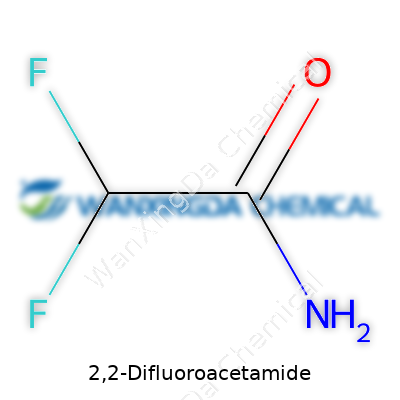
2,2-Difluoroacetamide: Chemistry, Challenges, and a Glimpse into the Future
Historical Development
Looking back on the emergence of organofluorine chemistry, 2,2-difluoroacetamide reflects a turning point that shaped molecular design over the past century. Early experimentalists faced major technical hurdles with organofluorine synthesis, battling both unpredictable reactivity and a lack of reliable methods. Electrophilic substitution barely scratched the surface, and only over decades did robust techniques for introducing difluoromethyl groups materialize. By the late twentieth century, as research on pharmaceuticals and agrochemicals accelerated, chemists appreciated how subtle electronic changes—like those delivered through the CF2 group—could transform molecular behavior. 2,2-difluoroacetamide soon caught the eyes of medicinal chemists who saw opportunities to modify bioactivity and stability in their lead compounds. Instead of an obscure laboratory curiosity, it represents the persistence and creativity that’s always driven synthetic chemistry forward.
Product Overview
2,2-Difluoroacetamide stands out as a molecule that pulls significant weight for its size. Its backbone—two fluorine atoms bonded to the same carbon adjacent to an amide function—places it among the building blocks favored by chemists looking to fine-tune the properties of larger compounds. From a practical point of view, this material doesn’t take top billing in volume-driven markets. Its real strength rests on its unique combination of electronic effects. Fluorine, as the most electronegative element, draws electron density away from neighboring atoms. When chemical designers need to suppress metabolic degradation or boost binding specificity in a new medication, they often reach for small functional groups like the difluoromethyl unit. This kind of thoughtful modification helps shape everything from drug candidates and agricultural agents to specialty polymers and advanced materials used in electronics.
Physical & Chemical Properties
The character of 2,2-difluoroacetamide comes through most clearly in its physical and chemical features. As a small, polar molecule, it usually appears as a solid or low-melting compound, dissolving easily in water and polar solvents. Strong hydrogen bonding, courtesy of the amide group, influences its solubility and plays a role in how it interacts with other molecules. The difluoromethyl group gives a significant electron-withdrawing kick. This often boosts chemical stability and slows down reactions that would normally break down similar, non-fluorinated amides. Chemists pay close attention to the acidity of the hydrogen atoms next to the fluorines, which can shift quite a bit compared to ordinary acetamide, and that can matter during synthetic steps or metabolic transformations. What you get with 2,2-difluoroacetamide is a building block with just enough chemical stubbornness to stick around under tough conditions—an asset in the lab and in practical applications.
Technical Specifications & Labeling
Most researchers working with 2,2-difluoroacetamide expect rigorous purity standards, mainly because minor impurities can dramatically change the outcome of scientific studies or manufacturing runs. Typical labeling follows international guidelines, listing the IUPAC name, major synonyms, and molecular formula (C2H3F2NO). Handling and shipping labels must spell out hazards linked to organofluorines and provide instructions for ventilation and skin protection. Lab facilities that manage this compound regularly keep detailed logs—not only to monitor purity but to ensure safe storage in cool, dry spaces, often shielded from incompatible reagents or moisture-sensitive chemicals.
Preparation Method
Synthetically, 2,2-difluoroacetamide poses a memorable challenge even for seasoned chemists. The most effective methods rely on the introduction of two fluorine atoms at the alpha position of an acetamide backbone. Traditional routes use halogen exchange reactions, with difluorocarbene precursors generated from compounds like chlorodifluoromethane or difluoroiodomethane. Newer strategies, inspired by green chemistry, swap out harsh reagents for milder conditions and safer starting materials. Electrophilic fluorination and transition-metal-catalyzed coupling have gained more traction in research circles, promising higher yields with fewer side products. Each route comes with trade-offs: efficiency, cost, and scalability matter differently in development, academic research, or pilot-plant production. Progress here has tangible impacts, often leading to increased access for those probing new chemistries or tested drug modifications in the pursuit of better medicines.
Chemical Reactions & Modifications
Once in hand, 2,2-difluoroacetamide offers up a series of reactions valuable for synthesis and materials development. That difluoromethyl group, with its inductive withdrawal, stands as both a stabilizer and a gatekeeper—shaping how the molecule behaves with acids, bases, or nucleophiles. Direct alkylation or acylation at the nitrogen is straightforward, expanding its value from a basic amide to a family of functionalized analogs. The methylene carbon, highly activated by the two fluorines, can participate in condensation or substitution reactions, opening up new rings or scaffolds that lead the way toward novel drugs and agrochemicals. Researchers have also found ways to swap out the amide group under specific conditions, extending 2,2-difluoro derivatives toward keto acids or nitriles—each with a chance at a different spectrum of applications.
Synonyms & Product Names
Looking through the literature, 2,2-difluoroacetamide hides behind a few aliases: difluoroacetic acid amide, N-difluoroacetylamine, and sometimes simply DFA. Chemical suppliers stick to standardized names, but chemists often shorten or code names for rapid communication. Navigating databases, you find these synonyms used interchangeably, especially in the context of patents or synthetic methodology papers.
Safety & Operational Standards
Safety with 2,2-difluoroacetamide centers on a respect for organofluorine reactivity and potential for biopersistence. Despite low volatility, the compound can irritate eyes, skin, and mucous membranes. Researchers working with concentrated solutions or heated reactions avoid direct contact, handle powder within fume hoods, and store all waste for specialized disposal. Local regulations demand detailed risk assessments and up-to-date Safety Data Sheets. I’ve seen seasoned scientists run thorough hazard reviews and regular safety briefings whenever a new batch arrives, recognizing that muscle memory and overconfidence are the enemies of safe chemistry. Best practices, including double-checking personal protective equipment and never working solo in the lab, keep accidents and exposures to a minimum.
Application Area
On the application front, 2,2-difluoroacetamide shows up in a surprising array of R&D projects. Its electronic properties transform how target molecules interact with enzymes and receptors, making it an attractive candidate for medicinal chemistry programs aiming to optimize drug behavior in the body. Antiviral, antifungal, and anticancer agents have all incorporated difluoroacetamide substructures, sometimes to slow metabolic breakdown, sometimes to fine-tune activity or selectivity. Agrochemical developers have leveraged similar tricks to keep pesticides stable in tough environmental conditions, while material scientists turn to the difluoromethyl group to add chemical resistance or tweak dielectric properties in new polymers and coatings. The breadth of use keeps this compound on the radar for anyone with a foot in applied chemistry, from pharmaceutical pipelines to pilot-scale material innovation.
Research & Development
Research into 2,2-difluoroacetamide brings me right back to hands-on problem solving—where theory alone won’t cut it. Over the past decade, R&D teams throughout academia and industry have worked to improve how efficiently and safely this compound can be synthesized. Enantioselective processes remain a hot topic, especially for those chasing biologically active targets where the arrangement of atoms makes all the difference. Advances in catalysis and the use of alternative fluorine sources have unlocked new reaction pathways, occasionally dropping costs and environmental impact. Many groups have also turned their attention to better analytical tools—NMR, mass spectrometry, and high-resolution chromatography—to chase down trace impurities, confirm purity, and map degradation pathways. All the while, every gain made in synthetic strategy or purification underpins new discoveries, whether it’s in bench-scale synthesis or production runs that feed early clinical trials.
Toxicity Research
Toxicology studies for 2,2-difluoroacetamide have lagged behind some of its close relatives, but a growing number of publications draw attention to both acute and long-term risks. Most lab studies note low-to-moderate toxicity in animal models under typical exposure levels, yet repeated or accidental high-level contact raises concern about organofluorine persistence in biological systems. Metabolic studies show slow breakdown, probably due to carbon–fluorine bond strength—a trait that helps in drug design but complicates disposal and environmental impact. Researchers working in regulatory science push for broader, deeper investigations into chronic exposure and possible endocrine disruption, not just for 2,2-difluoroacetamide but its transformation products. Regulatory agencies keep a close watch here, requiring comprehensive data before approving any compounds based on these building blocks for commercial use.
Future Prospects
Chemistry rarely stands still, and the story of 2,2-difluoroacetamide shows just how far persistence and small shifts in molecular design can take a field. Efforts today focus on making synthesis greener and safer, trimming down hazardous reagents and energy use while chasing higher yields. There’s a clear hunger for late-stage functionalization—routes that add difluoromethyl groups directly to complex molecules with precision, skipping steps and saving resources. In medicine, researchers look for more ways to harness the subtle tuning offered by difluoro substitution, whether that’s making cancer drugs more resistant to degradation or designing new, safer crop protection tools. Sustainability also pushes researchers to map environmental fate and develop options for safe disposal or recycling. It’s chemistry that never quite rests, shaped by the challenges of today and the possibilities in the next discovery just around the corner.
Walking through a university chemistry lab, you’ll notice shelves filled with bottles of chemicals, some of them small, white powders or clear liquids with names that sound like science fiction. 2,2-Difluoroacetamide is one of those oddities. For most people outside of research or pharma, the name doesn’t ring any bells. Yet, if you’ve studied pharmaceuticals, agrochemicals, or synthetic chemistry, you know these niche compounds form the backbone of many big discoveries.
Let’s break it down: 2,2-Difluoroacetamide is a building block—literally, a molecule that helps scientists put together more complex, targeted compounds. Two fluorine atoms swapped in for hydrogens on a standard acetamide structure change everything. A chemist looking for a way to protect a group in a reaction or to dial up the potency of a new drug can use this little molecule. Those tiny fluorine atoms alter how the whole compound behaves: they’re stubborn, resisting breakdown by enzymes, so anything built with 2,2-difluoroacetamide often lasts longer in the body. That helps drug developers craft antibiotics or cancer therapies that don’t get chewed up before doing their job.
I recall a graduate project where we studied how certain antibiotics failed to work because the bacteria could gobble them up too quickly. Adding fluorine changed the game. The difference came down to minor chemical tweaks, yet the impact for pharmaceutical research is massive. Antivirals, antifungals, anti-tumor agents—you name it, they all benefit from smart design at the molecular level. Fluorinated amides pop up in efforts to make medicines that stick around, work better, and need lower doses.
This doesn’t just matter for the medicine cabinet. The agricultural industry leans on similar chemistry to produce pesticides and herbicides that fight weeds or bugs without needing to be sprayed every week. Adding a difluoroacetamide group can slow degradation in sunlight or rain, letting crops soak up the benefits for longer stretches. Yet it’s not just about efficiency. Overusing or misusing persistent chemicals can build up in soil and water, which means researchers have to balance effectiveness with environmental risks.
If scientists want to do more with less environmental damage, the conversation starts in the lab. Using 2,2-difluoroacetamide opens the way for more potent pesticides or drugs, but researchers think carefully about where and how to deploy molecules that resist decomposition. Advances in green chemistry make a difference here: newer lab processes try to use fewer hazardous reagents and less toxic waste. Funding bodies and journals place a premium on sustainability these days. Creating a pesticide that sticks around just long enough to do the job—but breaks down safely later—takes real effort and collaboration.
Better regulation, smarter chemistry, and constant testing help keep the benefits of compounds like 2,2-difluoroacetamide out in front of the possible downsides. Pharma and agriculture depend on a steady march of innovation, but safety doesn’t take a back seat to progress. For me, the excitement in this field comes from seeing how chemistry, policy, and public health all fit together. The expertise in the lab blends with real-world testing and, hopefully, honest conversations about risk and reward, pushing us closer to smarter medicines and safer fields.
The Chemistry at the Core
Knowing what goes into common chemicals gives a stronger grasp of what they might do or how they might behave in practice. In the case of 2,2-Difluoroacetamide, things get straightforward with its chemical formula: C2H3F2NO. This formula speaks volumes, not just as a dry collection of letters and numbers, but as a practical code that shows its structure. You get two carbons, three hydrogens, two fluorines, a single nitrogen, and an oxygen. This tells chemists and anyone working with molecules enough for the basics: it’s a small molecule, but with two electronegative fluorine atoms, it’s more powerful than its size implies.
The molecular weight clocks in at 97.05 grams per mole. This number doesn’t just sit there for academics to admire—it’s key for anyone weighing or measuring this compound for reactions or lab work. Getting the mass right matters, whether you work in a research lab, an educational setting, or in industrial production where efficiency and safety aren’t just buzzwords, but everyday realities.
Experience Connects the Lab and the Real World
There’s a real satisfaction that comes from seeing theory line up with practice. My own run-ins with molecules like this started in undergraduate labs. Measuring out compounds like 2,2-Difluoroacetamide called for precision. The molecular weight played a direct role in everything—order too little, and an experiment stalled; add too much, and the risks shot up, especially with fluorinated organics. These lessons stuck because they didn’t just teach chemistry—they taught respect.
As for the structure, those two fluorine atoms make this molecule more than a textbook curiosity. Fluorine atoms pull hard on electrons, changing how the molecule interacts with others. That pull toughens certain bonds but also changes the way the amide group reacts in chemical syntheses. This is why chemists choose it—sometimes newer, fluorinated compounds like this offer more control or open up routes to new drugs and materials.
Why the Details Matter for Everyone
Understanding details like formula and weight goes beyond lab work. It touches on public health and environmental safety. Fluorinated compounds have their upsides, such as bringing stability and resistance to breakdown. At the same time, persistent chemicals sometimes slip through water supplies or resist traditional cleanup. That’s why better knowledge of structure and mass guides responsible use: more targeted clean-ups, safer labs, and smarter regulation.
A molecule’s formula isn’t just a technical fact; it’s a clue to promise and risk. One smart step is keeping thorough records and clear labeling. Transparent research also benefits the broader public. Publishing detailed data, like structure and molecular weight, lets scientists across the world replicate results, cross-check findings, and spot risks early. Real progress comes from direct collaboration and sharing—not secrecy.
Building a Smarter Path
In the end, even a modest molecule like 2,2-Difluoroacetamide makes a strong case for why understanding the basics isn’t optional. Clear data supports safe handling, helps cut down on waste, and strengthens public trust in chemistry. Keeping a close eye on each number isn’t just for the folks in lab coats—it reaches all of us, shaping products, safety, and scientific progress day by day.
What's in a Name?
2,2-Difluoroacetamide sounds like a mouthful. Most people haven't heard of it, but this compound pops up in research labs and sometimes in chemical production. It belongs to a group of chemicals that keep showing up in specialty manufacturing and advanced chemistry. People in science circles tend to look closely at substances like this because of the way small changes—like the addition of fluorine atoms—can alter toxicity, environmental impact, or health risks.
What Science Tells Us
The available research on 2,2-Difluoroacetamide doesn't stretch as far or wide as some might like. Still, a few facts come into play. Fluorinated chemicals don’t behave the same as their non-fluorinated cousins. Studies on similar items often flag concerns about environmental persistence, bioaccumulation, and effects on living cells. In other words, what works well for chemistry doesn’t always play nice in water, soil, or lungs.
The structure of 2,2-Difluoroacetamide makes folks cautious. Halogenated amides can cross biological membranes. Some related compounds have shown toxicity in aquatic tests or caused irritation in animal studies. I’ve talked to researchers who have learned to treat these molecules with respect. Skin contact shouldn’t be taken lightly, and nobody wants these compounds washing down the drain into wastewater systems.
Regulatory Viewpoint
Regulators look for hard evidence before slapping on strict rules, but that doesn't mean absence of guidelines equals safety. At the time of writing, global databases offer little in terms of clear hazard ratings for 2,2-Difluoroacetamide itself. This tells us more about a gap in testing than about the actual safety of the chemical. It’s a familiar story—chemicals keep getting synthesized faster than regulators can keep up.
Best Practices and Safer Handling
Lab veterans know that a low profile in chemical registries doesn’t excuse slack practices. Protective gloves, fume hoods, and clear disposal plans should be standard whenever handling novel organofluorine compounds. Accidental exposure to chemicals with similar backbones has sent more than a few researchers to medical staff, often with skin or respiratory complaints. I learned early on never to trust a compound just because the bottle looked new or the literature didn’t shout “danger.” Many of the worst surprises came from under-regulated compounds.
Eco-conscious readers might wonder what happens after disposal. Fluorinated molecules can stick around. Some persist in the environment long after initial use, ending up in waterways or even drinking supplies. That’s led to a growing push for green chemistry, where researchers look for alternatives with a friendlier track record.
What Lies Ahead
The world doesn’t run out of new chemicals. Chemistry drives innovation, but every new substance deserves a thorough safety assessment. 2,2-Difluoroacetamide hasn’t made headline news for toxicity, but if its structure shares the stubbornness and toxicity of some relatives, caution makes sense. Calls for more research and stricter oversight aren’t a knee-jerk reaction—they come from years of watching overlooked hazards pop up down the road.
Stronger chemical stewardship means treating unknowns with the level of care we’d want for our neighbors or ourselves. The best step is to keep pressing for broad toxicity tests, environmental checks, and public transparency around new chemicals, whether they’re old news in the lab or just hitting the market.
There’s no shortcut to storing chemicals properly, especially something as unpredictable as 2,2-difluoroacetamide. I’ve spent long hours inside research labs, sharing workspace with compounds that barely tolerate mishandling. From those days, I know one sloppy stashing job can ruin more than a semester’s work, or worse, endanger lives.
Why Storage Choices Matter
Some folks might assume this chemical could share shelf space with the regulars—simple solvents, harmless buffers. That’s not the case here. 2,2-difluoroacetamide carries enough volatility to make improper storage a major risk. It can react with moisture in the air or incompatible substances, leading to decomposition or pressure build-up, which turns a storage cabinet into a potential hazard zone.
Older colleagues used to joke that every bottle in the fridge had its own personality. Jokes aside, they had a point. Each requires specific handling, with attention to air exposure, light, and temperature swings. With 2,2-difluoroacetamide, expectations rise. Reports from various chemical safety guidelines—like those from the American Chemical Society—suggest storing it in cool, dry, tightly sealed conditions, away from direct sunlight and incompatible chemicals. That means choosing a spot in a ventilated chemical storage cabinet, not just any shelf.
Easy Mistakes Lead to Hard Lessons
I’ve seen even seasoned lab partners get a bit casual, leaving sensitive chemicals out after use, capping them loosely, or mixing reagent families in a hurry. 2,2-difluoroacetamide doesn't forgive such lapses. The presence of water or acids can encourage dangerous reactions, and the chemical's own fumes could corrode nearby materials or harm users. Once, after an overnight accident involving a similar fluorinated compound, our team spent days cleaning up, missing valuable research milestones that, honestly, a locked cabinet and working fume hood could’ve protected.
Labels and Segregation: No Room for Guesswork
One practice I picked up early: marking everything, double-checking seals, keeping an up-to-date inventory. It’s too easy to forget what’s behind a tinted cabinet door on a busy day. Clear labeling, including the chemical name, hazard class, and date received, cuts confusion and speeds up emergency response if something goes wrong. Never stack this stuff next to oxidizers or bases—separation by compatible groups goes a long way in preventing unforeseen reactions.
Solutions: Training, Culture, and the Right Tools
Some labs invest in smart storage systems that monitor temperature and humidity. While that sounds high-tech, basic vigilance still counts most. Regular chemical hygiene training keeps the dangers fresh in everyone’s minds. If management fosters a culture where safety trumps speed, then even new lab techs feel free to pause, double-check, or raise a concern. A decent cabinet, thick gloves, eye protection, and the discipline to check each bottle—these cost less than any clean-up or, far worse, a hospital bill.
Storing 2,2-difluoroacetamide isn’t just about meeting rules. Setting up the right storage space is a sign that you care about your team and the long-term health of your research. Trust me, a little extra care in storing these bottles pays off every single day in the lab.
Packing Chemicals Means Balancing Safety, Costs, and Demand
Anyone who’s spent any time working with specialty chemicals knows the job doesn't end in the lab. At some point, the question comes up—how do you get your hands on 2,2-Difluoroacetamide, and do you need a little or a lot? It’s not a household chemical, so we’re not talking gallon jugs on store shelves. Just like most niche reagents, it lands in a limited assortment of options, designed to keep the material stable and those handling it safe.
Different Labs, Different Needs
University teams and small companies often want just a handful of grams. Research budgets only have so much room for bulk buys, and sometimes you really don’t know if a molecule will work out in the early screening phase. Chemical suppliers cater to this, and the most common route for small-volume acquisition of 2,2-Difluoroacetamide is the 1-gram or 5-gram glass vial. Screw caps and PTFE liners keep out moisture and oxygen, and these vials help folks measure out only what they need in the hood.
On the other side, there’s the process chemists and scale-up engineers. Buying one gram at a time adds up quickly if you’re trying to make a kilo of an intermediate. In these cases, you’ll see packaging scale up to the 25-gram, 50-gram, and 100-gram bottles. Sometimes larger, like 250 grams, if a reliable market exists and the shelf life holds up. Most of these ship in high-density polyethylene bottles or amber glass, chosen to reduce light damage and minimize risk of leaks.
Shelf Life and Transport Call the Shots
I’ve seen more than one promising chemical go south after a supplier tried bulk packaging without considering air and moisture intrusion. Fluorinated compounds like 2,2-Difluoroacetamide can be sensitive to atmospheric conditions, so suppliers rarely push the limits on package size. A bulk drum might seem efficient, but in reality, even industrial buyers tend to request their lots packed in multiple smaller bottles within a single order. This way, they only open what they need, keeping the rest sealed tight for next time. It’s a simple move that slashes waste and cuts exposure risk.
Regulations also nudge choices. If a chemical poses a hazard, you’re not going to see it in floppy bags or thin-walled jars. DOT and IATA rules restrict both the package size and type. Suppliers can talk about best practices, but the law sets the floor for safe transit. In my experience, compliance means the pool of “available sizes” stays within a safe, predictable range.
Room for Improvement
Finding better packaging solutions could help both researchers and commercial users. Pre-measured single-use ampoules would go a long way toward speeding up workflows and cutting down on waste. Protective films and moisture-absorbing inserts are another area worth developing, especially for sensitive reagents. Every year, more customers demand tamper-evident packaging, too—and for good reason, since purity affects outcome in any serious lab.
Limited choices in size shouldn’t force anyone to buy more than they can safely store or afford to waste. Industry can learn from lab users, and vice versa. Honest conversations with suppliers help steer packaging formats in a smarter direction. Sizing isn’t just about convenience; it links directly to safety, budget, and scientific progress. The choices available for packing 2,2-Difluoroacetamide reflect these priorities, and our feedback shapes how those options evolve down the road.