2,2,2-Trifluoroethyl Formate: Exploring an Unassuming Chemical with Far-Reaching Impact
Historical Development
Chemistry often rewards those with keen curiosity, and the discovery and development of 2,2,2-trifluoroethyl formate is no exception. This compound drew attention from researchers interested in fine chemicals and pharmaceutical intermediates. The compound traces its roots to mid-20th-century efforts to introduce fluorine into organic molecules, part of a broader movement in organic synthesis that recognized the power of fluorinated groups to steer reactivity and stability. Chemists who navigated the wave of fluorinated compound research in the 1970s and 1980s viewed fluoroalkyl formates as specialty building blocks. That era’s hunger for improved agrochemicals, new pharmaceuticals, and materials with unique traits set the stage for diligent exploration of trifluoroethyl derivatives, with this formate fitting neatly into the toolbox of synthetic chemists. Echoing the way synthetic aspirin or nylon emerged from persistent tinkering, trifluoroethyl formate came about from pragmatic work driven by industrial, medicinal, and academic curiosity.
Product Overview
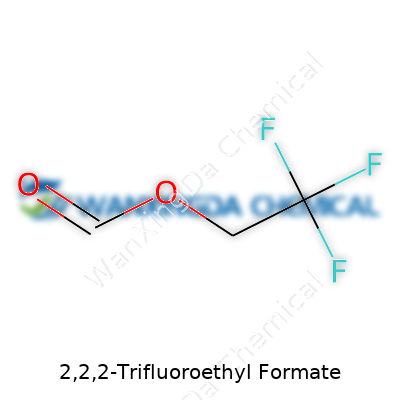
2,2,2-Trifluoroethyl formate stands out as an organic ester with a structure that mixes a formyl group with a trifluoroethyl backbone. Its molecular formula, C3H3F3O2, might look simple, but the presence of three fluorine atoms makes a big difference compared with related non-fluorinated esters. Fluorination isn’t just for novelty—the trifluoroethyl group resists metabolic breakdown, adds lipophilicity, and alters how the molecule behaves in synthesis and applications. Folks in the lab recognize trifluoroethyl formate as a useful piece in making API intermediates, a mild acylating agent, or even a flavor modifier, especially because it interacts with other molecules in ways that non-fluorinated pieces simply don’t.
Physical and Chemical Properties
As a liquid at room temperature, 2,2,2-trifluoroethyl formate has a modest boiling point and a distinctive, often sharp odor reminiscent of other small esters. Its density runs higher than many hydrocarbon-based organics, reflecting the heavy fluorine content. The molecule doesn’t dissolve easily in water; it leans toward mixing with organic solvents, a trait shared by many trifluoroethyl derivatives. Chemical stability plays in its favor—thanks to the electron-withdrawing punch of those fluorines, the formate resists easy hydrolysis and oxidation compared to its hydrogen-carrying cousin, ethyl formate. For a chemist, the trifluoroethyl group tunes the molecule’s reactivity, dampening unwanted side reactions and empowering more selective transformations.
Technical Specifications & Labeling
In the lab or plant, clear labeling of 2,2,2-trifluoroethyl formate goes a long way. The material usually carries a CAS number, along with hazard pictograms to cover its slight volatility and mild skin or eye irritation risk. Specifications for purity, moisture content, and appearance help keep quality on point and contamination low. Typical analytical data, drawn from methods like gas chromatography or NMR, confirm its identity for those with strict process requirements. Trace metals, residual acids, and other process byproducts pull attention too, as these can sabotage delicate reactions if left unchecked. In my own work, a tidy, well-labeled bottle of this compound always meant fewer headaches downstream, whether in bench chemistry or scale-up.
Preparation Method
Synthesis of 2,2,2-trifluoroethyl formate tends to follow the same logic as other simple esters, but with a twist thanks to the trifluoroethyl group. A common route stirs together trifluoroethanol with formic acid or its derivative, sometimes under acidic catalysis to coax the reaction along. Some shops prefer a dehydrating agent or invest in acid chlorides, swapping hydrogen chloride for water to speed things up and improve yields. The overall method isn’t groundbreaking, but careful drying and exclusion of water matter more due to the high reactivity of the starting materials and the cost of trifluoroethanol. What stands out is the need for glassware and equipment that can shrug off acid—fluorochemicals eat cheaper lab gear alive. Anyone with past experience knows the misery of a reaction gone sour from moisture or corroded valves.
Chemical Reactions and Modifications
Chemists who work with trifluoroethyl formate like its predictable behavior. Under gentle heating or base, it yields up the formyl group, making it a reliable acylation agent. Nucleophiles home in on the carbonyl carbon, ready to attack and displace the trifluoroethyl moiety in planned syntheses. The fluorines help slow down unwanted side plays, steering the reaction more cleanly than ethyl formate would. Folks searching for new fluorinated compounds find it a flexible jumping-off point; with modification, it shapes into unusual amides, ureas, or trifluoroethyl-substituted building blocks for more advanced targets. This flexibility gives medicinal and materials chemists leeway to design molecules that would otherwise be tough to access.
Synonyms and Product Names
Along the supply chain, this compound shows up under a handful of aliases. Besides "2,2,2-trifluoroethyl formate," I’ve seen shipment crates marked "trifluoroethanol formic acid ester" and "formic acid 2,2,2-trifluoroethyl ester." International catalogs sometimes offer translations or abbreviations—despite that, the substance inside the bottle stays the same. Naming confusion tends to irritate chemists more than help, so keeping an eye on structure and CAS data sidesteps shipping mistakes and lab mix-ups.
Safety and Operational Standards
Safety in the world of fluorinated compounds takes on everyday importance, and trifluoroethyl formate is no exception. Its volatility brings exposure risk through vapors, so proper fume hoods and gloves form the first line of defense. Inhalation or direct contact can spark irritation or central nervous system effects, so no one in the lab ignores fresh air and quick cleanup. Adequate labeling not only avoids accidental exposure but also lines up with regulatory standards around handling and disposal of halogenated organics. Training sessions and vivid signage do more to keep workers safe than any binder of outdated MSDS sheets. Keeping air monitors and spill kits close has saved more than one new chemist from a rough first day.
Application Area
Trifluoroethyl formate brings value across research, pharmaceuticals, and materials science. The pharmaceutical sector calls on this ester as a masked formyl group, allowing complex intermediates to weather tough conditions without breaking down. Process chemists appreciate its use in introducing the trifluoroethyl group, a favorite trick to boost bioavailability and stabilize molecules against metabolic destruction in the liver. In flavor or fine chemicals, the ester’s volatility and crisp scent lend well to formulating new compounds that edge away from the routine. Materials research leans in here too: custom surfactants, advanced polymers, and specialized surface treatments all trace their traits back to starting points like trifluoroethyl formate.
Research and Development
Investing in R&D around 2,2,2-trifluoroethyl formate rests on more than just curiosity—it emerges from a chase for new molecular scaffolds and a demand for sustainable processes. Research groups continue to spin off new reactions using this ester, uncovering greener catalysts, milder conditions, and more resource-efficient syntheses. Environmental focus urges scientists to work up routes that cut waste and avoid toxic byproducts, so every published improvement on trifluoroethyl formate’s preparation has practical value and commercial appeal. In my experience, teams that stay nimble—adapting methods to new findings—see the fastest gains both in yield and in process safety.
Toxicity Research
Concerns about toxicity run deep for any organofluorine compound, and the science community looks hard at 2,2,2-trifluoroethyl formate’s risks. Early studies flagged potential for irritation and the long-term impacts of trifluoroethyl-containing byproducts if released unchecked. Ongoing toxicology screens dig into breakdown rates and persistence, especially since some organofluorines stay in soil and water for years. While data on trifluoroethyl formate suggest moderate risk under routine conditions, strict control on air emissions and wastewater discharge remains non-negotiable. The push towards safer alternatives and smarter containment doesn’t slow down—every regulatory cycle turns up the pressure, demanding stronger data and leaner environmental footprints.
Future Prospects
Looking ahead, prospects for 2,2,2-trifluoroethyl formate hinge on innovation in synthesis and sustainable practices. Accelerating demand for smarter drugs and advanced materials keeps chemists busy rethinking how to use this flexible ester. There’s optimism in coupling this compound to new green processes, especially as global producers aim to shrink costs and trim environmental liabilities. Modular manufacturing, real-time monitoring, and circular chemistry hint at a future where trifluoroethyl formate finds wider and safer uses. My experience suggests that the real leap won’t come from a single breakthrough, but from persistent, step-by-step improvements—lowering losses, refining logistics, and designing reactions that make every fluorine atom count.
Inside the World of Laboratory Chemistry
In all my time working with compounds in a small organic chemistry lab, there’s always been something fascinating about tracing the structure of a molecule back to its formula. Anybody with years of pipetting and mixing behind them knows that even a slight swap of atoms—sometimes a single fluorine—can completely change how a compound behaves. This is the case with 2,2,2-Trifluoroethyl Formate. Its name might not roll off the tongue, but every part tells a story about the molecule it’s built from.
The Formula: Getting Specific
2,2,2-Trifluoroethyl Formate carries the chemical formula C3H3F3O2. That string of numbers and letters looks simple on the surface, but it unlocks a lot for chemists. The “trifluoroethyl” bit signals three fluorine atoms have replaced hydrogens in the ethyl group, which does more than just bump up the molecular weight. Fluorine’s presence impacts boiling points, solubility, and, crucially, how the molecule reacts with others. Working with these compounds, it’s easy to notice they handle differently than their non-fluorinated cousins. Sometimes that’s helpful, sometimes it gets in the way.
What often gets missed outside of chemistry circles is how much effort goes into pinpointing these formulas. In the lab, mislabeling or mistaking a single atom can send an experiment down the drain or spark a safety issue. Every molecule documented in journals and databases—especially ones like 2,2,2-trifluoroethyl formate—builds on research done over decades. The process of confirming the formula means running it through NMR, IR, and mass spectrometry, things no one gets to skip if they want reliable results. That effort up front saves a lot of wasted time and keeps chemistry publications trustworthy, an expectation in research communities that value accuracy over speed or cost-cutting.
The Broader Picture: Why Surface Detail Matters
Even outside pure science, this bit of chemistry matters. Fluorinated molecules keep popping up in pharmaceuticals, agrochemicals, and fine chemical manufacturing. The trifluoromethyl group in this formate brings certain biological activity or durability not always seen elsewhere. A drug’s safety or an agrochemical’s environmental breakdown sometimes comes down to a few extra fluorines. Knowing the formula helps regulators and developers anticipate how a compound might linger in soil or water, or how bodies process it after a dose or exposure. That matters for communities near production sites and for future regulations as scientists learn more about persistent pollutants.
Challenging the Assumptions in Chemistry
It’s easy to assume most molecules fit quietly into textbooks, their formulas only important for exams. Working with volatile or reactive chemicals, the formula becomes far more than classroom trivia. Even small labs feel the ripple effect of accuracy in formula reporting. A single line in a peer-reviewed paper could guide a student’s thesis or shape a regulatory debate. Inaccurate reporting doesn’t just slow research—it risks lab safety and public trust.
This is one of chemistry’s most persistent lessons: getting the details right, down to every last atom, isn’t busywork or nitpicking. C3H3F3O2 stands for more than numbers and letters; it represents a shared language in science, one that keeps researchers aligned and communities safer. That kind of clarity is non-negotiable in a world shaped by synthetic molecules.
Hidden Muscle in Modern Chemistry
Walk through any active lab working on drug development, and you'll see a whole shelf of bottles with names only an organic chemist could love. One of those odd-sounding chemicals, 2,2,2-Trifluoroethyl Formate, rarely grabs headlines but fits right into the kind of modern chemistry that shapes real-world breakthroughs. The molecule has a lot going for it: a volatile nature, strong electron-withdrawing power from those three fluorines, and a formate group that opens doors for reactions. None of that means much unless you've seen how this stuff fits into an experiment that pushes something useful out the other end.
Key Player in Pharmaceutical Development
Drug research often looks for shortcuts, safer steps, and new paths around old problems. This is where 2,2,2-Trifluoroethyl Formate brings value. It's a building block chemists turn to when they need to slip fluorine atoms into a carbon chain. That matters because adding fluorine changes how drugs behave in the human body, helping them last longer or sharpen their effects. Over the years, researchers noticed that many drugs with at least one fluorine atom wind up making it further in clinical trials. No magic, just clever chemistry, and molecules like this make it possible.
You don’t need a Ph.D. to realize that the road to a new pill in your medicine cabinet gets a lot shorter when tools like this molecule exist. The quest for new antivirals, cancer therapies, even next-generation anti-inflammatories, all dig into chemical spaces unlocked by smart use of fluoro-organic building blocks. Without them, a lot of molecules never stand a fighting chance inside the body, and start-ups or big pharma both see the value here.
Emerging Uses in Advanced Materials
It’s not all about medicine. Some of the new plastics, coatings, or electrolytes for batteries borrow technology originally designed for the pharma world. 2,2,2-Trifluoroethyl Formate slides into polymer synthesis, helping scientists fine-tune surfaces that shrug off water or resist wear. In industries where corrosion eats profits—think aerospace, biotech, microelectronics—chemists need molecules that stand up to everything from acids to high heat. With that trifluoroethyl group, this compound unlocks ways to build chemical links that don’t quit so easily.
Battery researchers, especially those chasing safer, longer-lasting lithium-ion cells, look hard at fluorinated intermediates. They help design new electrolyte additives, coatings for metal parts, or even flame-retardant components. While the details stay buried in patents, the use of building blocks like 2,2,2-Trifluoroethyl Formate keeps showing up in research papers.
Pursuing Safer and More Sustainable Practices
Every tool in a modern chemist’s kit opens up questions about health, safety, and the planet’s future. Strong fluorinated compounds carry a reputation for sticking around, so attention turns to how cleanly these molecules get used and disposed of. Any responsible use needs a plan for waste, closed systems that keep fumes in check, and tight paperwork. Workers who spend their days synthesizing new materials know safety is non-negotiable—not just to stay in compliance but because good labs run on people who feel valued and safe.
Building Real Solutions, Not Just Theories
If the future depends on designing new molecules with sharper performance and safer side effects, the supporting cast matters as much as the star players. 2,2,2-Trifluoroethyl Formate stays out of the public eye but shapes research that touches medicine, energy, and technology. With more attention on greener processes and thoughtful stewardship, its benefits can keep pushing chemistry in directions that really make a difference. Facts and experience both say that the main applications worth fighting for are always the ones with real-world rewards.
Why Chemists Pay Attention to Boiling Points
Walking into a laboratory, it doesn’t take long to realize how much everything depends on boiling points. They shape decisions on safe handling, storage, and even the choice of glassware. A compound like 2,2,2-Trifluoroethyl Formate sticks out to me, since its trifluoromethyl group dramatically alters everyday chemistry compared to simpler esters.
2,2,2-Trifluoroethyl Formate carries a boiling point of about 55–57°C. That’s more than 50 degrees cooler than water. Set a bottle of this on a warm bench and you’d smell its sharp, fruity scent within minutes. Nothing in a busy research space puts safety more front and center than a low boiling point. Vapor pressure rises with temperature—double the concern if that vapor is a reactive or toxic one.
What the Boiling Point Tells Us
Taking the boiling point of 2,2,2-Trifluoroethyl Formate at face value sounds pretty straightforward. On a technical level, it means that the liquid turns to vapor in open air below average boiling water temperature. Pouring it into an open dish, it drifts away almost before the surface tension breaks. That has practical impacts—flasks need stoppers, and fume hoods stop being optional.
Many commercial esters don’t share this volatility. The trio of fluorine atoms pulls electron density, lowering intermolecular forces compared to non-fluorinated cousins. You see more rapid evaporation, easier distillation, and—if you aren’t watching—higher exposure risks. For those running syntheses, separating 2,2,2-Trifluoroethyl Formate from a reaction mix works at lower temperatures, so thermal decomposition doesn’t threaten your product nearly as much.
The Human Story Behind Numbers
There’s a practical side often missing from reference charts. Working with substances like 2,2,2-Trifluoroethyl Formate was a crash course for me in paying attention to little details. During grad school, underestimating low boiling esters led to lost product on a rotovap and a cloud of vapor that sent folks scrambling. You remember lessons like that.
Laboratories everywhere post Material Safety Data Sheets for a reason. A boiling point tells you how quickly things go from liquid to gas. Higher volatility can mean greater risk for inhalation or spills. A proper ventilation system and sealed containers don’t just save money by cutting down loss; they guard against headaches, coughs, and even serious exposures.
Better Handling for Better Outcomes
Solutions for dealing with low-boiling chemicals don’t need rocket science. Store them in tightly sealed bottles, away from heat. Always work in a fume hood. Label flasks clearly—nothing’s more humbling than discovering a mystery liquid was a missed volatile. Rotovaps, cold traps, and ice baths aren’t just recommended—they’re essential for anyone trying to capture every last drop.
At the end of the day, facts like a boiling point of 55–57°C aren’t just trivia for manuals. They’re the first thing a good chemist thinks about when planning an experiment. Respecting the physical properties of solvents and reagents makes research safer and more successful, and it helps everyone breathe a little easier.
Why Chemists and Students Keep Asking About 2,2,2-Trifluoroethyl Formate
People in labs keep bumping into compounds like 2,2,2-Trifluoroethyl formate. Ask most chemistry students, and they’ll recall that nervous moment before a reaction—Googling everything they touch, even if their professor shrugs it off as just another simple ester. But once the word “trifluoro” shows up, folks perk up. Fluorine chemistry usually means you pause and double-check. The concern isn’t overblown. It comes from a mix of hazy safety data and stories about fluoro-organics going wrong.
Looking at What’s Known About Its Hazards
Search through material safety databases, and you’ll find 2,2,2-Trifluoroethyl formate hiding among dozens of other specialty esters. Its structure is simple—a formate connected to a fluoroalkyl group. There’s no wide industrial use, so in many public references, details on toxicity just trail off. This has led some to treat it like any other volatile organic compound—unwise. Many organic formates have irritating vapors. Adding a trifluoromethyl group, as we see here, means inhalation risk and possible acute toxicity go up.
Most sources lump it with esters that can irritate the eyes, nose, and throat. If you spill it on skin, burning or redness can follow. It’s flammable—across academic labs, summer heat and careless handling have triggered fires with similar chemicals, so that’s not a hypothetical threat.
There’s another side to fluorine chemistry, too. Trifluorinated organics, if mishandled or degraded, may yield breakdown products that are hazardous. In rare lab accidents, fluorinated acids and fluorinated formates can release small molecules such as HF, which is highly toxic. Cautious chemists respect that, even if most work never brings it to the point of decomposition.
Why Proper Handling Makes All the Difference
Safety habits around any volatile ester matter. No one wants to end up as the story others tell as a warning. Once I accidentally splashed a simple methyl formate—and spent the afternoon feeling dizzy from the fumes. If that had been a fluorinated compound, symptoms could’ve been worse.
Gloves, goggles, and good ventilation matter—even more if you’re dealing with something exotic like trifluoroethyl formate. Lab safety audits show that injuries often come from underestimating small-volume or “boring” chemicals. We all get cocky, especially in university settings, thinking the real danger only lurks in bright yellow or marked bottles.
It’s tempting to skip reading the safety sheet, but that’s where problems start. Too many researchers and students believe that if a compound isn’t flagged with a skull-and-crossbones, it’s fine. I’ve seen people learn the hard way—unexpected rashes, headaches, or ruined clothing because they gambled on easy handling.
How Science Gaps Create Blind Spots
The biggest issue with compounds like 2,2,2-Trifluoroethyl formate is the lack of detailed toxicological profiles. Regulators focus attention elsewhere—on big-volume chemicals, pharmaceuticals, or known disaster cases. That leaves a strange limbo: not enough data, so nobody draws strong lines.
Without precise knowledge, the only reasonable choice is to handle them with extra care. Even outside a professional lab, mixing up chemicals purchased online or for DIY projects without clear hazard information makes accidents more likely. It’s best to treat every little-known reagent as if it could be risky until proven otherwise.
The only real solution is better transparency and shared data. If industry and academic groups publish more on what happens after exposures, everyone’s safer. Until then, we all become the test case—so caution beats bravado, no matter how much we think we know.
Why Care About Handling This Chemical?
Ask anyone who’s spent time in a chemistry lab and they’ll tell you: a careless moment with volatile chemicals can rewrite a day. 2,2,2-Trifluoroethyl formate fits the bill as one of those substances you don’t want to mishandle. This compound pops up in research labs because of its use in synthesis and specialty chemistry. That same appeal—being versatile for reactions—makes good storage and handling practices not just recommended, but essential.
Real Risks of Skipping Precautions
Let’s get practical. If stored in an open or poorly sealed container, fumes can escape. These vapors can irritate eyes, nose, and throat. Anyone who has cracked open a chemical bottle without PPE and felt that familiar sting knows how easy it is to shrug off warnings—until it ruins your next few hours. Too many rely on hoods and fume extractors without checking if their seal really holds up. It only takes a leaky lid to remind you how persistent an odor can be.
Another big point—flammability. Organic solvents and derivatives, like this formate, tend to ignite if they catch a spark or get exposed to high temperatures. It’s tempting to stash bottles on a crowded shelf. That habit sets up a risk that often gets dismissed in daily routines, but even a minor spill could make a small fire travel far too quickly.
Lessons from Experience
Years working next to colleagues handling volatile liquids gave me a front-row seat to why clear protocols matter. A distracted coworker once left a cap slightly loose, thinking the hood would handle everything. By break time, an odd smell worked its way across the benches, distracting everyone. We traced it to that one bottle. We fixed it before someone suffered more than watery eyes, but not before it wrecked trust in routine.
After a chemical spill in another lab across the hallway—triggered by improper shelving and bottle stacking—safety procedures doubled overnight. Bystanders had to evacuate. The shipment involved got quarantined until a full risk review cleared things up. Nobody needed hospitalization, but the lesson stuck: everyone’s safety depends on the next person following the rules.
How to Make Storage and Handling Work in Real Labs
Safe storage starts with a well-fitting cap and a bottle that won’t degrade from the inside out. Glass bottles, with PTFE-lined caps, block both evaporation and corrosion. Avoiding sunlight and heat cuts down on the risk that something will spark spontaneously or pressure will build up inside the container. A cool, dry, and well-ventilated space—preferably in a chemical cabinet designed for flammable liquids—keeps surprises to a minimum.
Access control wraps up the main risks into something manageable. Only people trained for chemical handling unlock those cabinets. In places I’ve worked, storeroom managers kept daily logs, so no bottle changed hands without a note. PPE—nitrile gloves, eye protection, and lab coats—serves as a daily reminder that skin is no match for a chemical splash.
Building a Culture of Responsibility
What stands out over decades in science isn’t just smart equipment or careful labels but a shared understanding that chemical safety reaches beyond personal habits. Open conversations matter. Newcomers who see experienced chemists double-checking caps and inspecting cabinets will pick up those routines. Anyone catching a break in procedure can call it out—no matter what role they fill.
The mindset around 2,2,2-trifluoroethyl formate, and any chemical with similar volatility, boils down to simple respect for risk. It’s not about paranoia—it’s about keeping one mistake from spiraling into a bigger problem, for everyone.