2,2,2-Trifluoroethylamine: Tracing the Story, Uses, and Challenges
Looking Back: The Path to 2,2,2-Trifluoroethylamine
Chemists have spent decades looking for compounds that give them a new edge in pharmaceuticals, materials science, and life sciences. 2,2,2-Trifluoroethylamine sits in a corner of this history, gathering attention for its three fluorine atoms stacked on a single carbon, bringing a set of chemical behaviors quite different from its non-fluorinated cousins. Fluorinated amines like this one emerged once chemists learned to handle and tame these tricky compounds in the mid-1900s, riding the wave of organofluorine chemistry. Understanding how to work with and prepare these molecules has made them go from the oddity shelf to key tools for researchers and manufacturers alike, often showing up in projects aiming to fine-tune molecule properties for industries as varied as medicine and electronics.
Decoding the Basics: What Stands Out About 2,2,2-Trifluoroethylamine
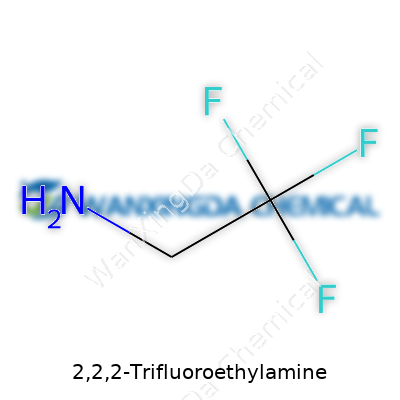
When it comes to 2,2,2-Trifluoroethylamine, the jump from regular ethylamine couldn’t be clearer. Replace three simple hydrogens in the methyl group with fluorines, and everything from reactivity to biological interaction changes. This small change makes the molecule more electronegative, more resistant to metabolism, and a lot less prone to unwanted reactions. It’s a clear signal to any chemist: add fluorines, and you remake the whole personality of the substance. This property has proven valuable for companies hunting for drugs with better metabolic stability or materials with unique dielectric properties.
Getting a Handle on Its Properties and What They Mean in the Real World
Start thinking about handling 2,2,2-Trifluoroethylamine in the lab, and you notice right away it’s a colorless liquid with a strong, ammonia-like odor. Its boiling point sits lower than plain ethylamine, reflecting the volatility that fluorine imparts. Its miscibility with water isn’t as broad as some common amines, which means storage and handling need more thought, especially for large-scale operations. Chemists also note the molecule’s outstanding resistance to oxidation and unique acid-base behavior, which has practical implications for everything from clean-up protocols to reaction planning.
Labels Aren’t Just Bureaucracy—They’re Safety
Reading the technical specifications and label instructions for trifluoroethylamine always leaves an impression: safety comes first. Reports from decades of industrial chemistry teach a lesson or two about this. Despite being less reactive than some cousins, it can give off toxic fumes, trigger allergic reactions, or pose a significant flammability hazard in poorly controlled environments. Labels and safety data sheets developed over years of collective learning now stress the importance of gloves, eye protection, and proper ventilation. The voice of experience says: treat new chemicals with respect, and always rely on proven protocols.
The Art and Science of Making Trifluoroethylamine
Making trifluoroethylamine has challenged chemists for years. The most quoted method involves the nucleophilic substitution of 2,2,2-trifluoroethyl halides, typically with ammonia or a primary amine as the nitrogen source. Handling the halides brings its own risks, given their toxicity and volatility, requiring fume hoods and trained hands. Improvements in synthetic methods have focused on reducing waste, controlling side products, and driving up yield, all goals shaped by rising costs and tighter environmental regulations. Recent research has moved toward greener synthesis pathways, an encouraging sign that the future points to sustainable chemistry.
Pushing Boundaries: Chemical Creativity and Modification
The versatility of 2,2,2-Trifluoroethylamine becomes crystal clear in the ways chemists twist and tweak it for more complex molecules. The amine function gives a handle for acylation, alkylation, and even cyclization reactions, opening routes to trifluoromethylated pharmaceuticals and agrochemicals. Medicinal chemists have latched onto these transformations, looking to build compounds with stronger receptor binding, altered bioavailability, or improved metabolic profiles. The knowledge built up from these efforts now feeds into automation and high-throughput discovery, where trifluoroethylamine acts as a reliable backbone for entire libraries of advanced compounds.
A Lot of Names, Same Chemical
Some people in research circles still call this compound trifluoroethanamine or even ethanamine, 2,2,2-trifluoro, highlighting the way naming conventions swing with trends and regional habits. The core identity stays rooted in the CAS number and chemical structure, letting laboratories and regulatory agencies cut past confusion. For researchers moving between countries or regulatory environments, knowing these synonyms streamlines information sharing and helps avoid costly mistakes with ordering or compliance.
Safety Standards and the Lessons of Hard Experience
Safety practices surrounding 2,2,2-trifluoroethylamine tell a story of learning through near-misses and detailed reporting. Early workers sometimes underestimated the risk, judging its hazards against less volatile amines. Today, the rules are clear: full-face protection for handling large quantities, flame-proof cabinets for storage, and never underestimate the risk of skin contact. Regular training for laboratory staff builds muscle memory around spill response and ventilation checks, which over time reduces accidents and ensures more consistent product quality. My own time in the lab hammered home the truth—skipping safety steps for the sake of speed rarely ends well.
Where It Shows Up in the World
The biggest practical push for 2,2,2-trifluoroethylamine remains its growing use as a building block in pharmaceuticals, especially for drugs built around nitrogen-containing scaffolds. The trifluoromethyl group sharpens drug properties, giving chemists a way to design molecules that last longer in the body or hit their biological targets with greater precision. Beyond medicine, electronics manufacturers chase the unique properties fluorinated molecules bring to specialty polymers and advanced coatings. In agriculture, modified amines feed into pesticide development, where new chemical shapes hold the answer to rising resistance and environmental pressure. Each sector brings its own demands, pushing researchers and producers to stay agile, innovative, and responsive.
Research, Toxicity, and the Balance with Progress
Scientists keep digging into the toxicology of trifluoroethylamine, knowing that every new application brings new exposure risks. Animal studies have shown potential effects on the liver and central nervous system, pressing the case for careful dose control and long-term monitoring. At the same time, researchers explore how trifluoroethylamine fits into green chemistry initiatives, considering life-cycle impacts and strategies for safe disposal. Industry leaders have a clear responsibility to weigh benefits and risks—an area where transparency and strong governance make a tangible difference.
Where Are We Headed?
The story of 2,2,2-trifluoroethylamine still unfolds. With the drive to create drugs that last longer and do their jobs with greater accuracy, this little molecule keeps its place in the toolkit. The expanding global focus on sustainability and responsibility challenges both the production methods and end uses of fluorinated compounds. Researchers dig into next-generation approaches: biocatalytic transformations, waste reduction pathways, and designing molecules with built-in degradability. If my own conversations with chemists are any indication, the spirit of careful experimentation and relentless curiosity will keep pushing this corner of organofluorine chemistry forward, always looking for that next discovery, and always weighing risks and rewards in a world that expects more from both science and industry.
A Look at Its Place in Modern Chemistry
2,2,2-Trifluoroethylamine doesn’t get much attention outside of labs and pharmaceutical meetings, but this compound shapes several industries that most folks rely on every day. With its three fluorine atoms and a simple amine group, it’s more than another quirky molecule—it fills a specific need among chemists hunting for new drugs and advanced materials.
Why Do Chemists Care So Much About It?
Creating new medicines these days feels less like cookbook chemistry and more like problem-solving at the atomic level. 2,2,2-Trifluoroethylamine gives researchers a special tool in that puzzle. Adding its trifluoroethyl group to potential drug molecules can make a huge difference. Sometimes the whole behavior of a drug changes—across my own work, I’ve seen how drugs that break down too fast inside the body start to last longer simply because this small fluorinated group resists the enzymes that usually chop other molecules up.
Drug companies often face the headache of balancing how well a medicine works with how long it sticks around without causing side effects. Swapping in 2,2,2-trifluoroethylamine can mean a jump in both durability and selectivity. That’s not marketing talk—that’s a matter of less frequent dosing and reduced toxicity for patients. In research, I’ve helped tweak molecules aiming for better brain penetration. The trifluoroethylamine building block shows up often in these projects, since it can boost a drug’s ability to cross cell membranes without spiraling out of control in the body.
Beyond Pills: Other Practical Applications
The story doesn’t stop at pharmaceuticals. Agrochemicals—the chemicals used to protect crops—benefit from the same stubbornness against breakdown. Farmers need products that last through rain and sunlight, and the trifluoroethylamine group adds that level of durability. In herbicide and fungicide development, swapping in this group can tip the scales toward better crop yield with fewer chemical applications.
In specialty materials and polymers, the trifluoroethyl piece holds value for its chemical resistance. Some companies use it in making fluorinated coatings or polymers that fight off stains and corrosion. While you won’t spot trifluoroethylamine on the ingredients list of your favorite nonstick pan, chances are high that chemists used it—or something like it—somewhere in the development process.
The Big Question: Responsible Chemistry
Having seen environmental debates up close, I can’t ignore the concern: persistent molecules like those containing trifluoroethylamine tend to stick around in soil and water longer than many natural chemicals. There’s a real push in the industry to track what happens throughout the whole life cycle. Responsible chemistry means constantly developing safer routes for making and breaking down compounds like this.
Research continues, aiming for more biodegradable versions or processes that minimize release. Regulations shift quickly. Chemists now spend as much effort on end-of-life planning for molecules as they do on initial design. Open discussion between regulators, researchers, and industry shapes the future—finding that sweet spot between performance and impact.
Looking Forward
For scientists, this molecule represents many years of effort to fine-tune performance at the smallest scale. While the average person may never hear about 2,2,2-trifluoroethylamine, their health, food supply, and household products are all touched by the choices made in the lab. That’s worth paying attention to as the call for safer, more sustainable chemistry gets louder each year.
Understanding the Makeup: C2H4F3N
People in the lab talk formulas like a second language, but for most folks, staring at a formula like C2H4F3N can be like reading a license plate. That’s 2,2,2-Trifluoroethylamine — a compound that does a lot more than its name suggests. Its makeup comes down to two carbons, four hydrogens, three fluorines, and one nitrogen. Jargon aside, this tiny cluster of atoms fuels progress in pharmaceuticals, agriculture, and specialty chemicals.
The Reason Chemists Keep Coming Back
2,2,2-Trifluoroethylamine is not one of those chemicals you’d come across in your daily life, unless your day involves a lab bench. In drug labs, its trifluoromethyl group stands out. Adding fluorine atoms to molecules tends to boost the medicine’s ability to survive longer in the body. It’s no exaggeration that the pharmaceutical industry leans on molecules like this to make drugs work better, sometimes with fewer side effects. Ask anyone who’s wrestled with drug design — swapping out a hydrogen for a fluorine can change everything.
It’s easy to overlook basic amines like this because the name sounds plain. Once you get into organic synthesis, though, it becomes clear that the building blocks matter as much as the skyscraper. With three fluorines hanging on one end, this molecule changes the rules. Compounds containing trifluoro groups tend to be less reactive in ways that help chemists steer reactions just where they want them to go.
Health, Environment, and Industry Impacts
There’s a push for greener chemistry worldwide. Fluorinated chemicals, including this one, get a second look for environmental impact. High persistence in the environment — what some call “forever chemicals” — causes headaches for regulators. That matters for a reason: chemicals that stick around in water or soil can enter food chains, affecting health and wildlife. Anyone who’s seen headlines about perfluorinated substances knows that discussion won't end soon. Companies weighing chemical choices can’t leave sustainability out of the equation.
How to Balance Progress and Responsibility
Progress never stands still. Medicine, crop protection, electronics all reach for molecules tailored for durability or activity. The trick comes in making these advances without leaving a mess behind. The chemical industry now aims for molecules with useful properties but less long-term persistence. Focusing research on biodegradability or designing compounds that break down more easily helps prevent buildup in the environment.
Rethinking the future of chemicals means listening to researchers, regulators, and the public. Finding safer replacements, or designing molecules wisely from the start, has grown into both an ethical and practical goal. Funding for green chemistry, better testing, and open discussion can give everyone a stake in where things go next.
Real-World Choices Matter
The story of 2,2,2-Trifluoroethylamine isn’t just one formula in a pile — it’s a reminder that even simple molecules pack big consequences. Decisions in the lab shape what ends up in products on shelves, in waterways, and impacting health far beyond the research bench. Getting the most benefit out of chemical innovation means weighing every new formula carefully, looking for the balance between usefulness and responsibility. As new challenges keep cropping up, the lessons from compounds like this will stick around far longer than anyone expected.
What We Know about 2,2,2-Trifluoroethylamine
2,2,2-Trifluoroethylamine is a small molecule, and anyone in the field of chemistry or pharmaceuticals knows it comes up often in synthesis projects. This chemical does its job by helping chemists tweak the properties of compounds, especially when they need to add a trifluoromethyl group. Its role seems minor, barely making a blip outside a laboratory, but handling such chemicals isn’t anything to shrug off.
Hazards and Toxicity Concerns
The first red flag with 2,2,2-Trifluoroethylamine comes from its chemical structure — trifluoromethyl groups boost both reactivity and volatility. I have memories of handling small amines in graduate labs, and my rule of thumb stayed the same: work in a well-ventilated chemical fume hood, never treat these as harmless, even for a moment.
Fluorinated compounds tend to show higher persistence in the environment and sometimes in living tissue. The trifluoromethyl group ramps up molecular stability, so spills or accidents pose real problems, not just for the lab but for broader waste streams. Although peer-reviewed toxicological data for 2,2,2-Trifluoroethylamine is limited, known effects from related amines raise enough warnings. Inhaling vapors or absorbing the compound through skin can irritate mucous membranes and respiratory tracts. Some amines, especially with fluorinated tails, may pass through skin fairly easily — I remember gloves being discarded frequently, and safety goggles fogging from the constant cycling of air from hoods.
Regulatory guides, such as those from the European Chemicals Agency, often set exposure limits based on structure and analogy, even without extensive human data. They categorize many small fluoroamines as hazardous unless proven otherwise. It's not uncommon for these chemicals to earn hazard labels for acute toxicity, environmental persistence, and irritation risks.
Why the Conversation Matters
Chemists tend to downplay some risks, especially when pressed by research deadlines or budget stress. The reality is this: even with a chemical less known than its cousins, the approach always involves respect for unknowns. Short-term effects — like headaches and skin irritation — matter, but there can be subtler, longer-term exposures that are trickier to trace. If someone spends every day in proximity to 2,2,2-Trifluoroethylamine without safeguards, the small risks start to add up.
What worries me more is the gap between professional laboratory protocol and standards in other industries where this chemical turns up. Not all users have chemical training, yet the dangers don’t diminish outside a lab. Industries without robust risk controls or trainings can unwittingly expose staff. The reality is that industrial accidents often stem from familiarity and haste, not outright ignorance.
Towards Safer Handling and Responsible Use
Best practice involves a chemical fume hood, gloves rated for organics, goggles, and snug-fitting lab coats — these steps don’t just protect from splashes, but from vapors and residues that collect on hands or sleeves. Routine air monitoring or even simple ventilation upgrades reduce long-term exposure. Safety data sheets are useful starting points, but nothing replaces face-to-face safety briefings or peer observation.
We also need better systems for chemical waste. Fluorinated amines should not go down the drain or out in regular trash streams. Specialized collection and incineration facilities exist, and chemical companies or institutions must make them accessible. Community education matters, too, so folks outside the lab understand these chemicals can cause harm and deserve to be treated with caution.
Recognizing potential hazards in chemicals like 2,2,2-Trifluoroethylamine isn’t about paranoia or red tape. It comes down to self-care, responsibility to neighbors, and respect for science — lessons any chemist probably picks up within the first few weeks at the bench, and ones that matter just as much on the hundredth day as the first.
Chemistry that Packs a Punch
In any lab, it pays to respect the quirks of the chemicals on hand, especially when dealing with something as sharp as 2,2,2-Trifluoroethylamine. This compound doesn’t behave like the stuff you spill on your kitchen counter. Belonging to the family of fluoroalkylamines, it has proven valuable for drug discovery and specialty synthesis. But, anyone who’s spent time in a lab knows: one wrong move with a volatile amine can make for a story you don’t want to tell.
Moisture and Air: Not Its Friends
Tales passed along from lab to lab signal the same advice: keep it dry and tightly capped. Trifluoroethylamine interacts poorly with air and moisture. Letting it contact water in any form can spark unwanted reactions and may degrade the purity of the compound over time. Some chemists share stories about surprising color shifts or sticky bottles left open for too long. Their troubles aren’t wild legends—they show that even slight humidity can change the game with this amine. A simple glass bottle with a tight-sealing cap works, but trusting old stoppers isn’t wise. Rely on modern seals for proper protection.
Choosing the Right Spot
Leaving this chemical on just any shelf invites trouble. Warm spots near heat sources can increase evaporation, which means both safety concerns and wasted money. Locking up trifluoroethylamine in a cool, well-ventilated cabinet keeps both its vapors and temperature in check. My own experiences in university research groups hammered home the risk of cramming all reagents together. Fume hoods serve as ideal homes for containers that get regular use. For bulk storage, a flammables cabinet—away from acids and oxidizers—sets the safest scene.
Escape Routes: Vapor Pressure and Ventilation
Those who’ve ever caught a whiff of a leaky bottle know how fast vapor can spread. Elevated vapor pressure means a container left even slightly open fills a small space with fumes. That strong odor is more than an inconvenience; it signals potential for headaches or worse. Chemical storage rooms with dedicated ventilation lower this risk. If in doubt, open containers under a fume hood and reseal right away.
Small Details Add Up
Label everything. Trust falls short when bottles look alike. Even experienced chemists have lost time or run into hazards thanks to the wrong cap or an unreadable label. Store bottles at eye level—not too high, not on the floor—so spills are simple to spot and clean before anything escalates. Keeping compatible absorbent material, like spill pads nearby, makes a big difference the rare time mistakes happen. Getting lazy with these habits, as I learned after a minor bench mishap, costs both safety and sanity.
Staying Ahead with Good Habits
Ensuring safe storage of 2,2,2-Trifluoroethylamine rarely gets headlines, but anyone invested in chemistry, medicine, or safety can appreciate the stakes. In my experience, solid habits around sealing, separating, and cooling chemical stocks pay off every single day. Encouraging teams to check their setups, share best practices, and keep an eye on ventilation protects both the people and the results we work for.
The Real-World Value of a Small Molecule
2,2,2-Trifluoroethylamine may sound technical, but this small organofluorine compound plays an influential role in labs and manufacturing. With experience handling a range of fluorinated chemicals, I’ve seen how the details—the stuff on the label most folks overlook—can sharpen decisions and keep people safe. Knowing what you’re working with shapes everything, from how you store a bottle to the way a chemical behaves when mixed or heated.
Physical Properties: Not Just Numbers
2,2,2-Trifluoroethylamine has a strong, penetrating odor that signals its volatility. This liquid doesn’t hide—its sharp smell reminds folks not to drop their guard around the bench. A boiling point hovering under 54°C says it won’t stick around as a liquid for long if left open in a warm room. That puts a spotlight on handling: even a little carelessness sends vapors into the air. In smaller, poorly ventilated spaces, this can mean problems fast.
Density checks in around 1.2 g/cm³ at room temperature. The higher density compared to water comes from those three fluorine atoms weighing down a small, simple frame. Heftier molecules flow differently, pool in odd ways inside glassware, and sometimes settle where you least expect. The flashpoint sits much lower than most might guess, which increases risk in routine lab work. Mishaps with flammable vapors have ruined more than a few afternoons.
As a colorless, clear liquid, it doesn’t give away much by appearance. I’ve accidentally dipped a gloved finger into a nearly invisible puddle before the smell hit. The lesson there: don’t trust your eyes—use tools and keep PPE on at all times.
Why This All Matters
Many folks see trifluoroethylamine listed in pharmaceutical research or specialty synthesis. They might assume that since chemists use it, strict protocols cover every base. Yet research shows the top incidents with small amines—and their vapor emissions—stem from overlooked hazards or unfamiliarity with the compound’s properties. Real stories from the bench remind us that good protocols start with knowing your chemicals. If a reaction runs hot or the ventilation hiccups, the wrong boiling point or unnoticed odor can mean more than a ruined experiment.
Storing this compound alongside incompatible chemicals increases the risk. Even containers with tight caps leak vapors over time. I always tell newcomers in my lab: store fluorinated amines in ventilated cabinets away from acids, and check seals twice.
Solutions Grounded in Experience
Teaching new lab techs about the hazards of trifluoroethylamine goes further than handing over safety data. Hands-on training, walk-throughs of spill responses, and strict adherence to fume hoods help reduce the day-to-day risks. Fume extraction should stay on, and bottles return to ventilated storage as soon as possible.
Working with this compound also means watching out for symptoms of vapor exposure—headaches or irritation may creep up fast. Company policies should promote buddy systems and frequent air checks, especially in smaller spaces.
On the manufacturing floor, chemical engineers who pay attention to these physical traits design safer storage and transfer systems. Equipment rated for high vapor pressure avoids leaks and unexpected releases. In my experience, simple habits—closing a vial tightly, using gas-tight syringes, double-checking ventilation—make the biggest difference.
Final Thoughts
2,2,2-Trifluoroethylamine is not just another number on a safety chart. A good grasp of its real-world qualities can prevent problems before they escalate. For anyone working with specialty chemicals, facts about volatility, density, and odor aren’t trivia; they’re tools for running a safe, productive operation.