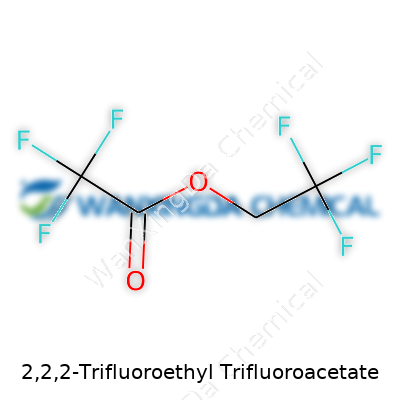
2,2,2-Trifluoroethyl Trifluoroacetate: Chemistry Shaped by Curiosity and Application
Historical Development
The story of 2,2,2-Trifluoroethyl Trifluoroacetate traces back to the changing landscape of organic fluorine chemistry. Fluorinated compounds gained serious interest through the latter half of the twentieth century. Chemists, inspired by the unique properties imparted by fluorine atoms—stability, volatility, and changing reactivity—pushed to craft molecules that could do what others could not. The ongoing search for robust intermediates in pharmaceuticals and material design opened doors for compounds that were once little more than laboratory curiosities. Academic papers through the 1960s and 1970s began to highlight the value of esterifying trifluoroacetic acid with trifluoroethanol—a move that not only produced a versatile reagent, but one that fused the chemical resilience and activity of two distinct trifluoro groups. As research ramped up across global labs, this ester found its niche at the intersection of synthetic methodology and applied innovation.
Product Overview
2,2,2-Trifluoroethyl Trifluoroacetate can look rather unassuming in the bottle, but it packs an interesting punch in the lab. Built from the sturdy backbone of two trifluorinated components, this compound stands out among esters for its reactivity and chemical compatibility. Researchers exploring new chemical routes value its role not just as a reagent, but also as a structural unit that lends heightened electronic effects to target molecules, making it a solid candidate wherever strong electron-withdrawing groups might be needed.
Physical & Chemical Properties
This compound boils off at temperatures far below most conventional esters, a clear sign of the influence fluorine wields: low boiling points, high volatility, and a tendency to evaporate quickly from glassware. Chemically, those six fluorines mean business. The molecule resists simple nucleophilic attack and shrugs off weak acids and bases, making it a reliable player in certain aggressive reaction conditions. Its colorless, often pungent liquid state is typical of volatile esters, demanding careful handling and proper ventilation in practical settings.
Technical Specifications & Labeling
Labels on containers tell the story of how this molecule moves from warehouse to benchtop—hazard pictograms call for respect, signaling the volatility and irritant potential of the liquid. Specifications focus less on consumer-level purity and more on the clean, clear chemical signature observed by NMR, IR, and mass spectrometry, which all confirm its structure. Regulatory standards require clear identification of the substance due to hazardous traits, reinforcing the need for safe storage and strict inventory control.
Preparation Method
Classic synthetic routes build this molecule from the union of trifluoroacetic acid and 2,2,2-trifluoroethanol, most often under the influence of a condensation catalyst or dehydrating agent. This isn’t a kitchen-table process: high-purity starting materials, anhydrous conditions, and attention to temperature prevent unwanted side products and loss through vaporization. Once distilled, the ester emerges as a sharp, economical reagent ready for further transformations, freed from the constraints of lingering water or acid.
Chemical Reactions & Modifications
In the synthetic toolkit, trifluoroethyl trifluoroacetate has carved its own path. Its use as an acylation agent stands out—chemists regularly turn to it to introduce the trifluoroacetyl group onto nucleophilic substrates where regular acetates fall short. The presence of multiple fluorines tilts the electron cloud, making the carbonyl more susceptible to addition without the usual side-reactions that simpler esters trigger. Interest picks up in advanced settings, with trifluoroethyl cleavage and replacement transformations giving researchers a stepping stone into the world of fluorinated drugs, agrochemicals, and specialty materials.
Synonyms & Product Names
Beyond its mouthful of a systematic name, this molecule answers to a few aliases in the literature and in commerce—trifluoroacetic acid 2,2,2-trifluoroethyl ester and TFE-TFA are common shorthand. Each label points to a different professional circle, from organic synthetic chemists to those involved in advanced material supply. Scientific naming isn’t just jargon here—it divides the casual from the expert, as each synonym signals experience in different corners of chemistry.
Safety & Operational Standards
Handling this ester starts with a healthy respect for its volatility and irritant potential. Without proper fume hoods, gloves, and goggles, exposure becomes more than a minor inconvenience—it demands preparedness and unwavering attention. Spilled droplets evaporate quickly, and as with many fluorinated reagents, inhalation or skin contact can provoke sharp reactions. Industrial hygiene standards recommend closed systems and regular air monitoring to keep exposure well within safe limits. Training isn’t a formality here; it’s a necessity that separates safe, productive research from unnecessary hazards.
Application Area
In my time with academic and industrial laboratories, this ester earned a reputation as a ‘fixer’ in tricky synthetic routes. Its strong electron-withdrawing properties allow transformations off-limits to more basic acetic or propanoic esters. Medicinal chemists count on it when tailoring molecules for increased stability or altered biological activity. Process chemists reach for it when aiming for precision in fluorinated building blocks that feed into new-generation pharmaceuticals, pest control compounds, or even advanced polymers that need to resist harsh chemical exposure. The surge in demand for stable, highly fluorinated molecules continues to drive its adoption far beyond the traditional organic laboratory.
Research & Development
New uses for trifluoroethyl trifluoroacetate emerge as the chemical industry evolves. Scientists push the boundaries, integrating the ester into protocols for selective acylation, labeling, and synthesis of complex targets. With the growing intersection of traditional synthetic chemistry and green chemistry practices, modifications to preparation and use focus on reducing waste and improving yield. Advanced reactors cut down on solvent use and energy input, reflecting a broader push toward sustainability. Publications and patents detail how a tweak to this reagent enables more efficient syntheses, or how small changes unlock new routes to molecules previously out of reach—all evidence of a field that continues to innovate.
Toxicity Research
Data on acute and chronic toxicity of heavily fluorinated organics never sits still, given shifting regulations and the relentless quest for safer labs. 2,2,2-Trifluoroethyl Trifluoroacetate’s volatility increases exposure risk, prompting ongoing review of its short- and long-term effects. While the overall toxicity profile remains less severe than some classic alkyl esters, repeated inhalation or skin contact can lead to specific, sometimes severe reactions. Toxicologists track not only the parent molecule, but also decomposition products that result from heating or contact with strong bases, looking for fluorine-rich byproducts that might present additional hazards to workers and the environment.
Future Prospects
Rising demand for highly tailored fluorinated molecules keeps the future for 2,2,2-Trifluoroethyl Trifluoroacetate bright. Advances in medicinal chemistry, where precise molecular tweaks spell the difference between success and failure, drive new appreciation for reactive, stable esters that survive increasingly demanding reaction setups. Shifting industrial focus—toward greener, more efficient chemical production—fuels interest in making this compound safer to handle, easier to recycle, and even more versatile in cross-coupling and acylation roles. The world of chemistry thrives on adaptability, and this ester’s ability to fit into new niches speaks volumes about its ongoing relevance. As new hazards are uncovered and new applications discovered, practical experience and a commitment to continuous learning will ensure its safe and effective use for decades.
A Real-World Glimpse at Its Chemical Nature
The world of chemicals can feel overwhelming. Even a name like 2,2,2-Trifluoroethyl trifluoroacetate gives off a complex vibe. Yet, at the core, this compound boils down to the simple formula: C4H3F6O2. Each character in that formula matters. Four carbon atoms hold the structure together, hydrogen and fluorine take up the edges, and oxygen links it all in an ester. People in the lab, especially folks working in organic chemistry, see these building blocks as familiar friends. That’s because trifluoroacetate esters, like this one, play a big role in synthetic work—often when protection or special reactions are needed. Fluorine’s reputation for stability isn’t for nothing. Its presence can shift how molecules react, stay safe from breakdown, and interact with other chemicals.
Why Do Chemists Turn to Compounds Like This?
Ask any synthetic chemist about their favorite reagents, and sooner or later, something with trifluoro- in the name pops up. The strong pull toward these molecules traces to their ability to resist many types of chemical attack. Long ago in my grad school days, the boss handed me a bottle with “trifluoroethyl trifluoroacetate” scribbled on a fading label. The task seemed simple: use it to protect sensitive parts of a new molecule. The reaction needed something that kept water and other troublemakers at bay. The fluorine atoms in both parts of this molecule made it a perfect guard. Sometimes, lab solutions aren’t fancy equipment or million-dollar contracts. They’re smart use of a stable, stubbornly protective compound.
The Real Impact of Formula Knowledge
Knowing the formula isn’t just trivia. Chemicals with heavy fluorine content can behave very differently from their cousins with hydrogen or chlorine. They stick around in the environment a lot longer. This matters, not only for lab safety but for anyone looking to keep new products—like specialty medicines and crop protection agents—out of trouble down the line. Environmental studies have leaned on facts about these compounds to track them in soil and water. Half-lives aren’t easy to ignore when designing for safety.
Have a spill in the lab? The formula tells responders what gear to wear and which neutralizer to grab. In my own work, the dread of an accidental trifluoroacetate spill forced me to always double-check labels and keep proper documentation. I saw labs get warnings over improper handling because the underlying formula suggested long-lived residues. In one case, researchers traced a persistent problem in a water sample back to a single careless pour-out of a trifluoromethyl compound.
What Comes Next for Trifluoroethyl Trifluoroacetate?
Safer use of chemicals with these formulas needs more than rules on a wall. It calls for building good habits early. I’ve watched new chemists learn fast after seeing what can go wrong. Updated safety training, clear labeling, and simpler disposal options work better than threatening emails from environmental officers. Green chemistry trends don’t ignore fluorinated molecules—they look for ways to design out their risks, or to recycle them safely. By knowing the formula, professionals in science and safety start one step ahead.
Behind the Scenes of Modern Chemistry
Hidden behind impossible-sounding names and long chemical formulas, specialty compounds move science forward in ways most people never think about. I’ve seen a few chemicals in my time that open doors, and 2,2,2-Trifluoroethyl Trifluoroacetate fits that list. Few outside of labs discuss it, but chemists who work with organic materials value it as a powerful intermediate and building block. It’s the kind of ingredient that can take a decent toolbox and add a set of torque wrenches, giving researchers more control.
A Reagent With a Job to Do
This compound stands out because of its two trifluoromethyl groups, making it popular for fluorine chemistry. Anyone who’s spent time developing new pharmaceuticals will have run across the hurdle of stability and bioavailability. Fluorinated groups help molecules resist breakdown, holding up under tough conditions. In the pharmaceutical world, adding trifluoromethyl groups isn't just a random tweak—it can dramatically change how a compound behaves.
Where 2,2,2-Trifluoroethyl Trifluoroacetate steps in is as a reagent. I remember working through syntheses with tricky alcohols and acids. Reagents like this one speed things up, and they help introduce trifluoroethyl groups where needed. Medicinal chemists use it to tweak drug candidates, increasing metabolic stability or trying to bump up receptor selectivity. A seemingly small change—one or two trifluoromethyl groups—can mean a drug doesn’t get chewed up by enzymes in the liver after all.
Down to Industrial Basics
Outside pharma, specialty fluorinated compounds show up in agrochemicals too. The pressure for crop protection pushes companies to reach for more precise, effective options. Compounds prepared using 2,2,2-Trifluoroethyl Trifluoroacetate show up in herbicide and fungicide research because these groups can alter how the final product interacts with plants, soil, and the broader environment. Fluorine chemistry allows designers to manage absorption, persistence, or even limit unwanted side effects.
Materials science takes another slice. High-performance polymers and specialty coatings often benefit from fluorinated building blocks. A coating that shrugs off oil, stains, or extreme temperatures relies on the quirks of these fluorinated chemicals. The acetate serves as a way to bolt fluorine onto the backbone of new polymers, supporting properties that other elements just can’t provide.
Challenges in Using Active Reagents
Working with 2,2,2-Trifluoroethyl Trifluoroacetate isn’t plug-and-play. It’s reactive, and improper handling poses health risks. My early days in the lab, safety came up every time fluorinated reagents arrived. Good fume hoods, gloves, and careful planning matter. Factories have to keep emissions and waste under a sharp eye, since regulatory bodies keep a close watch on fluorochemicals. Reports from the EPA and European agencies show increased oversight for these types of reagents, especially as environmental persistence draws more attention.
Towards Better Alternatives
The science world faces a tough line: demand for new fluorinated molecules keeps rising, but so does the need for responsibility. Smarter green chemistry methods have started to emerge. Some chemists look for ways to recover or recycle fluorinated intermediates, cutting waste. Others try to design molecules that keep the benefits of fluorinated groups, but degrade more easily after use. This is slow progress, but seeing innovation move in that direction brings hope for solutions that don’t trade one risk for another.
Powerful reagents like 2,2,2-Trifluoroethyl Trifluoroacetate keep research humming—helping new therapies, stronger materials, and safer agrochemicals reach the world. The next step comes from staying sharp about both the promise and the price of these hidden helpers.
A Closer Look at Responsible Chemical Management
Most folks who’ve worked in a lab—or spent time in industrial settings—know that anything labeled “trifluoro” tends to be a bit touchy. 2,2,2-Trifluoroethyl trifluoroacetate shows up in research environments, especially where specialty organic synthesis is on the table. Chemical safety doesn’t just belong to industrial engineers or the safety officer; it matters for every pair of hands in the room.
From what I've seen, this compound likes to make an impression. It’s volatile, so it has a habit of showing up in unexpected places if left to its own devices. The label “trifluoro” isn’t just for show. These molecules can be unforgiving when proper storage and practice fall through the cracks.
The Right Place for a Sensitive Substance
If someone has spent weeks troubleshooting unexplained reactions or frustrated by persistent odors in a lab, they know the value of disciplined storage. 2,2,2-Trifluoroethyl trifluoroacetate thrives in cool, dry settings. A dedicated chemical refrigerator works well, not because it’s a luxury, but because ambient temperatures lead to pressure build-up or leaky containers. Air and moisture get in when seals aren’t tight or flasks are carelessly left open, turning stable solutions into unpredictable risks.
Some folks cut corners by stashing reactive stuff with acids, bases, or just about anything non-flammable. That choice—often born from crowded shelves—creates a headache months later. This isn’t just theoretical: incompatible neighbors start silent fights, and when things go wrong, they go wrong fast. Mixing volatile organic compounds and aqueous solutions, for example, can send concentrations airborne or invite unwanted reactions.
Sensible Precautions Speak Louder Than Protocols
If you ask any lab veteran, he or she will say—without parroting official language—that the smallest drop spilled on a bare hand tells the story better than a thousand warning labels. Spills happen, and this chemical’s fumes hit the nose and throat right away. For that reason, gloves made of nitrile or neoprene work best. Latex doesn’t last against most highly fluorinated compounds. Always use goggles, no matter how experienced you think you are. That slight vapor cloud can turn into an hours-long headache, and a hurried cleanup only invites trouble down the line.
Ventilation is key. I’ve been in rooms where the smell lingers much longer than expected because someone skipped the fume hood. Having worked with trifluoro compounds, I never trust open benches or makeshift ventilation—unfiltered air moves these compounds right to where people congregate.
Beyond Storage: Building Good Habits
As technology and chemicals evolve, relying on past experience only works up to a point. Rely on up-to-date Material Safety Data Sheets, not urban myths passed down from senior researchers or older colleagues. Never decant into makeshift bottles or relabel containers with a worn-out pen; faded writing leads to mistakes. Walk through your storage area regularly—expired, contaminated, or compromised containers show up more often than most expect.
Some people act as if “just for a moment” with the wrong solvent or tool is fine. The best labs rarely suffer near-misses because habits get enforced by everyone, not just the most cautious. If someone asks “why the extra care”—point to cases, not just theory. Every major incident I’ve heard of, whether a near-miss or full-blown chemical fire, grew from shortcuts and ignored warnings.
A Culture of Respect for Every Bottle
Safe storage and careful handling show respect for both science and those who practice it. Newer chemists learn most from real talk and lived experience, not just laminated signs taped to the wall. If a bottle demands attention—treat it like it matters. In my own experience, chemicals remember carelessness far longer than people.
Chemistry labs carry a unique smell – often sharp and a bit intimidating. I grew up around beakers and warnings on labels. Among the notes scrawled on bottles, few things catch the eye quicker than a long name with “trifluoro” repeated. Now, looking at 2,2,2-trifluoroethyl trifluoroacetate, those instincts kick in. The words say a lot: a complicated organic compound, plenty of fluorine, and the structure hints at volatility. The next step is always the same – ask, is it dangerous? Is it toxic?
Trifluoro compounds usually don’t hang around the kitchen, but you spot them in labs, and sometimes in industry. Add enough fluorine to a molecule, and the whole structure changes. The compound in question comes with a reputation for strong odors and a tendency to react. That’s not the end of the world on its own, but it demands respect. Many similar compounds aren’t on friendly terms with skin, lungs, or eyes. Experience in university labs taught me always to avoid contact, gloves and goggles on, every time.
Information isn’t just common sense and caution. It’s right there in the safety data. 2,2,2-Trifluoroethyl trifluoroacetate lands on lists for both toxicity and environmental risk. Direct exposure, even a splash, can irritate skin or eyes. Inhalation of the vapor feels worse – think sore throat, coughing, discomfort that lingers. Over years, scientists have learned that organofluorines cross membranes and break down slowly. If something doesn’t degrade easily, it can stay in soil and water, building up where it shouldn’t.
Current Research and Known Effects
Most toxicity data points toward irritation, but research doesn’t stop there. Trifluoroacetate esters deserve extra attention because organofluorine chemistry sometimes brings unpredictability. For instance, metabolic byproducts from similar esters can cause trouble – more so than the starting chemical itself. Animal studies of comparable fluorinated compounds show liver changes and nervous system effects when exposure repeats or levels get high.
The risk doesn’t only affect the person using the substance. Accidental releases create problems for plants and aquatic systems nearby. Fluorinated chemicals in general resist breakdown. If they reach groundwater, they might stick around far longer than most ordinary solvents. Just a few micrograms per liter can spell trouble for small invertebrates in the food chain.
Controlling Hazards and Managing Risk
Keeping chemicals like this safe means strong habits, not just a set of rules. Fume hoods need proper airflow, and users should check the fan before uncapping a bottle. Disposable gloves make a difference, but double-gloving stops a splash in its tracks. Local exhaust and sealed waste containers keep fumes and residues from sneaking out too. Training matters most – every person needs a walkthrough before using any organofluorine.
Quick access to updated safety guidance helps limit exposure. Spills, even tiny ones, should never get ignored. Neutralizers for acids and adsorbents for volatile organics keep accidents from growing. Waste heads for special disposal rather than typical sink drains. Risk drops steadily when awareness, equipment, and careful planning all work together.
Anyone considering work with 2,2,2-trifluoroethyl trifluoroacetate will benefit from treating it with the respect it deserves. Regulations and guidelines succeed only if people choose to follow them with care – every day, every use. The compound’s quirky name hides a potential for real harm, but mindful practice keeps danger in check.
Hunting down chemical reagents like 2,2,2-Trifluoroethyl Trifluoroacetate might seem like a niche problem, but for research labs or chemical manufacturers, it’s a very real part of day-to-day science. I’ve seen countless colleagues stuck in procurement puzzles, scanning catalogs, waiting for quotes, and trying to figure out shipping timelines. Ever since tighter regulations started reshaping the global supply chain, finding specialized compounds has turned into a real test of patience—and persistence.
What Pulls This Compound Into Conversation
In synthetic chemistry and pharmaceuticals, 2,2,2-Trifluoroethyl Trifluoroacetate plays a crucial part in organic transformations. For anyone making fluorinated compounds, this reagent becomes a tool you don’t easily substitute. Its unique structure offers a powerful way to introduce trifluoroethyl groups, which isn’t something you swap out with some off-the-shelf replacement. That’s why skilled chemists trust suppliers who can vouch for purity, batch consistency, and reliable documentation.
The Practical Reality of Availability
Anybody hunting for this chemical will run into a trade-off between accessibility and regulation. Major scientific suppliers—and by that I mean established names with industry clout—stock 2,2,2-Trifluoroethyl Trifluoroacetate if the demand lines up with their supply chain. Sigma-Aldrich, TCI, Alfa Aesar, and other heavyweights often list it online, sometimes with various grades, but stocks might thin out or backorders could stretch from weeks to months. There’s no guarantee of next-day shipping. Some suppliers restrict sales to businesses, licensed research facilities, or government labs. Documentation runs deep: purchase orders can trigger requests for end-use declarations or proof of expertise.
As soon as the request spills across borders, things get even more tangled. In my experience, import rules spark extra paperwork. Customs can delay or even confiscate chemicals if safety or security standards don’t line up. Sometimes, customs agents get hung up on a single missing piece of paperwork, and the shipment languishes until someone chases down the oversight. When the product lands in ports categorized as hazardous, logistics slow even further. That’s the reality: regulatory oversight isn’t a box to tick; it can mean the difference between a project running smoothly and grinding to a standstill.
Who’s Actually Buying It—and Who Might Get Turned Away
Professional communities such as academic labs, biotech companies, and some pharmaceutical startups find themselves sifting through purchase channels much more than hobbyists. Qualified researchers generally gain approval if paperwork aligns. Hobbyists or unofficial buyers rarely succeed—not because the chemical is classified as the highest risk, but because suppliers play it safe. Sending out toxic, reactive, or niche chemicals to unknown addresses isn’t just a bad business move, it’s an ethical decision that sits at the core of building trust in science.
The internet might tempt some people to look for loopholes via online marketplaces or gray market websites, but this opens a can of legal and ethical headaches. Product authenticity can’t be verified. Packing and shipping standards fall away. Those who cut corners often find themselves in legal hot water or, worse, endanger themselves and others. Trustworthy suppliers require credentials—and for good reason.
Where Can Availability Improve?
Transparent supply chains would change the conversation. If manufacturers and distributors listed stock numbers and shipment timelines up front, labs could plan projects more reliably. Predictable pricing and better cross-border tracking would keep more scientists focused on experiments, not inventory management. Building relationships with trusted vendors pays off, and keeping licenses up to date speeds up regulatory checks.
Securing specialty chemicals takes more than a quick search and a click. It takes trust, planning, and—sometimes—a bit of footwork. Those looking to purchase 2,2,2-Trifluoroethyl Trifluoroacetate should expect a few hurdles, but with preparation and patience, the reagent reaches those equipped to use it responsibly.