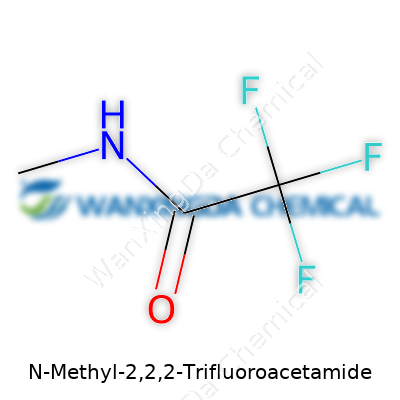
The Remarkable Story of N-Methyl-2,2,2-Trifluoroacetamide
Historical Development: Tracing Steps in Chemistry’s Progress
Chemistry fans like me often see progress through the arrival of new molecules that carve out unique places in the lab. N-Methyl-2,2,2-Trifluoroacetamide has that kind of story. Looking back decades, organic chemists sought to push the boundaries of fluorinated compounds, hoping for new reactivity and durability in synthetic challenges. The inspiration came during the boom in medicinal chemistry, when introducing fluorine into molecules could transform their behavior—altering bioavailability, stability, and even biological targeting. Sometime in the last century, labs started playing with methylated amides and strong electron-withdrawing groups like trifluoroacetyl, chasing those benefits. That’s how N-Methyl-2,2,2-Trifluoroacetamide found its way from chemical curiosity to a regular character in research, often tasked with selective acylations or protecting groups. Its journey mirrors how academic and pharmaceutical research relied on tweaks to molecular structure to open up broader fields.
Getting to Know the Compound: Physicochemical Identity
A closer look at N-Methyl-2,2,2-Trifluoroacetamide makes its place in the lab clear. Sporting a trifluoromethyl group attached to an acetamide framework, this compound stands out for its volatility, stubborn resistance against hydrolysis, and its unique solubility profile, which often makes it a favorite in organic reactions dealing with harsh conditions. The trifluoromethyl part throws its weight around, suppressing nucleophilic attack and lending strong electron-withdrawing power, so reaction outcomes can shift compared to amides without fluorinated groups. Most bottles contain a clear, nearly colorless liquid, sometimes with a faint, sharp chemical aroma. Boiling points and density matter a lot in practice, especially in purification and reaction design—N-Methyl-2,2,2-Trifluoroacetamide sits in a midrange that allows both distillation and solvent extraction. Its practical value often comes from its predictable handling in the lab, with minimal surprises for those who use it right.
Labeling, Specifications, and Getting It Right
Any chemist who spends time in the stockroom knows the importance of sharp labeling and clear technical specifications. With N-Methyl-2,2,2-Trifluoroacetamide, you’ll run into synonyms like Methyl Trifluoroacetamide or sometimes even common shorthand in older papers. Correct molecular structure and purity underpin safe use—impurities can completely redirect a reaction or introduce safety concerns. Reliable suppliers attach strict technical specs, from minimum purity by GC to established boiling point ranges. Detailed labeling, including the correct CAS number and warnings about storage conditions, helps prevent confusion—especially since misidentification can snowball through a research project. These practical details might seem bureaucratic, but they protect against costly mistakes and save a lot of troubleshooting.
Method of Preparation: The Synthesizer’s Approach
Setting out to make N-Methyl-2,2,2-Trifluoroacetamide calls on experience and a solid grasp of reactivity. Most typical routes start with methylamine and trifluoroacetic anhydride or acid chloride, producing the amide in good yield under controlled, dry conditions. The choice of base, solvent, and temperature tip the scales on yield and purity. One thing I learned after years at the bench: reagent quality affects the process as much as the workup. After the main step, washing and distillation remove side products and solvent traces. Sometimes, the route uses alternate starting materials, like esters, but the anhydride pathway remains the stand-by for efficiency and scalability. Complications do arise—humidity can ruin yields, exothermic mixing catches the careless off guard, and the reaction’s exergonic nature means set-up and monitoring matter more than most textbook routes suggest.
Reactivity and Tinkering: Handling Chemical Relationships
Once in hand, the chemistry of N-Methyl-2,2,2-Trifluoroacetamide reflects both its structure and the hefty influence of three fluorines. It can act as a mild acylating agent, sometimes helping install trifluoroacetyl groups onto nucleophiles, though it doesn't match the reactivity of the chloride or anhydride versions. The methyl group on nitrogen closes off some attack sites, boosting stability over simpler analogs, but certain strong bases or nucleophiles can still coax it into reaction. Modification usually means swapping the methyl for something bulkier, or manipulating the backbone to prepare derivatives useful as enzyme inhibitors or building blocks in drug discovery. The delicate, yet reliable behavior of the compound in challenging syntheses gives synthetic chemists a dependable tool, especially for protecting group strategies or introducing robust electron-withdrawing power. In my own experience, its relative inertness compared to similar compounds can sometimes frustrate progress, but it’s this mildness that encourages its use in multistep syntheses where over-reactivity can spell disaster.
Synonyms and Aliases: The Language of Chemistry
Names matter a lot. N-Methyl-2,2,2-Trifluoroacetamide has worn several over the decades. Lab books might show Methyl Trifluoroacetamide, N-Methyltrifluoroacetamide, or just TFMA in shorthand. These synonyms crop up in different languages, supplier catalogs, or old articles, adding to confusion if only a single name is checked. Being thorough in searches saves time and avoids ordering mistakes, and reviewing chemical abstracts service listings helps clarify structure versus isomer or analog confusion. In the decade I’ve spent digging through journals and patents, missed aliases have derailed more than one search for a crucial reaction recipe, especially with these mid-century compounds where nomenclature rules shifted over time.
Safety: A Chemist’s Realities
Safety discussion never gets old for those who actually use chemicals like N-Methyl-2,2,2-Trifluoroacetamide. The trifluoromethyl group brings promise but also creates handling risks. This compound typically resists rapid decomposition, but under high heat or with strong bases, it can spit out toxic or irritating fumes—think HF and acyl fluoride, both unwelcome in any uncontrolled environment. Gloves and eye protection quickly move from suggestion to necessity, especially with the volatility and potential for skin or mucous membrane irritation. The strong aroma has helped more than one researcher notice a leak or spill before instruments did. Proper ventilation is non-negotiable, both in scale-up and simple bench work. I’ve seen and heard of accidental exposures that serve as constant reminders—not just for legal compliance but for daily health. Waste streams demand careful tracking, as organofluorine residues can persist and complicate environmental management.
Where It Shows Up: Application Areas
N-Methyl-2,2,2-Trifluoroacetamide appears across applications, especially where the stability and selectivity of fluorinated amides make a difference. Synthetic organic chemistry appreciates its mildness as a protecting group or acyl source—the ability to resist unwanted side reactions can make or break a route, particularly in pharmaceuticals, where selectivity cuts downstream costs. The electronic effects help medicinal chemists tweak a drug’s absorption or metabolic pathway, searching for molecules that slip past enzymes or transporters. Sometimes it’s a tool for making radiolabeled or isotopically tagged trifluoroacetamides, used to trace metabolism or binding in vivo. Analytical chemists tap into its volatility for preparing test samples in gas chromatography-mass spectrometry (GC-MS). These real-world roles mean demand comes both from research and production chemistry, with new niches opening up as the field of fluorine chemistry keeps growing.
Innovation and the Long Game: Research & Development
Chemists rarely leave good tools alone, so N-Methyl-2,2,2-Trifluoroacetamide gets stretched into new territory every few years. Recent research directions hit on greener and safer production methods, chasing ways to avoid exotic or hazardous reagents that defined early routes. Computational chemistry and machine learning teams are beginning to explore the subtler electronic and conformational effects of this compound, looking to design new derivatives that target everything from cancer enzymes to specialty polymers. My own reading shows more universities and small biotech companies exploring libraries of trifluoroacetamide-based molecules, hoping to unlock new therapeutic opportunities. Life science and agrochemical researchers also hope new analogs with modified activity can minimize environmental persistence (a growing concern for fluorinated organics). These efforts depend on a cross-pollination of synthesis know-how and rigorous testing, so people working in traditional chemistry and computational modeling both play crucial roles.
Toxicity: Knowing the Risk and Seeking Balance
The toxicity profile of N-Methyl-2,2,2-Trifluoroacetamide refuses to be ignored, largely because organofluorine compounds have caught the attention of environmental chemists and regulators. Though acute toxicity in standard lab use seems moderate, long-term environmental fate studies push users to rethink old habits. Chronic exposure, either through skin or the environment, draws concern—organofluorines breakdown slowly and sometimes bioaccumulate. Current research digs into both the direct biological impact and the way this molecule and its breakdown products interact with air and water. Some studies note enzyme inhibition, others flag irritation or even dubious links to developmental toxicity at high exposures. These insights drive industry and academia to explore safer substitutions, better containment, and comprehensive waste treatment, instead of just focusing on quick short-term handling.
Glimpses Ahead: Future Prospects
Staring at the future for N-Methyl-2,2,2-Trifluoroacetamide, I see more than just another specialty reagent. With society keeping a sharper eye on fluorinated chemicals and their impact, research teams and regulatory groups are pressing for less persistent, safer analogs and greener synthesis. Demand pops up wherever fluoroamides offer advantages in selectivity, durability, or biological let-through, but that comes with the challenge of reducing legacy risks. New catalysts, tailored degradation pathways, and improved recycling join with open-source chemical safety standards, all hoping to keep the door open to these valuable tools without repeating the mistakes of the past. Researchers—especially those crossing boundaries from chemistry to biology—are steering discovery both toward improved medicines and a lighter environmental footprint. The journey of N-Methyl-2,2,2-Trifluoroacetamide reflects how chemistry adapts, trading immediate performance for long-term responsibility, and demanding new generations of scientists who appreciate both sides of the equation.
Most people never hear about N-Methyl-2,2,2-Trifluoroacetamide unless they spend plenty of late nights in a lab. This colorless liquid rarely gets a spotlight, but folks working in analytical chemistry or organic synthesis know its value. Instead of talking through jargon, let’s break down what it does, the issues that come with it, and why regular people outside the chemistry bubble should pay attention.
Why Chemists Rely on It
Those bottles marked with a daunting name like N-Methyl-2,2,2-Trifluoroacetamide mostly live on shelves in research and pharmaceutical labs. Chemists lean on this liquid when they're prepping samples for gas chromatography (GC) or mass spectrometry (MS). These machines give answers on what’s in a mixture, but some compounds naturally stick to themselves or break apart too easily. Picture trying to analyze a sticky caramel and having your equipment gummed up. Here, N-Methyl-2,2,2-Trifluoroacetamide steps in, turning those tricky molecules into forms that slide through detection systems without fuss. This process, called derivatization, helps boost accuracy and reproducibility.
Everyday Impact Hides Beneath the Surface
Once in a while, what happens in the lab hits the kitchen table. Consider pharmaceuticals where tiny impurities, left unnoticed through sloppy sample prep, can matter when making drugs. We’re not always talking huge life-or-death stakes, but sometimes health depends on rock-solid analysis, and small mistakes can break the trust we put in our medicines. The humble vial of N-Methyl-2,2,2-Trifluoroacetamide greases the gears of a system that checks for quality and safety before tablets reach a pharmacy shelf.
Weighing Benefits Against Hazards
I’ve spent my share of days handling reagents that demand respect. N-Methyl-2,2,2-Trifluoroacetamide, like many chemicals, earns a spot on the “handle with care” list. Spills can irritate the skin or eyes, and fumes shouldn’t float around anywhere people like to work. It highlights an ongoing debate in labs about safety culture and whether training matches the real risks. Regulations lay down the basic rules, but regular reminders, protective gear, and shared stories about close calls go a lot further. Labs that don’t keep safety front and center risk more than just ruined samples—they put people at risk, too.
Looking for Greener Choices
Honest conversations inside the scientific community focus on finding less hazardous alternatives. The need for solvents and reagents that give clear results without piling up environmental debt keeps coming up. It’s not about fearmongering—plenty of tools exist to make labs safer and more sustainable, from better ventilation to improved waste disposal. Many researchers argue that good science must go hand-in-hand with reduced hazards. Companies and universities that support greener chemistry often attract people who want to solve real-world problems instead of adding to them.
Wrapping Up the Unseen Influence
N-Methyl-2,2,2-Trifluoroacetamide might not trend on social media, but its role as a behind-the-scenes helper in analytical science deserves notice. Decisions about chemicals in labs echo far beyond the bench. Keeping that in mind whenever we talk about lab safety, pharmaceutical quality, or environmental sustainability helps keep the science trustworthy and the people who rely on it protected.
Chemistry Is Closer Than Most Think
Walk into any discussion about the chemistry behind research labs, pharmaceuticals, or even flavor science, and a long-winded name can appear out of nowhere. Take N-Methyl-2,2,2-Trifluoroacetamide. The name’s a mouthful, and for anyone not knee-deep in chemical catalogues, it sounds like pure jargon. Truth is, each piece of that name paints a clear portrait of what the molecule looks like and how it behaves. The chemical formula mirrors the name: C3H4F3NO.
Why Chemical Formulas Matter to Everyone
There’s a practical side to knowing formulas. College chemistry forced me to memorize endless strings of numbers and letters that barely meant anything at the time. Only after working with tangible substances—measuring out grams, reacting powders with liquids, waiting for color changes—did those formulas start making sense. The formula C3H4F3NO isn’t just a random code. It tells researchers exactly how many atoms go into each molecule. That precision brings safety, predictability, and logic to chemistry. Without formulas, even basic steps in manufacturing or research would become wild guessing games.
Breaking Down the Parts: Atoms With a Job to Do
This chemical involves three carbon atoms, four hydrogens, three fluorines, one nitrogen, and a single oxygen. That extra “trifluoro” bit means three fluorine atoms cluster on the same carbon. Methyl groups signal a single carbon came along with three hydrogens—melded into the molecular framework. The design isn’t accidental. Each atom changes how the molecule reacts, how it dissolves, and how it protects neighboring structures from breaking down or oxidizing.
Why Fluorine’s Presence Isn’t Just for Show
Adding fluorine sends a powerful message in chemistry. With trifluoroacetamide, those fluorines help block reactions that might tempt other molecules. That level of control got researchers excited back in the day; fluorinated amides create reliable, predictable outcomes in chemical synthesis. The hardiness of these molecules pays off in many specialties, including developing pharmaceuticals that need to resist metabolism and environmental chemistry where breakdown must be prevented for a certain length of time.
Bringing Trust Back to Chemistry
The chemical world has seen its fair share of public mistrust. Some worry about the downstream effect of strange-sounding ingredients, others get lost in lists that read like another language. Personally, honest conversations around what these compounds do—and what their formulas mean—start to mend that trust. Publishing data, sharing peer-reviewed research, and maintaining transparency about sources help level the playing field. Decades ago, knowing the formula C3H4F3NO could have been reserved for a small circle of scientists; now, genuine data is easy to access online, complete with safety records, research papers, and thoughtful breakdowns.
Problem-Solving Attitudes
Simple facts like chemical formulas go a long way in bridging gaps. Sometimes a small, specialty molecule opens the door to a new medicine, a less polluting solvent, or a reliable process in agrochemicals. If something about a compound sparks controversy or confusion, let’s not gatekeep the science. Delivering clear, direct answers, and encouraging ongoing education — whether through schools, science journalism, or open lab days — keeps the torch of knowledge burning. The formula C3H4F3NO proves there’s lasting value in pulling back the curtain, one molecule at a time.
Why Chemical Safety Can’t Be an Afterthought
Working in a lab dealing with small molecules every day, I’ve learned that N-Methyl-2,2,2-Trifluoroacetamide demands respect. It might look just like another bottle on the shelf, but cut corners with handling or storage and you’ll quickly see the downside. Anyone who’s ever sprayed a reactive solvent on their arm or watched a colleague clear out the lab because of chemical fumes understands that safety steps aren’t pointless bureaucracy. They’re real barriers that stop you from getting hurt.
Protecting Your Skin and Lungs
This compound can irritate the skin, eyes, and respiratory system, so skipping gloves, goggles, or a lab coat is asking for trouble. I once saw someone ignore eye protection on a busy synthesis day—after a quick splash, no one needed convincing about goggles ever again. Latex or nitrile gloves keep hands safe, while a properly fitted pair of safety glasses will guard against surprise splashes or spills. Fume hoods are a must for weighing or transferring N-Methyl-2,2,2-Trifluoroacetamide. Open bottles invite inhalation, and it doesn’t take much for throat irritation or worse to follow.
Clear Workspaces Stop Accidents Before They Start
Clutter on the bench and open containers are enemy number one. In my experience, a crowded workspace equals accidental mixing and hard-to-clean spills. Good habits—like labeling every container, immediately wiping up drips, and never leaving the bottle uncapped—will help you dodge plenty of headaches. An incident in our lab, where chemicals got mixed up due to lazy labeling, made everyone rethink their routines.
Flight Isn’t the Plan: Don’t Wing Emergency Readiness
Even the most careful scientist will one day run into a chemical mishap. You want the eyewash and safety shower ready, not rusted shut or blocked by cartons. Safety drills may make some workers groan, but I’ve been in a lab where one saved eyes and skin after a splash. Know exactly where the nearest first aid and safety stations are, and run through cleanup and reporting steps with everyone in the lab regularly.
Sensible Storage and Disposal: Don’t Build a Time Bomb
Heat, sunlight, and reactive chemicals can break down many lab reagents, and N-Methyl-2,2,2-Trifluoroacetamide is no exception. Storing it away from anything acidic or oxidizing reduces the risk of dangerous byproducts. Flammable cabinets help contain these types of chemicals, even if you trust your luck more than most.
Proper disposal speaks volumes about professionalism and environmental awareness. Pouring leftovers down the drain is irresponsible and opens up fines or even worse, shutdowns. Collect used solvent and contaminated materials in marked containers and turn them over to your waste contractor. If your department or building hasn’t reviewed proper disposal in a while, request a refresher.
Leadership Means Modeling the Right Behavior
In the end, a lab’s safety culture starts with its most senior members. I make it a point to gear up and do things by the book, even with small volumes or on slow days. People follow what they see. Cutting corners with N-Methyl-2,2,2-Trifluoroacetamide or any lab chemical only teaches newcomers to trust luck over skill and wisdom.
Chemical safety steps might seem fussy at first, but they turn into muscle memory. In every team, someone’s always watching your habits. Set the right example, and you’ll avoid the close calls altogether.
Respecting Chemistry to Protect Health and Research
I spent years working in the lab, surrounded by shelves of bottles marked with formulas and warnings. N-Methyl-2,2,2-Trifluoroacetamide might not be a household name, but anyone who’s handled specialty chemicals knows: small mistakes around chemicals can pile up, and sooner or later, someone gets hurt or expensive research gets set back. So, proper storage isn’t overkill, it’s common sense.
Avoiding Costly Mix-Ups and Accidents
This compound brings unique hazards to the table. With its volatility and potential to irritate the skin or respiratory system, it commands the kind of respect you'd show a live wire in the wall. Tossing a chemical like this into a crowded storage area with generic bottles and mystery jar labels just asks for trouble. A moment of carelessness—grabbing the wrong bottle, ignoring a faded label, uncapped containers—has ruined experiments and sent people for medical checks. In my experience, creating a “chemistry culture” in the lab meant putting obsessive attention on this kind of detail.
Staying Cool, Staying Safe
Most specialty amides, especially those with fluoro groups, don’t fare well in heat. Fluctuations creep into storage areas—think faulty HVAC systems or overstuffed fridges. This sounds mundane, but I’ve seen bottles swell or degrade because someone put caustic or reactive materials nearby. Keep N-Methyl-2,2,2-Trifluoroacetamide in a cool, dry spot. Dedicated refrigerators for chemicals (never household food fridges) make sense for this reason. Reliable temperature logs help track conditions, and backup plans for power outages spare headaches during storms or equipment failures.
Preventing Unwanted Reactions
Storing away from acids, bases, oxidizers, and moisture—this rule turns up in every safety manual, and with good reason. I’ve watched even experienced researchers forget that a single spill or whiff of moisture in the bottle risks contamination or runaway reactions. Color-coded storage, physical shelf barriers, and clear labeling go far in keeping incompatible chemicals apart. This isn’t bureaucracy; it’s how you end the day with all your fingers and your data intact.
Securing the Lab and Our Environment
Theft or unauthorized use might sound far-fetched for a niche chemical like N-Methyl-2,2,2-Trifluoroacetamide, yet the potential for misuse or accidental spillage always exists. Locking cabinets and limited access save everyone from much nastier outcomes. I remember incidents across university labs where unauthorized handling led to spills that cost thousands to clean up and resulted in environmental reports. Proper secondary containment—trays to catch leaks, dedicated chemical cabinets—can prevent a spill from becoming a full-blown cleanup operation.
Building Habits That Stick
Everyone in the lab, from grad students to seasoned researchers, carries the responsibility for getting storage right. Frequent training, posted reminders, and peer review of chemical storage habits foster a safer working environment. In my labs, we survived budget cuts and shifting staff because we made chemical stewardship a living part of the day. Complacency stays away if every researcher treats each bottle of N-Methyl-2,2,2-Trifluoroacetamide like it's waiting for a chance to make trouble.
Better Storage Means Better Science
Reliable storage practices go beyond safety—they uphold the integrity of our work. Safe chemicals, accurate results, fewer health scares, and less drama for the environment. By respecting the quirks of chemicals like N-Methyl-2,2,2-Trifluoroacetamide, we keep our science sharp and our labs running smoothly. I’ve seen the difference it makes, and I wouldn’t trade that peace of mind for anything.
Walk into any modern chemistry lab, and odds are you’ll find someone using N-Methyl-2,2,2-Trifluoroacetamide, often just called MSTFA. Even though the name sounds like something from a science fiction novel, this clear liquid does a lot of heavy lifting in both research and industry. For anyone who’s ever spent time doing chemical analysis, MSTFA brings a real-world solution to common problems in sample prep and chemical modification.
Bringing Clarity to Chemical Analysis
For researchers who work with gas chromatography and mass spectrometry, MSTFA has built a solid reputation. Many compounds in biology and environmental science—like amino acids, sugars, or steroids—don’t like to evaporate easily. Let’s say you want to look for these in blood or soil; you hit a wall, because they just won’t make the jump from liquid to gas when the time comes to analyze them. MSTFA steps in to help by transforming stubborn molecules into lighter, more volatile forms. This process, called derivatization, means suddenly you’re picking up the right signals in your instrumentation and seeing the whole picture rather than just blurry outlines.
I remember a long project where we tried to measure trace hormones in complex samples. Without a reagent like MSTFA, we’d miss details that pointed to biological changes or chemical shifts. MSTFA didn’t just boost our signal; it meant the difference between drawing real conclusions or staring at useless data.
Taming Tough Samples in Drug Discovery
Pharmaceutical labs spend years testing small tweaks in new compounds to find safer, more effective medicines. MSTFA earns its keep here since it can make a wide range of chemical groups more visible and measurable. If a molecule sticks itself full of hydrogen bonds, it clings to everything in its path, leading to smears on readouts. MSTFA breaks up these sticky groups, like pesky hydroxyls or carboxyls, and turns them into manageable forms. As a result, chemists can get reliable numbers and sharper snapshots of what’s really going on in their samples.
Some of the most important steps in early drug research wouldn’t run nearly as smoothly without reagents like MSTFA. It helps labs move faster from the guesswork phase to building real knowledge about candidates that might one day treat disease.
Environmental Science and Food Testing
It’s not just about fancy drug molecules or academic research projects. MSTFA gets called into action checking pesticide levels, pollutants, or food additives. Any time analysts need to look for very low concentrations of tricky-to-detect compounds, MSTFA acts as the behind-the-scenes fixer.
Food safety labs have to cover a huge territory of chemical tests, especially when processing everything from grains to fruits. A sugar by itself can clog up chromatography columns or evade clear measurement, but MSTFA turns it into a compound that plays nicely with analytical methods. Regulators and consumers both count on these results to protect public health, and MSTFA quietly holds up its end of the bargain.
Looking Forward
Researchers want fewer steps, faster results, and rock-solid answers. MSTFA is already known for its versatility, but some are searching for even safer, greener alternatives. There’s a growing push toward reducing waste and risk, especially in places where labs run hundreds of samples daily. Some innovations explore less toxic reagents or automated systems that can handle MSTFA safely with less exposure to chemists.
For now, MSTFA stays on the frontlines where analytical chemistry needs reliability and flexibility. As science moves forward, it helps bridge the gap between complex raw samples and results that matter.