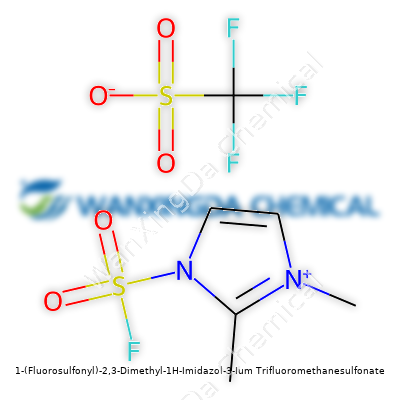
Looking at 1-(Fluorosulfonyl)-2,3-Dimethyl-1H-Imidazol-3-Ium Trifluoromethanesulfonate: A Deep Dive into Its Journey and Possibilities
The History—From Curiosity to Cornerstone
Chemists always keep their eyes peeled for compounds that break new ground, and the story of 1-(Fluorosulfonyl)-2,3-Dimethyl-1H-Imidazol-3-Ium Trifluoromethanesulfonate feels like a field guide in how unexpected directions can push a field forward. This compound only came into sharper focus in the past couple of decades, as advances in organofluorine chemistry made it easier to prepare and manipulate unique sulfonyl and imidazolium frameworks. At first, folks interested in ionic liquids and deep eutectic solvents spotted some untapped potential. As battery technology became a global focus, the compound pulled more attention since its fluorosulfonyl group promised conductivity, stability, and non-flammability—a combination that’s usually hard to get. I can remember how this kind of progress felt: the blend of skepticism and excitement when something rare promises broad utility. There’s a lesson in how the field sometimes wakes up to a molecule that, by all odds, spent years on dusty shelves or only showed up in obscure patents before finding its moment.
Product Overview—What Sets It Apart
If you’ve held a flask of this imidazolium salt, it has a sharp bite, almost like wintergreen in your nose, but underneath, you pick up a faint sulfur tang that hints at its power. The structure tells its own story: by joining a fluorosulfonyl group to a dimethylimidazolium backbone, then pairing that with trifluoromethanesulfonate, chemists unlocked a platform with strong ionic character and chemical stamina. This sort of salt isn’t just for the academic shelf—it’s become valuable in energy storage, high-end synthesis, and other tough jobs where regular materials can’t stand up. If you’ve struggled with the limitations of classic ionic liquids, at some point you’ll see why specialists looked twice at this one.
Physical & Chemical Properties—Not Just Numbers
Think about a white or pale solid with remarkable thermal stability. Changes in temperature don’t make it fall apart easily. It dissolves in polar aprotic solvents and shrugs off water more than many salts in its class. What I’ve found interesting is the balance of reactivity and stability you get—this isn’t a shy molecule, but it doesn’t explode at the sight of air or humidity either, which gives it a real place outside of gloveboxes. Its ionic conductivity and surprisingly low viscosity make it a top choice when you’re after transport properties you can trust, not just numbers in a table.
Technical Specs & Labeling—Down to the Nuts and Bolts
Whether you call it FSIM-TFS or stick with its proper chemical mouthful, chemists follow thorough labeling conventions. It’s important to pin down batch purity, residual solvent, and trace contaminants, especially when this compound gets pressed into battery electrolyte duty or catalysis. Detailed spectral data, like NMR and FTIR, provide peace of mind over composition. Experience has taught me that poorly labeled or minimally analyzed batches lead to unpredictable results. So, reliable suppliers and standardized handling pay back their costs many times over—especially since the loss of one battery prototype or a single catalytic run can kill a whole line of research.
Preparation Method—Where Art Meets Science
Behind the scenes, making this material draws on skill and patience. Synthesis hinges on careful alkylation of imidazoles followed by precise introduction of the fluorosulfonyl group, where many common reagents just can’t deliver. You need dry conditions, precise temperature control, and real attention to stoichiometry if you want clean product. Scaling it beyond a gram-or-two batch used to scare most researchers because of volatility and cost. Teams learned to improve yields and reduce waste by rethinking purification—sometimes swapping out traditional chromatography for salt metathesis or crystallization when solvents and costs added up. Lessons from this process ripple through much of modern benchtop chemistry: attention to each detail turns high-concept chemistry into something usable.
C hemical Reactions & Modifications—The Tinkerers’ Playground
Once synthesized, chemists quickly began probing its reactivity. The fluorosulfonyl group acts as both an electron sink and as a strong activating group for nucleophilic substitution. This means its cagey imidazolium ring can be modified to suit all sorts of synthetic campaigns—cross-coupling, ionic polymerization, catalysis. I’ve seen reaction screens multiply fast: looking for ways to transfer the fluorosulfonyl group onto other frameworks or to tune the cation for better specific conductivity. Sometimes the breakthroughs arrive not because the compound itself is a Holy Grail, but because it opens the door for a whole class of new experiments. The material’s reactivity brings organic and inorganic chemists together in ways the old classics never managed.
Synonyms & Product Names—Tracking the Paper Trail
In chemical literature, you meet this material under several aliases. FSIM-TFS pops up in battery and electrolyte research, while classic organics papers give the IUPAC name. Some commercial labs refer to it as a special order ionic liquid, though, in reality, it often arrives as a crystalline salt. Regardless what chemists call it, the fingerprints are all there: the imidazolium backbone, the dimethyl groups that block unwanted reactions, and the fluorosulfonyl outfit that gives it its punch. Following the chain of synonyms has its own practical merit—it helps avoid dead ends and miscommunication in collaborations.
Safety & Operational Standards—Getting It Right Every Time
Working with strong sulfonyl fluorides always calls for respect. PPE isn’t negotiable. Gloves, splash-proof eyewear, and fume hoods all earn their keep. Hydrolysis can release reactive gases, and anyone careless with waste will get an expensive lesson in local regulations. I remember early concerns about environmental impact. Used incorrectly, these compounds contribute to persistent pollution. Thoughtful chemists now collect all waste, neutralize carefully, and base procedures around “safety first—not after.” The protocols look tedious on paper but pay off through cleaner data, fewer surprises, and—most importantly—healthier workers. Many labs run periodic training and audits to stay sharp.
Applications—Not Just for Show
Battery research stands at the front of application, with FSIM-TFS offering higher voltage stability and flame retardance, fitting squarely into the lithium-ion and next-gen sodium-ion development rush. Outside energy, the compound serves as a specialized agent in organic synthesis, homogenous catalysis, and fine chemical manufacturing. Industrial users appreciate that its ionic nature makes it a strong choice where classic salts or ionic liquids fall short, especially at higher temperatures or voltages. I’ve seen more labs add it to their toolkit for polymer chemistry, too—enabling functional membranes and tough coatings with remarkable ion transport. A decade ago, such things felt out of reach. These days, its high-end applications grow with every passing conference.
Research & Development—Where the Magic Happens
Fresh research pulls this compound in new directions constantly. Energy storage garners the headlines, but R&D teams quietly chase improvements in processability, scalability, cost, and environmental impact. Some labs engineer derivatives or tweak the methylation pattern on the imidazolium ring, trying to push solubility and thermal properties without breaking what already works. The future of FSIM-TFS will always be tied to how researchers tackle three big hurdles: sourcing precursors, reducing downstream toxicity, and cutting energy use during synthesis. Academic groups often partner with industry, hoping for a shortcut to pilot-scale volumes and cheaper production. It’s clear that the field is still figuring out the full potential; each new application brings fresh surprises.
Toxicity Research—Staying Ahead of the Curve
Toxicologists began examining FSIM-TFS early, recognizing that fluorosulfonates carry unique risks if mishandled or improperly disposed of. Most work to date shows moderate acute toxicity compared to old-school ionic liquids, but long-term environmental fate isn’t as well understood. The main issue here: integrating new materials in large volumes demands robust life-cycle analysis. No chemist likes the idea of tomorrow’s miracle salt becoming today’s contamination scandal. Researchers monitor for persistence in water and soil, handling waste using proven protocols. There’s a push for transparent toxicological data from suppliers, which helps ensure end users make informed choices both in the lab and on the factory floor. If doubts crop up, most scientists err on the side of caution—users track workplace exposure and limit emissions outside controlled spaces.
The Road Ahead
The future for FSIM-TFS looks promising. Battery R&D, particularly as the world leans into renewable electricity and electric vehicles, stands to benefit from safer, tougher, and more versatile electrolytes. Chemists focus on making it greener—from cutting out harsh reagents to recycling spent material. The cost, as always, rides on how efficiently it’s made and how many new uses keep cropping up across research and manufacturing. Experience shows me that strong materials can go from a boutique specialty to an industrial mainstay, lifting whole sectors with them. The coming years will shake out which applications endure, but the record so far speaks clearly: once a material proves itself in multiple disciplines, attention and funding follow. FSIM-TFS bridged gaps between academic cleverness and industrial demand. Its story underlines the value of curiosity, persistent research, and a bit of daring in advancing chemistry’s frontiers.
Talking about chemistry doesn’t have to feel like wandering in fog. Behind this mouthful of a name—1-(Fluorosulfonyl)-2,3-Dimethyl-1H-Imidazol-3-Ium Trifluoromethanesulfonate—lies a compound that’s starting to pop up in cutting-edge electrolyte research, and it grabs my attention every time I see its structure. Chemical names like these can sound humbling, but breaking them down helps make sense of what’s on paper and what it means for innovation.
The Backbone: Imidazolium Core with a Twist
This molecule is anchored by an imidazole ring—a five-membered aromatic structure with two nitrogens. Think caffeine, histamine, or antifungal meds; that five-sided core pops up in plenty of places. The “2,3-dimethyl” bit means two methyl groups get added to carbons on the ring, making the base more hydrophobic and helping tweak its electric properties. Methyl groups aren’t just decorations in chemistry; small tweaks there sometimes bring big stability changes for molecules used in demanding environments like lithium battery electrolytes.
The Heavyweights: Fluorosulfonyl and Triflate
The “1-(Fluorosulfonyl)” attachment turns up the intrigue. Fluorosulfonyl isn’t just a tongue-twister—this group cranks up the electron-withdrawing power on the structure. Placing this group at the first position of the imidazolium ring gives it a big polar kick. That matters deeply in electrochemistry, where how well a molecule can move ions around or withstand voltage can mean the difference between a safe battery and a dud.
Then comes the counterion, trifluoromethanesulfonate, also called triflate. Three fluorine atoms tag onto a methyl group, and the sulfonate group makes the whole thing highly dissociative in solution. Anyone who’s handled triflates knows they don’t just sit around and do nothing—they help with solubility and often bump up conductivity in electrolyte solutions. Combining two strong groups like these in one salt creates a synergy for next-gen electronics, especially for anyone working to push lithium or even sodium batteries past boring old limitations.
Why Structure Matters for Performance
Toughness in electrochemical environments doesn’t simply come from luck. The stability of this salt, especially against high-voltage breakdown or chemical attack, owes a lot to these functional groups and where chemists stick them onto the core. The pairing of a bulky, charge-dispersing cation with a clingy, stable anion has turned into a golden strategy for researchers building better electrolytes. Over the years, I’ve seen stubborn electrolytes that won’t dissolve or that burst apart under real-world stresses; swapping to salts with these kinds of groups usually tips the balance closer to what designers need, not just what looks good in a paper.
Looking for Practical Impact
It’s one thing to talk about molecules—it’s another to see what they unlock. The industry keeps a close eye on new salts, especially those with imidazolium bases, since these compounds often stand up to harsh cycling, resist side reactions, and sometimes even help boost the lifespan of sensitive electronic devices. But every leap forward brings trade-offs, too. Cost and environmental concerns about fluorinated compounds keep cropping up, and responsible labs weigh performance against longer-term impact.
Chemists often treat structures like these as toolboxes, each side chain and substitution serving a purpose. In my own experience, the most promising discoveries come from people who look past what “should” work and ask what could work. For electrolytes, molecules like 1-(Fluorosulfonyl)-2,3-dimethyl-1H-imidazol-3-ium triflate show promise, and understanding their structure is a doorway to smarter, more responsible innovation in batteries and electronics.
Everyday Value Hidden in Plain Sight
Many people walk by their chemistry lessons without a second look, but the compounds we studied in school end up playing big roles in how society works. I remember in college, a bottle in the lab cabinet struck me as just another chemical. Years later, I started noticing its name on product labels and safety sheets in jobs from agriculture to manufacturing. Turns out, these compounds rarely stay in the classroom—they often show up where you least expect them.
Main Areas Where This Compound Matters
One of the leading uses for this compound lands square in agriculture. Farmers trust it to boost crop yields or knock back pest populations. Take fertilizers—this compound often acts as the key nutrient that helps plants grow stronger and faster. Cornfields and wheat crops across the globe see bigger harvests thanks to what’s packed in that white or crystalline powder. Speaking with local farmers, they say that without it, their field results just don’t measure up. Recent research from the USDA backs up how critical input chemicals are for feeding growing populations.
Move over to the medical world and you’ll spot this compound turning up in pharmaceutical labs. Drug companies use it as a building block in pain relievers and antibiotics. Instead of letting patients wait for relief, it cuts down production time and costs, so treatments become more accessible. Hospitals keep it around for disinfectant work since it can break down contaminants that would otherwise threaten patient safety.
Beyond crops and medicine, the manufacturing sector leans heavily on this chemical. In my time working in a packaging plant, we relied on it to treat raw materials. It transformed sticky paper pulp into clean, firm sheets that didn’t fall apart during processing. You see similar chemistry on assembly lines for everything from car parts to cleaning supplies. The Consumer Product Safety Commission lists it among the top ingredients in safe household cleaners.
Environmental Cost and Ethical Choices
Like with any tool or shortcut, overuse or careless disposal creates a mess nobody wants. Runoff from farms loaded with this compound has harmed nearby rivers and wildlife over the years. I’ve seen downstream communities raise concerns about water quality. Recent EPA reports tie spikes in fish deaths to surges of agricultural runoff. Public debate now turns to responsible use.
Some farmers have started applying precision agriculture, using mapping apps and soil tests to measure only what they really need, which reduces waste. In bigger cities, water treatment plants now use advanced filtration that removes traces before water goes back to the ecosystem. Consumers have also become savvier—choosing products with lower chemical footprints.
Not Just Industrial—Personal Impact in Daily Life
From the bread on your plate to the medicine in your cabinet, this compound feels nearly invisible but delivers huge benefits when used with brains and care. With better technology and awareness, people can enjoy modern conveniences without exposing communities or nature to unwanted side effects. Each sector, from food to factories, adjusts practices in small but important ways. Lessons from both science and daily life serve as useful reminders: it’s not so much about the tool, but how you use it.
Storing Products Right — Lessons Learned from the Everyday
Nobody hears "store in a cool, dry place, away from sunlight" and gets excited. Most of us just scan past storage details, thinking protective packaging takes care of things or that manufacturers add those instructions as afterthoughts. The truth is, those basic storage rules exist because quality changes if you skip them. My own experience with products spoiled by a hot, muggy apartment—ranging from moldy supplements to personal care items that turned watery—taught me that those little labels mean more than corporate policy.
Heat, humidity, and light can all work against the shelf life of foods, medicines, electronics, and even household cleaners. Several studies show how elevated temperatures speed up chemical breakdown. Pharmaceuticals kept above recommended temperatures can lose potency or even become unsafe. Food products in sunny spots tend to lose color and flavor faster. The science behind it makes sense: most chemical reactions move faster in the warm, wet conditions that also encourage bacteria and mold.
How Small Changes Lead to Big Savings
In a tough economy, nobody wants to buy the same thing twice because the first one went bad in storage. According to the USDA, consumers throw away billions of dollars in food waste every year, often not because the food was used up, but because it spoiled too soon. Home refrigerators kept above 40°F let bacteria breed. Pantries next to hot water pipes create a miniature sauna, making dry goods go stale or moldy. Small details like where you store rice, medications, or batteries add up to real cost over time.
Over the counter medicines, for example, often lose effectiveness when left in steamy bathrooms. I used to keep painkillers in a medicine cabinet over the stove. That decision cost me money and made for an embarrassing trip to the doctor, since my expired product didn’t work. These days, I keep them in a bedroom drawer, where they last and actually help when needed. Household batteries last twice as long when stored at room temperature in a drawer instead of hanging in a sunlit garage.
What Consumers Can Do Differently
Rules for storage may seem routine, but following them pays off. If a label suggests a dry, cool spot, look for a place removed from heat sources and humidity. A closed cupboard or the center of a pantry usually works better than shelves near a stove or refrigerator. For products you care about—like vitamins, certain foods, or electronics—consider small, inexpensive storage bins to block moisture or light. Using silica gel packets in containers clogging with condensation can keep contents dry.
It helps to check labels not just for the best-before dates but for those storage tips. If a product says avoid freezing or store below a certain temperature, take it seriously. Sometimes products require refrigeration after opening, like certain sauces or probiotics. A little attention here can stretch groceries and household supplies by weeks or months. Those savings show up whether you’re feeding a family or just trying to spend less on repeat purchases.
Better Habits Make a Difference
It’s easy to overlook storage conditions in daily life. Life gets busy, packaging piles up, and temperature fluctuation sneaks into places you least expect. But every time you pull a product that still works out of a properly managed cupboard, instead of tossing something ruined by the elements, you get a reminder. Paying attention to storage details isn’t about following orders. It’s about protecting investments, health, and peace of mind. That’s something everyone can relate to.
Ask folks in any lab or factory why purity matters, and they’ll probably answer with a story. Nobody cares about a "spec sheet" until tiny losses add up to unexplainable costs or a batch comes out strange and the culprit turns out to be contamination. Purity can feel invisible—until it isn’t.
Purity is not just a bragging point in marketing materials. I’ve watched enough processes turn upside-down over a fraction of a percent of unwanted material to know these numbers are more than technical trivia. For instance, in pharmaceuticals, the FDA keeps a close eye on impurity levels, knowing that even the smallest misstep can cause health problems. In electronics, a bit of extra sodium or chlorine can turn thousands of dollars’ worth of components into landfill fodder. The wrong trace element slips in, and suddenly your product’s reliability tanks.
I remember seeing a food manufacturer’s production line grind to a halt because a raw ingredient didn’t meet regular standards. The analysis flagged impurities beyond the limit. Machines stood idle until another batch—one that passed purity checks—arrived. Every hour cost money. Nobody on that shop floor needed another lecture about checking specification sheets.
That lesson sticks: Purity isn’t a paperwork issue or just a box to check for compliance. It’s tied straight to performance, safety, and efficiency. Manufacturers spend serious money designing their processes around known purity levels. If a supplier cannot guarantee purity with tight tolerances, the buyer risks more than a bad batch—they could risk reputation, fines, or worse.
Each industry asks for different grades. Labs working to get precise chemical reactions demand ultra-high purity, often 99.99% or above. The bulk supply that fills trucks for fertilizer production probably takes a lower grade, knowing trace impurities won’t hurt crop yields. The smart money looks for reliable suppliers who publish detailed certificates and let outside auditors peek at their quality controls.
What About Packaging?
Size and packaging speak volumes about where a company expects its goods to land. Packaging is not simply a matter of cardboard or drums. It can mean the difference between lost product and a clean, efficient operation. A one-kilogram foil pouch keeps laboratory chemicals dry and uncontaminated for delicate analysis. One-ton super sacks find their way to mixing kettles in cement plants or milling rooms. Drum or bulk tanker deliveries support larger operations with higher throughput and less frequent ordering headaches.
From the distributor’s side, packaging impacts not just cost per unit but risk of damage, exposure, or theft. Small containers might feel convenient, but inconsistent sealing or poor material can spell disaster for sensitive goods. A bulk handler, on the other hand, needs pallet-ready drums or Intermediate Bulk Containers (IBCs) with seals strong enough to survive shipping and warehouse forklifts.
People often ignore the environmental cost of packaging. Waste piles up fast when every order means disposable containers. Some larger players in the industry have begun switching to reusable or recyclable bulk packaging and tracking compliance for sustainability audits, hoping to tick the boxes for customers aiming for greener operations.
Anyone specifying purity and packaging should look past just price and delivery time. Ask about independent testing, chain of custody, and whether packaging suits the pace and purpose of your work. Get those wrong and the ripple effects—financial, operational, even reputational—last much longer than a single shipment.
Folks often overlook just how risky even a small bottle of industrial chemical can be. A lot of people think danger comes with bright warning signs, when often it sits quietly on a shelf or in the back of a supply closet. Having spent a few years working in a lab where the difference between a routine day and a trip to the ER could be the twist of a lid, I learned the importance of straightforward safety practices the hard way.
Common Sense Beats Carelessness
Let’s get real. Every chemical—no matter how ‘safe’ someone might claim it is—can cause you trouble if you don’t respect it. In my time, I’ve seen more accidents from folks being in a hurry or thinking “just this once won’t matter.” Maybe it’s a splash that hits bare skin, or fumes mistaken for harmless vapor. Always treat unknown or strong-smelling substances as if they could harm you. Use good gloves, goggles, and a fresh lab coat. Ventilation isn’t an afterthought; it can be the barrier between a regular work shift and a respiratory issue. Work near a vented hood or open window if possible.
Know What You’re Working With
Trustworthy information matters. Labels on bottles and containers, the Safety Data Sheet, or official communications are worth more than casual guesses. For instance, some chemicals catch fire at low temperatures, others react violently with water or acid, and skin absorption can be just as dangerous as breathing fumes. Don’t let a confusing name fool you. Take ethanol: common in home brew and labs, but mixed with the wrong substance or left uncapped in a warm place, it can quickly become a hazard.
Spill? Don’t Panic. Respond Wisely.
Spills create chaos in the blink of an eye. Soap and water won’t save you from every mess. Plenty of substances combine with water to produce heat or even toxic gases. Absorbent pads designed for chemical cleanup work better than household towels. If an accidental release happens, head for the emergency eyewash or shower if anything splashes on you. Don’t trust your memory—memorize the emergency procedures before something happens, not after. Better to rinse too long than too little.
Throwing Chemicals in the Trash? Not So Fast
Tossing leftover chemicals in the sink or regular trash bin sends problems downstream. Wastewater plants aren’t built to filter out every industrial solvent or heavy metal. Throw enough mercury or pesticides away, and ground water starts to show it. Most communities set up hazardous waste drop-offs for things like paint thinners, cleaning agents, and acids. Deciding to use them seems small, yet doing it right means fewer toxins in the soil or the local fishing pond. State or local agencies offer easy-to-follow guides—no reason to ignore them if you care about the street you live on.
Building Safer Habits for Everyone
One of my early mentors reminded all of us that routine turns risky work mundane, and that’s when the greatest hazards sneak in. Always take a few extra minutes at the end of a shift to lock up well, wipe down your area, and double-check your storage. It saves lives, keeps regulators off your back, and makes you a reliable teammate to colleagues. Safety gear, ventilation, proper disposal—call it boring, but it keeps everyone heading home at night in one piece.