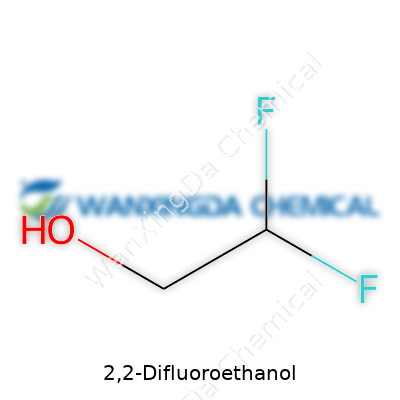
2,2-Difluoroethanol: A Fresh Look at a Unique Building Block
Historical Development
Fluorinated compounds keep drawing attention due to their strong roles across industries—from pharmaceuticals to refrigeration. 2,2-Difluoroethanol stands out as a simple molecule with two fluorine atoms clinging to a two-carbon chain. Chemists have eyed this compound since organofluorine chemistry shot up in the mid-1900s. Early fluorination techniques, not exactly safe nor straightforward, made this compound difficult to isolate. Today, improved synthesis and cleaner routes mean researchers get their hands on it without the perils of the past. The growth of demand in medicinal chemistry and materials has nudged more labs to pay close attention to its quirks and usefulness, something older manuals and databases once glossed over.
Product Overview
2,2-Difluoroethanol acts as a small, effective tool in chemical synthesis. Its alcohol group makes it reactive, while dual fluorines give molecules new behaviors—like improved metabolic stability and altered polarity. In practice, people seek it out for use in research and industrial settings. The market doesn’t flood with it, given its niche status, but what’s available shows up in laboratories chasing tough-to-make drug scaffolds or advanced polymer applications.
Physical and Chemical Properties
This colorless liquid brings a rather sharp, biting smell, a feature anyone handling it won’t forget soon. Lower boiling and melting points compared to regular ethanol signal the disruptive power of two fluorines. It dissolves in many polar solvents and in water, although you’ll notice some unexpected twists in solubility compared to its non-fluorinated cousin. It can act both as an alcohol and a moderately strong hydrogen bond donor, yet those electronegative fluorines tug at the electrons, dampening certain reactions and steering others in new directions. The reagent’s volatility and flammability make it important to handle in controlled conditions—nobody wants unknown vapors drifting in the lab.
Technical Specifications & Labeling
Suppliers bottle 2,2-Difluoroethanol with clear hazard symbols—flammable liquid, acute toxicity warnings, and aggressive tissue irritant tags. Purity varies, but high-grade material typically tests above 98 percent, sometimes 99 percent for critical research. Chemists working with this compound rely on sharp labeling—CAS number 359-13-7 and clear chemical names—to avoid confusion, especially given the narrow margin for error in organofluorine chemistry.
Preparation Method
Old days saw chemists fumbling with toxic fluorinating agents, trying to force difluoro groups onto small alcohols. Newer methods use safer, more predictable reagents like Selectfluor, Deoxo-Fluor, or DAST (diethylaminosulfur trifluoride). A common approach involves starting with a chlorinated or tosylated ethanol derivative and swapping in fluorine atoms. Specialist techniques, sometimes involving electrochemical routes, aim to boost selectivity and cut down nasty byproducts. Each method brings tradeoffs in cost, safety, and waste, but the shift toward greener chemistries marks welcome progress.
Chemical Reactions & Modifications
Once in the flask, 2,2-Difluoroethanol opens up options for making all kinds of organofluorine derivatives. That –OH group can get swapped, protected, or extended; meanwhile, the difluoro motif brings about new pharmacophores for drug design or changes digestibility in agrochemicals. One standout example: turning the alcohol into an ester or ether, often changing the biological profile of the molecule it’s part of. Electrostatic effects from the fluorines can slow or speed up reaction rates, so chemists have to test and tune conditions—one standard playbook rarely fits all. Years in the lab taught me that substituent effects in even a small group like difluoro can rescue or ruin a multi-step synthesis, meaning modern chemists don’t take this molecule lightly.
Synonyms & Product Names
Chemical circles might call it 2,2-difluoroethanol, ethylene glycol difluoride, or just DF-ethanol. Others in European research refer to it as difluoroethyl alcohol. These aliases sometimes trip up procurement, so anyone buying needs to match up CAS numbers or double-check with trusted suppliers. One instance in an academic project, a mismatch in ordering nearly brought a multi-week project to a standstill—a tough, but useful, lesson.
Safety & Operational Standards
Expect gloves, goggles, and fume hoods each time 2,2-Difluoroethanol comes out. Even minimal exposure stings the skin and eyes; higher doses provoke more serious symptoms, like respiratory distress or dizziness—no time to ignore a safety sheet. Local regulations ask for bunded storage away from sparks or heat, since its flash point drops lower than everyday alcohols. In the waste stream, difluoroethanols prove tough to break down, pushing labs to track every drop and consider incineration or controlled destruction. Institutions and regulatory bodies alike call for strict risk assessments before scaling up, and workers unfamiliar with fluorinated compounds get extra training.
Application Area
Medicinal chemistry snags the biggest spotlight, given the molecule’s role in modifying drug candidates. Adding 2,2-difluoroethanol pieces to compounds helps tweak solubility, stability, and biological interactions. Agrochemical research finds similar value, using it to alter metabolic pathways in plants or pests. Material science teams eye it for specialty polymers and surfactants, since fluorine chemistry often boosts resistance to heat or chemicals. Electronic industries—especially those pushing new concepts in dielectric materials—test its derivatives for improved current insulation. Researchers run into it again and again, not because it’s common in the wild, but because the effects edge past what traditional alcohols can deliver.
Research & Development
Academic labs around the world chase greener routes to 2,2-difluoroethanol, searching for cheaper, safer, and less polluting methods. Industry partners devote resources to scale-up techniques that dodge old-school reagents. There’s a pattern here: as chemical safety gets tighter and the cost of hazardous waste disposal keeps climbing, methods that avoid harsh fluorination win favor. Patents and literature hint at new, cleaner fluorinating agents and biocatalytic approaches, which, if they pan out, could shake up how small-scale and batch producers operate. The field could use more open sharing of these advances, since proprietary know-how sometimes bottles up sustainable solutions behind fences.
Toxicity Research
Toxicology lines remain a mix of well-established findings and open questions. Short-term handling leads to strong irritant effects, with acute inhalation or skin contact sometimes leading to hospitalization. Chronic exposure’s impact isn’t as well mapped—the fluoride ion and small-chain fluorocarbons have long, controversial reputations, making researchers cautious. Environmental safety studies say difluoroethanol doesn’t biodegrade easily, raising concern for water or soil exposure. Manufacturers and labs need to keep monitoring developments here, as more regulated environments and stricter disposal rules seem sure to follow.
Future Prospects
Cutting-edge drug discovery and eco-friendly material design both push toward more precise use of fluorinated building blocks. I expect demand for 2,2-difluoroethanol to grow—maybe not explosively, but on a steady, critical track—as designers ask for fine-tuned molecular effects in crops, pills, and coatings. Companies finding ways to produce it with less hazardous waste and at lower cost will set themselves apart. Regulatory pressure will push greener solutions to the top and might spawn fresh approaches in fluorine chemistry overall. There’s plenty of space for innovation, especially in methods to recycle or safely destroy spent products. The next years promise a tug-of-war between broader application and rising scrutiny, leading to more streamlined, responsible ways to harness this valuable tool.
Not Just Another Chemical Compound
Ask a chemist about 2,2-Difluoroethanol, and you’ll see a gleam in their eye. With two fluorine atoms attached to a two-carbon alcohol backbone, this compound packs a surprisingly big punch in research and industry. Most folks—outside organic labs—rarely hear its name, yet its fingerprints show up in areas as varied as pharmaceuticals, crop science, and even materials for new-age electronics.
Fuel for Innovation in Pharmaceuticals
Drug discovery often comes down to small tweaks—a new group here, a functional group there—that can make or break a medicine’s success. The inclusion of fluorine atoms gives pharmaceutical molecules extra stability. It helps them last longer in the body, slip across membranes, or resist breakdown from enzymes. 2,2-Difluoroethanol steps in as a building block, letting chemists create structures that aren’t possible with regular ethanol.
Instead of seeing it as a one-shot solution, chemists use it as a stepping stone to craft antifungal drugs, antivirals, or potential cancer treatments. The game of designing new medicine looks for these sorts of niche intermediates. Just a couple of fluorines can turn a good molecule into a great one.
Agrochemicals: Powering Modern Farming
Farmers want pest controls and herbicides that work fast, act selectively, and break down without sticking around too long in the environment. A fluorinated alcohol like 2,2-Difluoroethanol gives chemical companies a backbone for making next-generation crop protection tools. Engineers can hook other useful groups onto the molecule, delivering products that either disrupt pests or protect plants in ways that untreated crops miss out on.
The environmental aspect matters. Overdoing it with persistent chemicals has hurt land and waterways. Starting with building blocks that allow for efficient breakdown while doing their job means a smaller ecological footprint. If you follow research in crop science, 2,2-Difluoroethanol’s presence often signals moves toward smarter, more responsible chemistry.
Everyday Electronics Start With Odd Chemicals
You tap away on a smartphone, watch a TV, or scroll through news on a crisp electronic display. Fluorinated compounds have shaped much of this shift toward sharper screens and longer battery lives. While 2,2-Difluoroethanol isn’t the final ingredient in your display, its derivatives help create liquid crystals or polymer films that make those bright colors possible. Materials scientists use it to tweak flexibility, resistance to heat, or what happens when a screen stays on all day.
I’ve chatted with folks in materials engineering who admit that without smaller specialty chemicals like 2,2-Difluoroethanol, innovation slows down. The right building block accelerates lab work. It keeps projects moving forward rather than stuck in basic synthesis.
Safety, Access, and the Path Forward
No one likes reading about mishaps in chemical labs, and 2,2-Difluoroethanol brings risk of toxicity if handled carelessly. Following strict guidelines stays critical, especially as demand grows for more specialty fluorinated chemicals. Chemical research has to keep up, pushing for safer storage, better training, and new methods that limit exposure.
Finding alternatives that do the job with less risk stays important, too. People working in green chemistry want to swap hazardous solvents or intermediates with safer relatives. So far, few substitutes check every box, but persistent research keeps chipping away at those barriers.
Small Details, Big Differences
2,2-Difluoroethanol rarely headlines outside chemistry circles. But in the real world, its value comes from the possibilities it unlocks—a sharper medicine, safer crops, or a brighter digital display. Keeping an eye on compounds this small might feel like chasing details, yet these are the details that slowly change how we live and work.
Looking Danger in the Eye
Anyone who has worked in a chemistry lab understands: chemicals rarely forgive. 2,2-Difluoroethanol stands out as one of those substances that demands full respect. It's a clear liquid, often showing up in research circles and sometimes in industrial settings, and—like many fluorinated compounds—it offers plenty of fascinating science. Alongside the promise, though, comes risk. People sometimes underestimate small-molecule organofluorines, thinking they couldn’t possibly behave all that differently from their non-fluorinated cousins. That can be a costly mistake.
Why This Stuff Matters
A close look at 2,2-Difluoroethanol’s health effects reveals a familiar pattern if you’ve ever handled solvents: rapid absorption, strong central nervous system effects, and organ toxicity. The main danger jumps to mind right away: inhaling vapors or getting it on your skin. A 2012 toxicology review compared the risks to those associated with other fluorinated ethanols. In animal studies, exposure even at relatively low doses made for severe respiratory and nervous system symptoms. That tells a clear story: the margin for error is slim.
I learned early on to take skin and respiratory protection seriously, especially with compounds like this. Splash a little on bare skin or breathe in some fumes, and you aren’t just risking mild irritation. Neurologic symptoms, confusion, and even coma can follow higher exposure. Anyone who recalls treating a solvent-exposed coworker knows these effects play out frighteningly fast.
No Room for Complacency
It’s tempting for experienced lab workers to slide into old habits—just gloves and a standard fume hood, trusting the safety net. But real-world mishaps happen when attention wavers. 2,2-Difluoroethanol, with its volatility and toxicity, doesn’t play nice with shortcuts. The right gloves—often nitrile, not latex—make all the difference. Chemical goggles protect from unexpected splashes. A properly functioning fume hood buys time if something goes wrong.
Storage brings its own set of issues. 2,2-Difluoroethanol isn’t just flammable; heating or burning it can push out hazardous gases. Chemistry storerooms sometimes turn into poorly ventilated holding pens if no one checks the airflow. Putting this chemical on a high shelf or next to oxidizers and acids courts disaster. Most experienced teams keep it in a flammables cabinet, away from incompatible materials, with labels everyone understands.
Responding to Spills—and Learning from Them
Spills rarely follow a script. I remember a graduate student forgetting to cap a bottle during equipment repairs. In minutes, a sharp, odd odor reached us. We swept in: everyone evacuated the room, donned air-purifying respirators, used absorbent pads, and cleared up. The lesson stuck—react faster, and never work alone with hazardous organofluorines. Emergency practice and access to spill kits proved their worth.
Solutions That Start with Culture
Too often, safety takes a back seat to productivity or curiosity. Regular training is one step, but culture cements what training begins. Sharing stories of accidents and close calls—both real and near-misses—helps keep the hazard real. Supervisors, researchers, and lab managers who model best practices shape safer working habits. The chemistry does not change, but our response can.
2,2-Difluoroethanol reminds me that “routine” and “safe” never mean the same thing. Respect the risk, support each other, and the lab becomes a place for real progress. Ignore the warnings, and the cost can be much steeper than anyone expects.
Digging Into the Chemical Backbone
Most folks don’t find themselves daydreaming about alcohols with fluorine atoms, but the chemistry behind 2,2-difluoroethanol deserves some spotlight. Its structure stands out: two fluorines replace the usual hydrogens attached to the second carbon of ethanol. Imagine the familiar ethanol chain, but both hydrogen atoms on the central carbon swap places with two fluorines. The molecular formula lands as C2H4F2O.
What’s the real difference? Fluorine is no wallflower; its presence shakes up the chemical profile. Only a handful of molecules can pull off such dramatic changes with such small tweaks.
The Little Formula With a Big Impact
As someone who’s handled a variety of reagents and solvents in the lab, the impact of swapping two hydrogens for two fluorines gets impossible to ignore. The structure, written chemically as CHF2CH2OH, means the first carbon grabs both fluorines, and the second one holds the alcohol group. These fluorines tighten up electronegativity around that carbon center, pulling electrons in and twisting the old reactivity script.
This setup doesn’t just exist on paper. The inclusion of fluorine changes how the molecule behaves in chemical reactions, how it interacts with the body, and how it blends into other compounds in industry and research. The volatility shifts. The way it resists breakdown shifts. From a safety perspective, those fluorines can increase both toxicity and environmental persistence.
Why Getting the Structure Right Matters
Mistakes with this structure create trouble. It’s easy to assume it acts like regular ethanol, but the difference isn’t subtle for chemists or anyone dealing with its use outside the lab. Fluorinated compounds have notorious reputations: they stick around in the environment, ionize differently, and often mess with enzymes or metabolic processes in ways that basic alcohols don’t.
In my own chemical work, misjudging a fluorinated compound meant wasted products or, worse, creating something much harder to dispose of. The case isn’t just about recognizing C2H4F2O as a formula—it means truly understanding how small changes in a structure can cascade through an entire downstream process.
Challenges and Smarter Paths Forward
The best lesson from these tricky molecules? Respect the details. Researchers need to keep safety and environmental impact at the top of their minds. For companies relying on fluorinated chemicals, seeking greener substitutes or measuring out exactly what’s needed lowers both risk and waste. There is growing traction for alternatives with less persistent environmental effects; it’s up to both the industry and regulatory bodies to fund and reward these developments.
Teaching about these differences in classrooms, labs, and companies closes the knowledge gap. Being transparent about chemical responsibilities should never be just a box-ticking exercise.
On a molecular level, 2,2-difluoroethanol proves how chemistry refuses to play by simple rules. Even one small tweak—two swapped atoms—sets off a chain reaction that winds through the lab, the market, and right into ongoing questions about safety, responsibility, and smart chemical design.
Storing chemicals can often feel like a chore. Companies stash drums in warehouses, sometimes ignoring what might happen if the wrong container or an open window lets something dangerous loose. 2,2-Difluoroethanol demands respect. As someone who has spent years in laboratory and teaching settings, I’ll tell you: storage shortcuts have serious consequences.
A Clear and Present Risk: Volatility and Toxicity
2,2-Difluoroethanol isn’t just another bottle in the flammable cabinet. Volatility and toxicity combine in a way that takes safety up a notch. Inhalation of the vapor can trigger headaches, dizziness, and much worse with moderate exposure. Spills or leaks don’t just risk a mess—they risk lives.
These risks remind me of a close call a colleague once had after accidentally storing a volatile chemical near a heat source. The flare-up didn’t just singe eyebrows; it wrecked confidence in what should have been a routine process. Keep anything like 2,2-Difluoroethanol away from ignition sources, for the sake of your building and everyone inside.
Storage Containers Aren’t Just a Matter of Preference
Corrosive and reactive chemicals deserve proper containers. Using random glass bottles or mismatched plastic doesn’t cut it. Containers designed for hazardous solvents stop leaks and resist the slow, steady creep of vapors. Metal caps and certified safety seals work together to keep fumes locked in.
This is a lesson driven home by more than regulatory guidelines. I’ve seen containers swell and fail, spreading vapors after storage in the wrong material. Trust in proper chemical-resistant bottles—anything else invites trouble you can’t predict.
Temperature and Ventilation—Not Just Box-Checking
A lukewarm corner won’t cut it for something this volatile. Store 2,2-Difluoroethanol in a cool, well-ventilated area. Direct sun, heating pipes, and confined spaces heat chemicals up and ramp up vapor pressure fast. If you’ve ever walked into a stuffy storeroom and caught a whiff of something sharp, that’s your sign air exchange has failed.
Dedicated flammable storage cabinets with built-in venting and temperature control do more than keep inspectors happy. They buffer against unpredictable building changes: lost power, failed HVAC, or nearby construction. Having worked in spaces where a single hot day nearly turned storage areas into a hazard zone, I know how tempting shortcuts are—especially late in the day. Don’t give in. A few minutes’ diligence makes the work environment safer and preserves health.
Don’t Underestimate Training and Oversight
It’s easy to overlook the day-to-day details—labels rub off, keys go missing, logs aren’t updated. The best practice draws on ongoing training and oversight. Rotating safety briefings and double-checking inventories stop trouble before it starts. Not everyone in the lab will recognize a slightly corroded bottle or a label faded from time. Sharing stories of real-world accidents—without pointing fingers—helps underscore why the rules exist.
Many workplaces value rules on paper, but practicing good storage habits takes persistent leadership and staff buy-in. My experience says you spot the safest workplaces not by their newest equipment, but by the confidence workers show when handling hazardous materials and spotting warning signs.
Smart Storage Reflects Real Commitment
Respect for 2,2-Difluoroethanol means more than following codes. Every safe practice, every container decision, every regular check-in—these steps protect people first. Smart storage doesn’t just avoid accidents; it builds trust. It shows new students, young workers, and visitors that safety gets taken seriously—and that’s not something any workplace can fake.
A Look at What Sets This Compound Apart
People who work in chemistry labs get to know the feel, smell, and hazards of all kinds of unusual substances. Anyone who’s come across 2,2-difluoroethanol—a short, simple molecule with two attached fluorine atoms—knows it’s not your average alcohol. The stuff is colorless and liquid at room temperature, a detail you spot right away when opening the vial. But a lot more goes on beneath the surface.
Fluorine on a molecule changes almost everything. Swap two hydrogens for two fluorines on ethanol, and suddenly you’re dealing with 2,2-difluoroethanol, a substance far less friendly than the regular drinkable version. That chemical switch turns it toxic, corrosive, and sharper on the nose. The vapor can irritate eyes and skin, and breathing too much can cause real harm. Because a lot of industrial solvents or reactants evaporate quickly, you need good ventilation or even a fume hood if you’re handling this compound in the lab.
As for the chemistry, those fluorines are extremely electronegative. They suck electron density away from the rest of the molecule, making the alcohol group more acidic than ethanol’s. The pKa drops, so you’ll notice it reacts differently if you’re using it as a building block in organic synthesis. Some chemists count on that quality because it changes how things bond or break apart in a flask. Producers of pharmaceuticals or specialty chemicals might reach for 2,2-difluoroethanol when they want to add a difluoromethyl group to a more complex molecule—fluorine helps drugs last longer in the body and can even block certain metabolic pathways.
The compound’s physical side reflects in its boiling and melting points, which are higher than ethanol but lower than heavier alcohols. It dissolves well in water, unsurprisingly, because it still has that classic hydroxy group. But with its toxicity, you want no part in tasting or smelling it for too long. Even small exposures should be respected, and the EPA does list it as a hazardous material, which means disposal requires special care.
Experience tells you right away that handling a chemical like 2,2-difluoroethanol brings a layer of caution to every step. I remember colleagues running small-scale reactions with it, suiting up with extra gloves and double-checking ventilation, just to avoid an accidental splash or whiff. Chemical properties are not just numbers—they drive safer habits in real time.
Anyone seeking alternatives might ask if other alcohols without fluorine suit the same synthetic needs. The answer depends on your reaction’s goals. If you’re aiming for a specific difluorinated product, nothing else does the trick as cleanly. One fix for the risks would be using automated or closed systems that minimize human contact. Some labs already invest in these setups, especially as concerns over lab worker health keep growing.
I’ve always believed that understanding both the risks and rewards of a substance makes the work better—and safer. Chemicals like 2,2-difluoroethanol earn respect. With every property, from vapor pressure to reactivity, it shows how small tweaks at the atomic level end up shaping decisions, safety rules, and even whole production strategies in the world of chemistry.