, since costs per kilogram can stack up for large-volume applications. As green chemistry principles mature, people keep searching for more benign synthesis methods, scrutinizing both environmental impact and cost of raw materials. The ongoing back-and-forth between academic labs and industrial partners helps advance standardization in procedures and pushes the field from small-scale curiosity towards process-scale reliability. Groups focused on computational chemistry pitch in by modeling ionic structure and predicting new properties, speeding up discovery and reducing the time from hypothesis in a notebook to practical deployment.
Toxicity Research
Assessing toxicological risk keeps gaining importance, especially as ionic liquids move from bench to plant. So far, most studies suggest limited acute toxicity, with much lower flammability and volatility than many lab staples. Still, some investigations point out that chronic aquatic toxicity may arise if waste streams go untreated, so responsible disposal matters as much as personal protection during use. Research teams keep scrutinizing breakdown products to spot possible persistence or bioaccumulation. In actual lab life, I’ve seen extra caution with waste collection—liquid left over from reactions gets segregated, and researchers avoid pouring it down drains, opting instead for professional disposal channels. Progress in this area means growing collaboration with environmental chemists and regulatory bodies to proactively address concerns rather than react after incidents.
Future Prospects
Looking forward, interest in this ionic liquid shows no sign of slowing down. Energy storage, especially for next-generation batteries and capacitors, remains an obvious driver. Synthetic chemistry keeps unlocking new transformations that weren’t practical before. Life sciences may soon test modified versions as solvents for enzymes or pharmaceuticals as green chemistry standards tighten. Cross-disciplinary projects—pairing computational materials science with real-world engineering—hint at untapped potential in microelectronics or even carbon capture. The ongoing challenge, as always, is to balance innovation and stewardship. Making sure these advances grow in ways that benefit both science and society takes commitment from every corner: researchers, suppliers, regulators, and end-users all have roles to play. Those who keep investing in deeper understanding will shape not just the next application, but how we think about chemistry’s place in a world chasing sustainability and robust performance.
Understanding the Compound in Today’s Labs
Lab shelves today hold far more than old school acids and bases. As a chemist who’s spent years handling all sorts of liquids, I’ve watched the rise of “ionic liquids” with curiosity and some cautious optimism. Among these, 1-Ethyl-3-Methylimidazolium Trifluoromethanesulfonate—most folks shorten it to EMIM OTF—stands out for enabling things that regular solvents just can’t match.
How EMIM OTF Powers Synthesis
Running a tricky synthetic reaction, especially if it involves sensitive molecules, always demands a careful choice of solvent. Traditional options sometimes ruin yields or leave unwanted byproducts. EMIM OTF doesn’t mix with water, dissolves a wide range of organic and inorganic compounds, and resists breaking apart under heat. These properties let researchers mix molecules that usually refuse to mingle. In practice, that means better results in processes used for pharmaceuticals, advanced materials, or research into new battery chemistry.
Green Chemistry: No Nasty Fumes
Safer chemistry matters. Many older solvents produce nasty fumes, catch fire easily, or stick around in the environment for ages. EMIM OTF carries an edge here. It barely evaporates, reducing inhalation risks in crowded labs and making the air more tolerable for everyone. Plus, since it rarely catches fire, students and seasoned scientists both lower the risks of mishaps.
Electrochemistry on the Rise
Modern batteries and capacitors keep pushing efficiency—and the test bench keeps shifting. EMIM OTF started popping up in labs working on new ways to store energy. Its stability at different voltages and temperatures allows batteries to last longer and charge faster. I’ve seen it help researchers avoid the usual explosions or meltdowns that haunt those working with lithium or sodium devices. A safer electrolyte means research money and time actually go toward results, not cleaning up mistakes.
Challenges in Cost and Disposal
Of course, EMIM OTF isn’t all sunshine. These ionic liquids run expensive, especially when used by the liter. Labs need to handle them with care during disposal—local wastewater systems can’t deal with complex molecules like this. Recycling programs and research into breaking them down after use should get more attention. Universities and chemical manufacturers need to invest in ways to recover or neutralize spent ionic liquids, or their green promise falls flat.
Looking Ahead
No chemical solves every problem. EMIM OTF does open up reactions that seemed out of reach. Its low vapor pressure and electrochemical stability show promise for safer, cleaner research and manufacturing. I’d argue that the wider use of ionic liquids could shift the chemical industry in a more responsible direction—provided labs keep costs and disposal impacts in check. Years tinkering in university labs have shown me: new tools like these can bring real progress, if used mindfully.
Chemistry Deserves a Little Respect—and Attention
It’s easy to overlook the daily realities of chemical storage—until you’re the one responsible for keeping research or industrial operations running safely. 1-Ethyl-3-Methylimidazolium Trifluoromethanesulfonate, or EMIM OTf if you want to save your breath, offers a good lesson in how chemistry demands both understanding and discipline.
EMIM OTf stands out among ionic liquids. It refuses to evaporate like water, and it tends to keep its cool under pressure and temperature climbs. Some might shrug and ask, “If it’s stable, why fuss?” The truth is, chemical storage isn’t just about preventing accidents—it’s about respecting the entire process that stretches from delivery to disposal.
A Humble Bench, or a Better Plan?
Leave an open bottle of EMIM OTf on a lab bench and problems start to roll in. The main worry centers on water. This chemical plays well with moisture—and not in a good way. It soaks up water from the air, and over time, that hidden invasion shifts its physical and chemical behavior. Suddenly, what was once a pure and reliable reagent turns unpredictable. Folks in research and manufacturing already know how one mishap in purity can send results off track or sabotage expensive equipment.
Locked cabinets with clear labeling make for a good first step. The best practice involves choosing an airtight container, glass or high-grade plastic if you want to minimize unexpected reactions. Dry, cool areas win out over shelves near windows or under radiators. The last thing you want is a surprise reaction because heat sped up a process no one thought to double check.
Health and Safety Heads Up
Chemists or technicians who have spent some years in the lab often develop a habit—glancing at labels and grabbing gloves before uncapping a bottle. EMIM OTf isn’t the most volatile or toxic compound out there, but it doesn’t give you a free pass either. No one likes to talk about chemical burns or allergic reactions, but both can show up in any workplace with poor storage discipline.
Bottles with a tight seal prevent spills and minimize fume exposure. Anyone handling this chemical should know where to find eye wash or a spill kit, not just hope for the best. Standard operating procedures, along with refresher courses on safety, make a huge difference in cutting down the number of accidents.
Waste Not, Want Not: Long-Term Thinking
Sometimes a shelf grows crowded with half-used containers, each with someone’s sloppy handwriting and a forgotten use-by date. EMIM OTf loses its edge once compromised by moisture or light. Labs and factories do themselves a favor by rotating stock and tracking their inventory. Old bottles, especially those gathering dust for months, drift toward waste—and in many cases, hazardous waste. Unused chemicals shouldn’t just go down the drain or in the trash.
Teams that take time to establish clear waste collection procedures do more than follow regulations—they show the kind of responsibility that builds trust with co-workers and neighboring communities. That trust comes from visible routines: dated containers, regular audits, and transparent disposal routes managed by specialists.
There’s No Shortcut for Care
No magic trick lets a lab sidestep discipline with chemical storage. Each step—sealing the bottle tight, labeling clearly, tracking expiration, locking it away from moisture and heat—boils down to respect. Whether people work in academic labs, government facilities, or high-volume manufacturing, the message stays the same: don’t let comfort with a chemical dull your attention to storage details. That’s the real foundation for smart, safe science.
Understanding the Substance
1-Ethyl-3-methylimidazolium trifluoromethanesulfonate, sometimes shortened to [EMIM][OTf], belongs to the family of ionic liquids. Scientists and engineers rely on these salts in liquid form for batteries, chemical synthesis, and some industrial cleaning. The buzz around ionic liquids usually centers on claims of higher stability and lower vapor pressure compared to the usual volatile organic solvents.
Sizing Up the Hazards
Many folks outside chemistry labs hear “ionic liquid” and imagine a safer alternative to classic solvents. But not every innovation comes trouble-free. This substance doesn’t evaporate like acetone, but the story doesn’t stop there. For most of us, skin or eye contact would probably cause irritation. Inhalation might not be as much of a worry, thanks to the low volatility—though droplets or mist in the air could still enter lungs if handled carelessly. One study in the early 2010s showed some ionic liquids caused harm to aquatic organisms, raising questions about what happens after they wash down the drain. We often don’t have enough long-term health data, especially for workers handling tons each year. It’s not as familiar or well-studied as acetone or ethanol, which have entire books dedicated to their effects and antidotes.
Weighing the Evidence
Researchers have poked at these compounds for possible toxicity. You can find studies that measure lethal doses in fish, bacteria, and even soil worms. Some ionic liquids knock out enzymes and harm cellular membranes. For [EMIM][OTf] specifically, toxicity seems to fall in the moderate range compared with more aggressive chemicals like formaldehyde or benzene, but “moderate” feels slippery if you’re the one splashing it around without gloves. Chronic hazards—things like reproductive toxicity or carcinogenic potential—haven’t been spelled out with confidence. Lacking decades of workplace stories or big toxicology reports, it makes sense to treat new chemicals with respect, not only excitement.
What Safety Looks Like Day-to-Day
In my own days working in a lab, the main precaution always began with personal protective gear: gloves, goggles, a coat that zipped tightly. In the case of [EMIM][OTf], a splash on bare skin or eyes calls for a quick rinse with plenty of water. Clean-up means soaking up spills with absorbent pads—not sweeping things to the floor or down the sink. Nobody wants to trade one set of problems for another, so people need to keep ionic liquids out of the environment, just as with older solvents. For disposal, chemistry workshops work with professionals who can incinerate or contain waste, not flush it away.
What Could Make It Safer?
Companies and researchers should keep a close eye on exposure data as workers handle new substances. Regular training, proper labeling, and keeping detailed records of who uses what, where, and when, all help. If regulators think a substance creates too much risk for workers, wildlife, or drinking water, rules change or even ban some uses. Listening to feedback from workers who spot skin problems or headaches also alerts people to hazards official reports miss. Substitution sometimes helps—switching to a less toxic cousin—or refining handling protocols to trap every drop before it becomes an environmental release. Science doesn’t have every answer yet, but treating inventive chemicals with patience and humility keeps labs and factories safer for everyone.
A Look Beneath the Surface
Digging into the chemical structure of 1-ethyl-3-methylimidazolium trifluoromethanesulfonate exposes more than a pile of letters and numbers. This isn’t just an esoteric compound: it shows how chemistry crosses from lab curiosity into real-life problem-solving.
The Formula Unraveled
Let’s get to what makes this molecule tick. The cation, 1-ethyl-3-methylimidazolium, comes from the imidazolium family — basically, a five-membered ring with two nitrogens, decorated with an ethyl on one side and a methyl on the other. Pair that with the anion trifluoromethanesulfonate, also called triflate, which piles three fluorine atoms onto a sulfonate group. Chemically, the whole thing lands at C7H11F3N2O3S.
Looking at it on paper, it’s just a string of atoms — but there’s a lot packed in that formula. Imidazolium rings, for example, play nice with plenty of partners. The triflate anion doesn’t get in the way much; it’s stable and loves liquidity. Mixing them gives an ionic liquid with a low melting point, meaning this chemical stays a liquid over a wide temperature range, even well below water’s freezing point.
More Than the Sum of Its Parts
Ionic liquids like this one have built a reputation outside academic conference rooms. Their stability, negligible vapor pressure, and strong solvating ability let them step into roles that solvents like acetone or ether can't always handle safely or efficiently. It’s not hard to see the pull for green chemistry: less environmental blowback from evaporation, less fire risk, and they stick around long enough to be recycled.
Watching the hype grow around ionic liquids, I sometimes wish back to basic undergrad labs, with the sharp smell of volatile solvents. Safety goggles fog up, the constant drone of the fume hoods in the background. Fast-forward to labs experimenting with ionic liquids — no more irritating fumes, less stress about notching the air with clouds of flammable vapor. I’ve seen firsthand how safer solvents lower stress and open the door for more creative experiments, especially for students and new researchers.
Challenges in the Lab and Beyond
It's not all smooth sailing. Making these compounds, purifying them, and making sure they don't carry unwanted water or contaminants can run up costs for small labs. Their ability to dissolve a lot can be a problem, too, when it comes to isolating products or working up reactions. It’s a balancing act: better performance and safety, weighed against the reality of price tags and complicated recycling steps.
Researchers have started building connections — sharing methods for easier synthesis and purification, pooling resources, and lobbying for more support to develop greener, more affordable ionic liquids. The next chapter lies in bridging the lab and industry, scaling up without punching holes in budgets or giving up progress toward more sustainable chemicals.
Looking Forward
The structure and formula of 1-ethyl-3-methylimidazolium trifluoromethanesulfonate give clues to why it’s become a backbone for modern chemistry labs. There’s value in understanding not only what a compound looks like on paper, but what its structure allows us to do out in the real world. Sharing knowledge across academic and industrial boundaries, and keeping an eye on costs and risks, will shape how compounds like this are used for greener, safer chemistry down the line.
A Look at Ionic Liquids Beyond the Hype
People have been searching for better materials for electrochemistry for decades. In this search, ionic liquids often made headlines—mainly because they can jump many hurdles that regular solvents can't. One example is 1-ethyl-3-methylimidazolium trifluoromethanesulfonate, sometimes written as [EMIM][OTf]. Anyone who's ever fiddled with batteries, supercapacitors, or electroplating baths knows standard solvents don’t always play nice with high voltages or long runtimes. These new kinds of salts may look weird at first—they're liquids at room temperature and made entirely from ions—but their promise is real.
What Makes [EMIM][OTf] So Interesting?
I've worked on lithium-ion battery research and dealt with all kinds of solvents that break down at high temperature or get in trouble when currents get intense. [EMIM][OTf] offers a shot at stability that beats many common solutions. It won’t evaporate much, so you don’t get that choking odor in the lab or a mess in your glovebox. You can stretch its electrochemical window—meaning it can support reactions at much higher or lower voltages before breaking apart. This opens up room for bigger battery capacities or sleeker supercapacitor designs.
Conductivity comes up in any electrochemistry discussion. A liquid that can’t move ions is just dead weight. [EMIM][OTf] generally scores well here, though it doesn’t always top the charts. In lithium or sodium batteries, conductivity drops if you add too much salt or start using heavy, sluggish ions. Still, for many applications, it’s more than enough to do the job.
Downsides Often Hide in Plain Sight
Everyone talks up the green side of ionic liquids—no vapors, low flammability—but there are always hidden costs. Some ionic liquids end up being hard to recycle or pricey to make. I’ve watched more than one startup balk at specialty solvents after seeing a bill for a few grams. Plus, the triflate anion in [EMIM][OTf] gives it an edge in chemical stability, but it can mess with certain metals as it sometimes causes corrosion or unwanted byproducts in plating. End-users with a focus on cost and supply chain security need to ask tough questions about sourcing and lifecycle.
Another practical snag: water always tries sneaking in. Even in a well-sealed lab, these liquids tend to soak up moisture from the air. Too much water can wreck performance, dropping the electrochemical window or fostering side reactions that kill longevity, especially in high-voltage devices. Anyone moving out of the lab and into real-world settings—making big batteries, scaling up manufacturing—can’t ignore humidity controls and purification steps.
Where [EMIM][OTf] Shines and How to Fix Its Weak Spots
This liquid finds its best use in scenarios demanding high voltage and chemical resilience, like advanced capacitors or new plating baths for specialty metals. Researchers keep tinkering with different cations and anions, tuning the mix so it runs cooler, lasts longer, or gets cheaper. Partnering the right additives—say, water scavengers or corrosion inhibitors—could tamp down the biggest hassles and unlock even better performance. In my view, teams that spend time tailoring ionic liquids to their use cases, handling them like specialty ingredients instead of miracle cures, will see the biggest payoffs.
Bottom line: [EMIM][OTf] has a real future in electrochemistry for folks who know what they’re dealing with, stay vigilant about cost and water, and keep adapting their recipes as the technology matures. Big leaps often come from small tweaks, and ionic liquids like this one give us a colorful new box of tools for the job ahead.