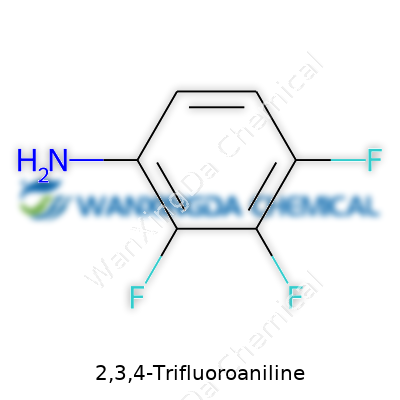
2,3,4-Trifluoroaniline: A Deep Dive into a Modern Fine Chemical
Historical Development
Chemists have eyed fluorinated anilines for more than a century, but the journey to developing 2,3,4-trifluoroaniline stands out. Early studies in aromatic amines laid the groundwork for subtle fluorine substitutions, leading to remarkable improvements in stability and reactivity. Tracing the arc from early dye intermediates through post-war agrochemical research, you see the stepwise emergence of selective fluorinated analogs. After the uptake of catalytic fluorination techniques in the late 20th century, researchers got more creative and deliberate about site-specific substitution, giving rise to molecules like 2,3,4-trifluoroaniline. Today’s chemical manufacturers and R&D labs use procedures that benefit from decades of stepwise improvements, each building on old lessons about aromatic chemistry, safety, and handling.
Product Overview
2,3,4-Trifluoroaniline has become a versatile intermediate across pharmaceuticals, agrochemicals, and specialty materials. With three fluorine atoms stitched onto the aniline ring, it offers boosted stability and unique reactivity compared to other mono- or difluorinated anilines. The molecule’s design suits the needs of scientists looking to tweak electronic properties or to add metabolic resistance for active ingredients. Research pipelines lean on intermediates like this to develop things like new drug scaffolds or to scheme up advanced coatings, often aiming for compounds that last longer and resist degradation. The upside to using 2,3,4-trifluoroaniline stretches across markets wanting more sustainable, longer-lasting products.
Physical & Chemical Properties
2,3,4-Trifluoroaniline delivers a blend of benefits chemists appreciate. Its melting and boiling points differ from those of the parent aniline because fluorine atoms really change basic physical behavior. With a moderate boiling point and solid aromatic character, this compound resists oxidation and stands up well in storage. Its solubility profile lets it mix into both polar and nonpolar solvents, making it handy in process development or analytical chemistry. The intrinsic electron-withdrawing nature of the trifluorinated ring tames the reactivity of the amine group, which opens up possibilities for selective functionalization that would otherwise turn messy with plain aniline. This small set of subtle changes means researchers can count on 2,3,4-trifluoroaniline for a repeatable process and a reliable outcome.
Technical Specifications & Labeling
Quality control has become a non-negotiable point in commercial shipments. Chemical suppliers address purity, often delivering 2,3,4-trifluoroaniline at upwards of 98 percent, with well-characterized residual water levels and certified identification by NMR or GC-MS. Safe labeling practices indicate hazard statements related to toxicity and personal protective requirements, as fluorinated aromatics usually demand attention. The bottles arrive with clear hazard pictograms, storage recommendations, and instructions relating to spill management, all meeting international standards for chemicals under GHS rules. Some batches carry extra analytical documentation tracing back synthetic routes, a direct response to pharmaceutical supply chain scrutiny.
Preparation Method
The path to making 2,3,4-trifluoroaniline usually starts with a pre-fluorinated benzene core. Chemists control fluorination carefully, often deploying selective chlorination and amination to position the amine group right where it’s wanted. Advances in catalytic fluorination or nucleophilic aromatic substitution have made these syntheses more accessible. In some workflows, direct amination leverages modest reaction temperatures and yields clean products after standard purification. Modern labs see each step as an opportunity to minimize waste streams or hazardous byproducts, especially since fluorinated intermediates draw close regulatory attention. Where earlier chemists might have settled for rough yields, current procedures rely on well-tuned protocols to balance safety, efficiency, and cost.
Chemical Reactions & Modifications
Researchers look at 2,3,4-trifluoroaniline as a launching pad for further transformations. The electron-deficient ring supports a suite of reactions, including N-acylation, sulfonation, and coupling reactions, all without losing the valuable trifluoro motif. Medicinal chemists have found that the trifluoro pattern helps steer the outcome of cross-couplings, providing access to libraries of bioactive compounds. The amine functional group can be protected, alkylated, or used to anchor side chains that imbue new solubility or reactivity. It’s this combination of reactivity and selectivity that persuades process chemists to stick with fluorinated anilines, particularly in drug discovery or fine chemical synthesis.
Synonyms & Product Names
You’ll find 2,3,4-trifluoroaniline under various names, such as 2,3,4-trifluoro-benzenamine or by its CAS number. The shorthand format—sometimes written as TFAN—shows up in industrial catalogs and research papers. Product listings occasionally mention “trifluoroaniline” along with positional numbering to avoid confusion with other isomers. Consistency in naming supports clear communication between labs, suppliers, and regulatory bodies, which matters a lot when the time comes for registration, customs clearances, or environmental impact assessments.
Safety & Operational Standards
Safe use of 2,3,4-trifluoroaniline needs vigilance, not shortcuts. Aromatic amines raise flags for toxicity and require gloves, goggles, and working chemical fume hoods. Material safety data sheets outline steps for spills and accidental exposure, with an emphasis on avoiding skin contact and inhalation. Waste handling protocols send spent materials through incineration or specialized chemical disposal, especially since many fluorinated byproducts resist biological breakdown in the environment. Safe handling does not end in the lab—proper labeling and training make sure everyone in the chain, from transporter to technician, understands what they’re working with. These practices not only protect people but also build trust in the chemical’s responsible use.
Application Area
2,3,4-Trifluoroaniline’s unique structure puts it on the short list for pharmaceutical synthesis, agrochemical discovery, and specialty polymers. Drug developers appreciate its ability to block metabolic breakdown by stabilizing active molecules, extending their half-lives in the body. In crop protection, engineers turn to trifluorinated intermediates for safer, more persistent herbicides or fungicides. Materials scientists see value in trifluorinated building blocks, using them to generate advanced coatings or polymers that shrug off harsh chemicals and resist UV light. The breadth of use ties back to the combination of aromatic character and tailored electronic properties, which only come from clever placement of fluorine atoms.
Research & Development
R&D teams consistently explore new uses for 2,3,4-trifluoroaniline. Structure-activity relationships hold the key, particularly for those searching for alternatives to more toxic or less stable amines. Ongoing studies try out different coupling partners or look to improve process efficiency with greener solvents and catalysts. The search for sustainable chemistry often runs up against the challenge of fluorinated waste, pushing scientists to devise better recycling or degradation methods. In pharmaceutical labs, the focus stays on feeding 2,3,4-trifluoroaniline into automated synthesis robots, churning out vast libraries for biological screening. These parallel efforts keep the molecule front and center in modern lab work.
Toxicity Research
Concern about toxicity accompanies all aromatic amines. For 2,3,4-trifluoroaniline, early studies suggest careful handling, as metabolic byproducts may be biologically active or persistent. Toxicology screens focus on short- and long-term exposure risks, looking at possible effects on liver enzymes or links to mutagenicity. Regulatory agencies often demand supplemental testing before greenlighting new uses, which slows down product pipelines but ensures safety. Researchers keep refining in vitro and in silico methods to predict toxicity profiles earlier in development, reducing the number of animal studies needed. Understanding risks means data-driven decisions, cutting down surprise liabilities for companies and protecting both human health and the environment.
Future Prospects
The future looks promising for 2,3,4-trifluoroaniline as industries prioritize compounds with improved performance and longer life cycles. Advances in selective fluorination and greener synthesis methods could open up new windows for safer, more cost-effective manufacturing. If researchers crack the code on degradable fluorinated molecules or better post-use handling, this could move trifluoroaniline-based products into sustainable chemistry’s spotlight. Pharmaceutical and agrochemical pipelines stay hungry for new scaffolds, and the unique hit that comes from fluorinated amines keeps 2,3,4-trifluoroaniline on the list for the next wave of breakthroughs. Environmental awareness and regulatory oversight remind everyone that progress depends on responsibility, not demand alone, framing the narrative for innovation in the years ahead.
Fluorine’s Grip: What Three Atoms Change
Chemistry hands you a benzene ring, throw on an amine, then pipe in three fluorines. That’s 2,3,4-Trifluoroaniline. At first glance, it sounds like a roll call from the periodic table. Under the microscope, though, those three fluorines transform everything in ways that matter for researchers, industries, and anyone thinking about the future of chemicals in modern life.
The Bones of the Molecule: Drawing It on Paper
Picture a hexagonal benzene ring. Take the aniline structure—a benzene ring with an amino group attached. Now, stick fluorine atoms at the second, third, and fourth positions next to the amine. Those positions matter because fluorine doesn’t just swap out for hydrogen and sit quietly. The chemical structure is C6H4F3NH2: a trifluorinated cousin of the humble aniline. What gives these three tiny atoms so much clout? Their strong electronegativity.
This isn’t just a chemistry trivia point. These fluorines pull electrons toward themselves, making the entire ring less reactive, especially for things like electrophilic substitution that work normally on anilines. The amine sticks around, but it isn’t as eager to interact as you might think. That electron-withdrawing power also gives 2,3,4-Trifluoroaniline a different flavor in synthesis and reactivity than unmodified aniline.
Why the Details Matter
In research, detail means precision, and precision helps people avoid mistakes. Anyone who’s wrestled with organic synthesis can tell you: a small change—like flipping the position of fluorines—can make molecules stubborn to react or form unexpected products. Fluorinated anilines sometimes end up as ingredients in agrochemicals, pharmaceuticals, and materials. That happens because fluorine can change how a molecule behaves inside living cells or in industrial reactions. Noticing the difference between a single fluorine and three, or moving them around the ring, can spell the difference between a therapeutic agent and an environmental problem.
There’s a broader story here, too. Fluorinated compounds don’t break down the way other organics do. Their persistence in the environment is legendary—a fact the chemical industry and environmental groups both know well. Part of being responsible in science and industry means respecting these differences, understanding the potential risks, and making informed choices about designing safer chemicals for people and the planet.
Moving Toward Solutions
Researchers looking ahead see an open field for improvement. One trend is to develop greener synthesis methods—thinking of less waste, milder conditions, and replacing hazardous reagents. Finding new ways to recycle or destroy persistent fluorinated chemicals could close the loop so environmental impact decreases. In the lab, teaching budding scientists the value of structure and substitution patterns leads to fewer mistakes, more innovation, and safer results.
2,3,4-Trifluoroaniline shows how a few simple changes to a molecule can rewrite the rules of reactivity and behavior. Keeping an eye on such details doesn’t just help chemists make better products. It’s a key ingredient for building a more responsible science that responds to real-world concerns and keeps moving forward.
Most folks scrolling through reports about chemicals might never give 2,3,4-Trifluoroaniline a second thought. Yet, in my years of digging through the tangled world of specialty chemicals, it’s the niche molecules like this one that quietly shape entire industries behind the scenes. 2,3,4-Trifluoroaniline doesn’t land headlines, but its fingerprints show up everywhere from drug development to crop protection.
Making New Medicines Possible
Drug discovery turns into a marathon of trial and error, and many researchers need building blocks that bring just the right blend of stability, reactivity, and uniqueness. 2,3,4-Trifluoroaniline delivers on those fronts. Chemists often turn to this compound to slip fluorine atoms into their target molecules. Why? Fluorine changes how a drug behaves—helping medicines last longer in the body or hit their targets with more punch. That trifluorinated aniline piece lays the foundation for a wide range of therapies, from cancer treatments to new antibiotics. I’ve chatted with medicinal chemists who treat compounds like this as prized puzzle pieces, because they give a lead compound that extra boost in a competitive research landscape. According to studies in medicinal chemistry, adding fluorine can improve the absorption and metabolic stability of new drug candidates, making 2,3,4-Trifluoroaniline a sought-after tool in the chemist’s toolbox.
Pesticides for a Changing World
Feeding a growing population means fighting off diseases and bugs that damage crops. A lot of modern pesticides lean on clever chemistry, and fluorinated compounds are especially valuable. 2,3,4-Trifluoroaniline serves as a starting point for many crop protection agents because these fluorine atoms help molecules resist breakdown under sunlight and in wet fields. Farmers look for solutions that last through tough weather, and this little compound helps chemists deliver just that. Companies in agricultural science adapt compounds like this to meet stricter regulations and still cut down on crop losses.
Specialty Materials and Color Chemistry
Not all impacts stay in the obvious places. I’ve seen applications drift over into the world of dyes and specialty coatings. Fluorine-rich anilines give pigments a shot at deeper hues and improved weather resistance. This makes a real difference for products left outdoors, where sunlight can quickly bleach color or damage surfaces. Whether it’s an automotive finish or the dye in your favorite outdoor gear, better durability can often trace back to specialty craftwork with trifluorinated building blocks like this one. Skilled materials scientists look for that golden balance between cost, performance, and long-term reliability, and 2,3,4-Trifluoroaniline offers just enough stability and reactivity to blend into breakthroughs.
Challenges and the Way Forward
No discussion about specialty chemicals would feel complete without talking about the responsibility that comes with such power. Fluorinated molecules have tough bonds that resist breaking down—great for product life, but a headache if they escape into soil or water. I’ve followed debates about persistent organic pollutants where poorly managed fluorinated compounds turned up in the wrong places. Solutions aren’t easy, but better waste management and greener synthesis methods hold promise. Many labs focus now on new catalysts and cleaner production chains, working to make the most out of what these molecules offer without leaving lasting problems behind.
2,3,4-Trifluoroaniline might be a mouthful, and it may never grace the front page. Yet its role in medicine, agriculture, and materials science shows how subtle advances push society forward, quietly shaping the modern world for the better.
Getting Close to Trifluoroaniline: What People Should Know
I once talked with a friend about safety in chemistry, and we both agreed: a moment's carelessness can leave a mark. 2,3,4-Trifluoroaniline doesn’t attract headlines, but it’s the kind of compound that quietly demands respect. It’s a liquid, aromatic, often clear or slightly yellow, and wears the badge of an aniline derivative. Labs and chemical suppliers move it around for research or specialty projects. Few outside a lab ever see it, but the risks stick around for anyone handling it.
Toxicity and Its Real Effects
I’ve seen what happens when people underestimate compounds in the aniline family—headaches, nausea, sometimes breathing trouble. Trifluoroaniline’s structure gives it the edge in reactivity and volatility. Exposure might start off small: skin contact, breathing in the vapor, maybe a splash near the eyes. On paper, safety data calls it harmful if swallowed or inhaled, and it can irritate skin and eyes. Some older research links similar chemicals to effects on blood and the liver. Once exposure piles up, small risks turn big. Folks handling it need to see beyond the label and realize no experiment is worth a trip to the ER.
Handling With a Level Head
The temptation to skip gloves or goggles always lurks, but this is where trouble begins. Splash protection, gloves resistant to chemicals, and working in a hood change the odds. I keep a habit: no open bottles unless airflow clears the fumes. Even if it’s just measuring out a few milliliters, spills and splashes happen. For trifluoroaniline, the right gear protects more than just hands; it shields lungs, eyes, and skin from something that lingers and soaks in fast.
Some think gloves are enough on their own, but the right glove matters. Nitrile or butyl types work well against most organics. Cotton or latex, on the other hand, can leave you exposed. Before breaking out any chemical with “fluoro” or “aniline” in its name, I always double check the SDS and rethink the workspace layout. Is my eyewash nearby? Is there spill material on hand? These steps sound simple but, in practice, save hours of regret.
Storing Without Surprises
Bottles of trifluoroaniline belong away from acids, oxidizers, and heat. I’ve seen shared chemical fridges crammed with incompatible materials, and that’s a nightmare scenario. Separate storage, tight caps, and clear labels kill confusion. Waste should never pour down sinks, and containers deserve more than just a quick rinse. Disposal means using designated collection—no shortcuts.
Being Ready for the Worst
Accidents don’t schedule themselves. Even seasoned chemists get caught off guard. Each lab session, I check my emergency procedures, review spill kits, and make sure exits stay clear. If someone’s exposed, the plan is to wash off, find fresh air fast, and call for help. Treatment might feel routine, but early action limits damage. Over the years, I’ve seen how simple routines—like reading labels twice and sharing safety reminders—build a culture that values health over speed.
Moving Forward: Culture Over Compliance
No rulebook can cover every situation. Good habits, open conversations, and real respect for risk—the trifluoroaniline story shows how chemistry keeps people humble. Small steps protect the person behind the beaker and everyone sharing the workspace. A bit more time spent on safety pays off with a lot less worry in the long run.
Not All Chemicals Are Created Equal
Anyone who's spent time with arylamines in a lab knows that trifluorinated compounds aren’t just about flashy molecular structure. The question of purity for 2,3,4-trifluoroaniline is the sort of thing that splits a smooth reaction from a wasted week. I’ve seen plenty of shelves lined with amber bottles, but only some of those contents can handle serious analytical or synthetic work. Here, the numbers matter more than the appearance.
Most chemists expect at least 98% purity from a reputable source, sometimes nudging higher for pharmaceuticals or electronic materials. Impurities—fluorinated or not—may seem minor, but they leave a trail of side reactions and analytic noise. Things get dicey when the last percent includes moisture, unknown organics, or traces of other fluorinated anilines. For the uninitiated, this means that your final yield can drop, or you might chase mysterious peaks across your data.
Beyond Numbers: Specifications With Real Impact
People often talk about melting point or GC-MS results like they’re just technical jargon. But these specs are the proof you can trust a shipment. A clear liquid at room temperature that hits the right melting point signals good things, but spectroscopic data (NMR, IR) nail down the identity, and anything less makes experienced chemists nervous. Years ago, we skipped a step on a small-scale run, trusting a supplier’s claim instead of their spectra. The result? A batch contaminated with 2,4-difluoroaniline. That lesson stuck.
Solubility also turns up during method development. Water or alcohol solubility gives hints about any lurking moisture or secondary amines, and color can signal trouble from oxidation or leftover iron. Sticking to a colorless or faintly yellow product means the purification has done its job. Regulatory specs cut in too, especially if you’re heading for anything related to pharma or electronics. Endotoxin levels or heavy metals aren’t just paperwork—they can trigger failed validations and recalls.
Trust and Verification: Choosing the Right Supply
Experienced researchers learn to ask for certificates of analysis, often comparing lots or cross-checking suppliers. Fresh chemists sometimes skip this step, and I’ve seen projects veer off because someone skimped on this basic check. The whole idea is to keep the experiment, and the data, clean. People in industry might swap notes on which supplier batches pass HPLC with flying colors or which ones tend to leave sticky residues in columns. These insights don’t make it to glossy catalogs, but they drive real decisions in choosing a source.
Ways To Cut Down Risk
I’ve seen a few solutions work over the years. If possible, ordering in smaller amounts—and testing each lot—saves headaches. Storing chemicals dry, away from light, and frequently re-checking the key specs stops surprises down the line. Some labs pool orders or request pre-shipment samples to test before committing to a bulk purchase; this goes a long way in high-stakes projects.
In the end, keeping purity high isn’t just about ticking boxes on a specification sheet. It’s about driving confidence in every step after that bottle gets opened. Cutting corners up front only hurts downstream. As someone who has scrubbed failed product from reactors and puzzled over bad TLCs, I’d say the value of knowing exactly what you have is worth every bit of extra care.
2,3,4-Trifluoroaniline might not be a household name, but anyone who’s handled chemicals in a lab or warehouse knows how crucial it is to treat it with respect. From the sharp smell to its potential health risks, this compound can disrupt much more than a routine day at work. Years spent around chemical storage rooms taught me that small oversights quickly turn into big headaches. The question isn’t about whether risk exists—it’s about staying ahead of it, every single step from storage shelf to delivery truck.
Averting Trouble With Simple Choices
Chemicals like 2,3,4-Trifluoroaniline demand careful handling. This stuff carries dangers that go beyond dry safety sheets: direct skin contact or a carelessly vented container can cause immediate health effects, including skin and respiratory irritation. So, the most basic requirement—don’t leave it out in open air, and don’t trust flimsy packaging. Store it in tightly sealed containers. Good chemical compatibility prevents surprise reactions. Once, after a poorly-chosen replacement drum led to corrosion and a minor spill, I learned that lesson the hard way.
Strong fumes alone convince most people not to stash compounds like this near workstations or food prep areas. Instead, aim for dry, well-ventilated, cool spaces that don’t see sunlight or wild shifts in temperature. Temperature swings can degrade the chemical, cause pressure build-up, or warp containers. I’ve seen labels fade and cap seals give out when companies cut corners here.
Keep Storage Spaces Honest
Physical separation of incompatible chemicals matters just as much. Storing 2,3,4-Trifluoroaniline near oxidizers, acids, or even plain old cleaning bleach is asking for trouble, sometimes in ways that become clear only during crisis cleanup. Paint clear lines on storage shelves or rooms, use locked cabinets, and never rely on memory or sticky notes for separation. A disaster I witnessed began with a simple misplaced container; the fallout included an entire building evacuation and weeks lost to investigation.
Safe storage includes honest, legible labeling. Those scribbled signs or fading stickers have no place near chemicals with toxic or flammable potential. Legally required documentation—Safety Data Sheets, access logs, and inventory lists—must be within arm’s reach, not buried in a back office. This isn’t just paperwork. Emergency responders and new hires both rely on these files in a bind.
Transport That Takes People Seriously
Getting 2,3,4-Trifluoroaniline from point A to B raises its own set of stakes. Secure packaging keeps leaks at bay, but smart transporters know this isn’t the end of the story. Use drums or bottles rated for hazardous substances, complete with intact, compliant seals. Every company should train drivers and handlers not just to move packages but to identify damage or warning signs before and during the trip. A single overlooked leak—especially in hot, vibrating conditions—can put entire neighborhoods at risk.
Trucks carrying toxic cargo deserve clear signage, with placards visible from every angle. Rules on what can travel together apply as much to trucks as storage rooms—never mix this chemical with foodstuffs or flammables in the same vehicle. Tracking and logging who touched each shipment and when reinforces accountability. I value companies that enforce routine inspections before departure, not just after an incident.
No Shortcuts—Ever
Responsible storage and transport for chemicals such as 2,3,4-Trifluoroaniline come down to putting prevention above convenience. The costs of skipping checks—ruined stock, sick workers, legal fallout—always outweigh five minutes saved. Asking tough questions, maintaining clear policies, and investing in safe rooms and robust packaging pays dividends, both in regulatory compliance and human health. If there’s one thing experience taught me, it’s that doing things by the book doesn’t slow you down; it keeps you moving forward without regret.