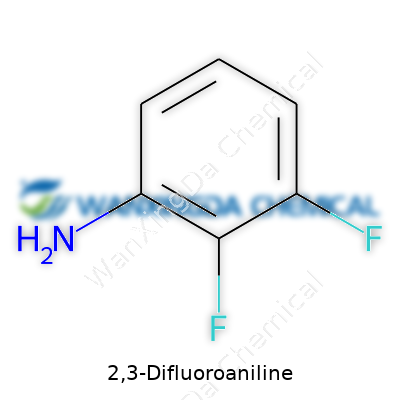
The Science and Significance of 2,3-Difluoroaniline
Historical Development
Fluorine chemistry reshaped many parts of modern science. The discovery and use of 2,3-difluoroaniline track alongside major leaps in organic synthesis across the last century. Synthetic chemists first saw the potential of fluoroanilines when new substitution techniques for aromatic rings appeared. With the rise of pharmaceutical chemistry and agricultural science in the mid-1900s, interest in small, versatile haloaromatics grew sharply. Scientists wanted new tools for building up biologically active small molecules, and the difluorinated aniline structure fit the bill. Pioneering journals from that period carried landmark studies on selective fluorination, which stood out for their ingenuity and precise placement of fluorine atoms on benzene rings. Today’s methods benefit from decades of clever refinements that allow for safer, scalable access to these once-exotic building blocks. This background matters: the roots of 2,3-difluoroaniline's story show its emergence was tied closely to the creative drive to probe new frontiers in chemistry.
Product Overview
2,3-Difluoroaniline offers a blend of reactivity and stability. In my experience working with halogenated aromatics, chemists often lean toward this compound for its unique dual-electron-withdrawing effect, which tweaks the reactivity of the amino group without shutting it down. The presence of two fluorine atoms next to the amine changes outcomes in N-arylation, cross-coupling, and further substitutions—options unavailable with non-fluorinated anilines. People value this for more than synthetic reasons: it brings new properties to final products, which means a compound’s pharmacokinetics, metabolic profile, or agrochemical activity can shift in useful directions. The drive to explore these tweaks fuels the steady demand in research labs and specialist manufacturers alike.
Physical & Chemical Properties
The small shifts that two fluorine atoms create never fail to surprise first-time users. 2,3-Difluoroaniline does not behave quite like its single-fluorine relatives. It appears as a colorless to light yellow liquid, which brings a distinctly pungent odor into the laboratory. Its boiling and melting points land lower than simple aniline, owing to fluorination’s effects on intermolecular interactions. You’ll often find it resists oxidation better than electron-rich analogs, which helps during storage and shipping. The molecule’s solubility skews toward organic solvents like dichloromethane and acetonitrile. Acids and bases interact with the amine as expected, but the electron-withdrawing ring means some standard electrophilic substitutions prove trickier. Skip careless assumptions about reactivity: every small tweak to the ring counts in synthetic planning.
Technical Specifications & Labeling
Of all the specialty chemicals I’ve handled, fluorinated aromatics demand hawk-eyed attention to purity and contaminants. Labs measure water, halide, and oxidizable impurities because even small traces can shift catalytic or coupling reactions. Commercial samples rarely stray far from the industry’s purity benchmarks, but stricter specs—such as GC over 98%—usually show up in pharma and fine chemical applications. Labelling often highlights these metrics alongside hazard and transport classifications, reflecting a recognition of both the chemical’s potential and the need to avoid mishaps. Proper labelling is not bureaucracy; it keeps researchers from unwittingly stumbling into reactive surprises or exposure.
Preparation Method
Preparative routes for 2,3-difluoroaniline reflect both the evolution of fluorination chemistry and the constant push for efficiency. Classic methods start from nitrobenzenes, which chemists nitrated and then subjected to halogen exchange or direct fluorination with reagents such as Selectfluor or DAST. In my work, handling these reactions means balancing yield against safety—the wrong protocol brings corrosion and volatility risks. More recent improvements see safer nucleophilic routes from 2,3-dihalonitrobenzenes or protected intermediates, with hydrogenation steps following to introduce the amino group cleanly. Large-scale manufacturers prize these upgrades both for safety and cleaner profiles, which decrease the cost and mess of purification. Watching the process evolve in patents and journals creates a map of chemists wrestling with real constraints, not just theoretical yields.
Chemical Reactions & Modifications
What sets 2,3-difluoroaniline apart is its flexibility as a synthetic handle. The electron-deficient ring resists overreaction and supports functionalizations at both the amine and aromatic core. Buchwald–Hartwig aminations, Suzuki–Miyaura couplings, and SNAr substitutions all draw on this framework, giving access to a swath of new molecules that lead out into medicinal chemistry, advanced materials, and even dendritic polymers. My time in medicinal chemistry highlighted the value of such “tunable” intermediates—adding or swapping functional groups downstream turns a single bottle of 2,3-difluoroaniline into a series of diverse lead compounds or key reaction partners. Modifications to the amine bring in acylation, sulfonation, or alkylation steps, while the fluoro groups themselves open doors to further nucleophilic substitution under basic conditions or activation for cross-coupling catalysis.
Synonyms & Product Names
Language in the chemical industry can trip up even experienced hands. 2,3-Difluoroaniline goes by names like o,o-difluoroaniline, 2,3-difluorobenzenamine, and CAS Number 367-25-9. These synonyms show up in procurement, catalogues, and databases—making clarity vital. In fast-paced communication, missing a digit or a prefix can slow down sourcing and delay results. I’ve personally seen project timelines stall over an ambiguous supplier code or a mix-up between isomeric products. Adopting consistent, accurate nomenclature is less about pedantry than keeping projects on track safely.
Safety & Operational Standards
Halogenated anilines bring real hazards. 2,3-Difluoroaniline can irritate the skin, eyes, and respiratory tract, and careless handling leads to serious incidents. The pungent odor first signals you’ve breached containment, but you miss it with low-level exposure. Anyone working with this chemical needs ventilation, gloves, and splash protection—no excuses. The colorless liquid may not look dangerous, yet accidental exposures accumulate and lead to chronic impacts. Proper storage in sealed, clearly labeled containers under cool, dry conditions heads off most incidents. In scaling up, engineer controls like scrubber systems and remote loading cut down the risk of inhalation or fire. Emergency procedures gain urgency—spill kits, showers, and mask fit testing underline this isn’t a compound for corners to be cut. Every research group handles “hazard fatigue,” but reminders from safety teams and incident statistics show why tight operational discipline remains critical.
Application Area
Pharmaceuticals, agrochemicals, and specialty materials all harness the unique features of 2,3-difluoroaniline. Medicinal chemists chase fluoro-substituted scaffolds for their ability to block unwanted metabolism and shape molecular interactions. Anti-infective and anti-inflammatory drugs built on this base show differences in both activity and side effect profiles when compared with non-fluorinated analogs. Custom pesticides and herbicides draw on similar logic: subtle changes in molecular shape and bioavailability favor persistent, selective action. In electronics, researchers work these structures into liquid crystals and high-performance polymers, leveraging fluorine’s impact on glass transition temperatures and dielectric properties. I’ve watched how even small amounts mixed into commercial products enable new device architectures or extend product lifetimes. The compound’s reach stems from its dual role: building block and property-modifying agent.
Research & Development
In research circles, 2,3-difluoroaniline is never simply “another aniline.” Academic groups and industry teams explore new coupling partners, regioselective reactions, green fluorination methods, and biologically active derivatives. Recent publications indicate a surge in metal-catalyzed processes—palladium and nickel systems, in particular—aimed at cleaner, higher-yield syntheses. Computational work models the electron density and predicts novel reactivity patterns, while bioassay panels sort promising medicinal leads from the pack. Partnerships between process chemists and regulatory scientists focus on scaling up routes that cut waste, use benign reagents, and simplify downstream purification. Patents reflect this drive: a single year often brings a handful of new approaches, each refining yield, safety, or application fit. The pace of change outstrips textbooks, and depends on real-world problem-solving, not just rote methodology.
Toxicity Research
Nobody in chemical safety takes fluorinated anilines lightly. Toxicologists flag 2,3-difluoroaniline for its potential risk to human health and the environment. Direct exposure can lead to methemoglobinemia—a blood disorder—alongside eye, skin, and lung irritation. Environmental chemists look at persistence and bioaccumulation: fluorinated aromatics tend to resist breakdown, and their movement through soil and water can lead to unexpected exposure routes. Risk assessments balance workplace exposure thresholds with environmental load. In my own work, meeting environmental standards often means limiting waste generation at the source, bundling unused material for controlled destruction, and careful monitoring of wastewater streams. More recent toxicity screenings aim to understand chronic impacts, subtle metabolic breakdown, and ecological effects. Transparency in reporting remains key, since underestimating fluorinated compound hazards never ends well.
Future Prospects
The appetite for fluoroaromatics continues to grow. As medicinal, material, and agrochemical sciences chase ever-tighter performance requirements, 2,3-difluoroaniline’s role as a modular starting point stays secure. Prospects point toward greener, more sustainable synthesis: direct C–H fluorinations, biocatalytic routes, and room-temperature processes all beckon as chemists seek to cut waste and cost. Regulatory attention on persistent pollutants may reshape permissible use and prompt new degradable versions. Research teams explore the interface between organically bound fluorine and biological systems, hoping to unlock selective, non-toxic behaviors or targeted delivery in medicine. A rising generation of chemists learns not just fluorination, but responsible practice, and the market grows accordingly. Two decades of innovation built the groundwork, but each new insight—whether in the lab or in the field—adds another layer to the promise and challenge of this deceptively simple molecule.
Breaking Down 2,3-Difluoroaniline
Curiosity drives much of chemistry, and 2,3-Difluoroaniline is a good example of where simple questions lead to deeper insights. On paper, its chemical formula reads as C6H5F2N. Some folks might picture complex glassware and mysterious colored liquids, but all we're talking about is a benzene ring holding onto two fluorine atoms—one on the second carbon, the other on the third—plus an amino group (NH2). That’s what shapes the formula.
A compound like this isn’t just a random product of the chemistry world. I remember my early days in the lab, learning how each little change on the ring makes huge ripples in how a chemical behaves. Add one fluorine, and the whole interaction with other molecules can tilt in a new direction. Add two, in just the right spots, and you’ve carved out a special path.
Why the Structure Matters for Real Life
Compounds with similar structures show up in plenty of places—especially pharmaceuticals and agrochemicals. Fluorine makes drugs more stable, more effective, and sometimes more easily absorbed. Amino groups play their own role in connecting building blocks and helping drugs sneak across the cell boundary. I’ve seen chemists in drug design comb through pages of molecular structures searching for just the right substitution. For some companies, tweaking a spot from one position to another slides a molecule from “promising” to “blockbuster.”
Why not just guess? Each position changes binding, activity, and safety. Still, the journey from formula to function takes more than lab work. Good science ties in trustworthy research, ethical oversight, and a practical approach to health and the environment. Scientists and regulators both need to know exactly what these formulas mean, not because the letters themselves matter, but because they translate to impacts in the real world—on people, crops, or water tables.
Addressing Concerns and Moving Forward
There’s a growing conversation these days about chemical safety and sustainable practices. In some places, strong opinions reign about how chemicals like these enter the environment after use. The presence of fluorine or other halogens can sometimes slow breakdown, raising concerns over persistence. The better we understand a molecule’s structure and properties, the more informed those conversations become.
Chemists need to keep clear records, share data openly, and stay alert to new research. Universities and industry groups, in my experience, do well to promote continuing education, making sure newcomers and old hands alike understand how something like “2,3-difluoroaniline” fits into bigger frameworks—food safety, medicine, even manufacturing quality checks.
Solutions don’t come from one direction. Education, innovation, and responsibility all play roles. We need more open access to chemical data, smarter regulation that’s informed by ongoing science, and conscientious industry approaches. Careful study of something as simple as a chemical formula helps fuel all that work—and can shape outcomes that reach far beyond the lab.
Chemistry on the Ground
Walk into any modern lab, and it’s easy to see how chemistry shapes the world. Every bottle and beaker holds a story of trial, error, and innovation. 2,3-Difluoroaniline doesn’t grab headlines, yet its role in chemical development runs deep. Years in scientific research have taught me that some compounds, though they sound obscure, keep the wheels of everyday life spinning. This one falls solidly in that camp.
Building Blocks for Serious Work
The nitty-gritty of 2,3-Difluoroaniline lies with its structure. Those two fluorine atoms attached to the benzene ring change the game compared to plain old aniline. In pharmaceutical labs, chemists count on molecules like these to construct new drug candidates. Drug design doesn’t get very far without starting points that bring stability, reactivity, and fine-tuned biological effects. Fluorinated building blocks like 2,3-Difluoroaniline add value by making molecules more resistant to breaking down in the body. Add a little fluorine, and often a pill lasts longer in the bloodstream, with fewer side effects. It isn’t just theory—it shows up in blockbuster drugs where tiny tweaks make all the difference in how they treat disease.
Pushing the Envelope in Agrochemicals
Farmers count on chemistry to protect crops and secure healthy yields. Many of the best-known pesticides and herbicides trace their origins to molecules much like 2,3-Difluoroaniline. Its backbone slots into larger molecules that target pests but leave the crop standing tall. The fluorine atoms can increase the activity against pests and extend how long a treatment sticks around, so a single spray does the job. This eases the pressure on farmers—less work, better harvest. It’s not rare to find compounds based on this structure in patent lists for modern agrochemicals.
Materials Innovation: Beyond the Obvious
It’s tempting to focus only on medicine and farming. The story doesn’t stop there. In electronics and specialty materials, the search for thermal and chemical resistance leads researchers to compounds like 2,3-Difluoroaniline. Its fluorinated ring can anchor bigger molecules that end up in polymers or coatings. These materials might find a place in devices that get hot or face exposure to tough chemicals—think smartphone insides or circuit boards. My time working alongside polymer chemists made it clear: a single molecular tweak can mean the difference between a product that melts in the sun and one that survives a heatwave.
Facing Challenges Head-On
With its benefits, the flip side needs attention. The introduction of new building blocks sometimes raises questions about environmental impact. Fluorinated molecules stick around in nature. Researchers must stay alert to where these compounds end up after use—especially outside the controlled lab setting. Green chemistry could hold the key. Approaches that avoid waste, cut out hazardous reagents, or use renewable sources might offer new pathways to safer production and disposal. Companies and universities have joined forces to find smarter methods for making and breaking down fluorinated compounds like this one.
Looking Down the Road
The world of 2,3-Difluoroaniline may not make the evening news, but its influence ripples through many corners of industry. Scientists need to keep asking the tough questions—how can these building blocks serve medicine, agriculture, and technology, while also respecting the environment and community health? From lab bench to final product, thoughtful use of chemical innovations remains the only way forward.
Getting It Right from the Start
2,3-Difluoroaniline might sound like something tucked away in a chemist's notebook, but it deserves more attention from anyone working with chemicals. I once watched a rookie in a small lab throw caution aside and cut corners with a similar aromatic amine—just a few months later, he dealt with way more headaches than he’d bargained for. Let’s put aside the jargon. Safe handling and smart storage for this compound are not up for debate.
The Real Risks
I learned a lot in a university laboratory, where we worked with different anilines almost daily. Most folks underestimate how easily aromatic amines react, especially those with halogen substitutions like fluorine. 2,3-Difluoroaniline gives off vapors that irritate the eyes, nose, and throat. Accidental skin contact usually leads to a burning sensation or rash, and constant low-level exposure builds up over time—enough to compromise health. Strong ventilation means more protection than a cracked window.
Moisture and sunlight chip away at most organofluorides, degrading purity or, even worse, forming unpredictable byproducts. From what I’ve seen, the rule among seasoned chemists is: keep it dry, cool, and dark. A locked, local chemical cabinet—away from acids and oxidizers—keeps things under control. Walking into a cluttered storage room, I’ve seen the aftermath of one overlooked bottle left too close to a heat source: warped labels, weird smells, and a mess to clean up. It’s never just a small mistake.
What Good Storage Looks Like
Rather than glass beakers balanced on a shelf, chemical-resistant containers with tight seals limit leaks and evaporation. Everybody in the lab should check dates and inspect containers for cracks or swelling regularly. Every misplaced cap or poorly wiped rim opens the door for problems. Once, our team had to halt an entire project because one mix-up led to cross-contamination. Equipment, records, and hours of effort gone in the blink of an eye—all because the storage system had a weak link.
Getting the Culture Right
Most folks put too much faith in warning labels and not enough in hands-on training. I’ve seen the right attitude turn a group of green interns into a reliable team who kept each other honest. Refreshing training every few months helps. Facts from agencies like OSHA and the CDC back it up: vigilance pays off. Clear areas, working spill kits, and no food or drink anywhere near the workspace. These steps build habits strong enough to cope with unexpected slip-ups.
Looking for Solutions
Packed schedules tempt people to take shortcuts, but the best labs make time for double-checks. Color-coded storage works better than any memo for separating incompatible chemicals. Digital tracking of inventory and condition helps catch issues early. Some companies use automated storage with climate control—an investment upfront saves money and trouble down the road. In my own experience, putting a few policies in plain English, alongside a crew that cares, outperforms a thick book of rules nobody reads.
At the end of the day, the way we store and handle 2,3-Difluoroaniline reflects how much we value safety, time, and each other’s health. Mistakes happen, but smart setups and a disciplined culture make them a lot less likely, and the science better for everyone.
Why Knowing What’s Behind the Chemical Label Matters
Chemicals with unassuming names sometimes carry surprising risks. I’ve spent enough time around labs and industrial settings to see how easily everyday work can go sideways when hazardous substances aren’t respected. 2,3-Difluoroaniline—a chemical used in dye manufacturing, pharmaceuticals, and specialty materials—demands more caution than many expect. No one wants to star in a workplace incident report, so let’s take a direct look at what makes this chemical a real safety concern.
The Risks You Might Not See at First Glance
Handling any aniline derivative calls for care, but add fluorine atoms and the stakes rise. 2,3-Difluoroaniline can release toxic fumes if heated or burned. I’ve watched the aftermath of a chemical fire—the damage lingers long after the flames go out. Workers exposed to aniline compounds have reported everything from headaches to more severe complications. Skin contact can mean persistent irritation, and breathing in fumes or dust may leave someone lightheaded or nauseous. Exposure to high levels of aniline compounds over time can affect the blood’s ability to carry oxygen, leading to cyanosis or worse, and symptoms may arrive slowly enough that people don’t notice until it’s serious.
For folks formulating new products or scaling up reactions, it’s tempting to lean on gloves and goggles and call it a day. That isn’t enough. Spills soak through the wrong gloves or splash onto unexpected surfaces, and regular nitrile gloves may not hold up for long. Chemical-resistant aprons and face shields help, but only if everyone actually uses them and keeps their protective gear in good shape.
Small Mistakes, Big Consequences
It’s easy to underestimate how quickly fumes can spread. Poor ventilation in a fume hood or workspace can move hazardous vapors throughout a building. I once saw a team’s afternoon sidetracked by a poorly contained solvent spill, forcing an evacuation and hazmat team visit. Cleanups never go as quickly as people hope in those moments.
Another big risk: storing 2,3-Difluoroaniline with incompatible chemicals. Strong oxidizers or acids can trigger reactions that create more toxic gases. Flammables stored in the wrong place contribute to fire hazards. Even something as simple as a cracked label or missing safety data sheet leaves workers guessing instead of responding with confidence.
Better Habits, Safer Workspaces
Training matters most. People pay attention when real examples get used—no one wants to repeat a colleague’s painful lesson. Regular refresher courses on chemical safety, spill response, and emergency protocols can make the difference between a scare and a tragedy. Every workplace benefits from clear signage, accessible eyewash stations, and proper chemical storage according to up-to-date safety guidelines.
Facilities can invest in better ventilation, regular safety drills, and personal protective gear rated for chemicals like 2,3-Difluoroaniline. For smaller operations, even a tight checklist and good communication can help avoid disaster. As soon as anyone feels weird after a possible exposure, seeking medical advice quickly makes a real difference.
Care and respect for what’s in the bottle won’t make headlines. Quiet preparation—training, protective gear, checking storage—makes sure no one gets famous for the wrong reasons.
Striking the Balance Between Research and Production
Anyone who has spent time in a lab or on a plant floor learns quickly that a chemical isn’t just a chemical. It’s not enough to ask if you have 2,3-difluoroaniline in stock; the real question is what grade you’ve got and whether it fits the task. In the case of 2,3-difluoroaniline, those variations in purity turn into a sticking point that decides everything from a reaction's yield to the cleanliness of your final compound.
In research, even a trace impurity can mess with your data or produce a result that looks good on paper but unravels under scrutiny. Organic chemists depend on high-purity starting materials. I remember a colleague spending weeks troubleshooting an unexpected byproduct, only to discover that the “technical grade” starting material was laced with just enough contaminant to throw the whole process off. It hit home how much headaches can be spared by paying attention to what’s inside the bottle before it hits the flask.
Differences Between Purity Grades
Purity isn’t just a number on a label. Lower-purity material can work for some industrial processes, especially if the impurities won’t make a difference in the final product. In basic screening or process optimization, researchers sometimes compromise, picking up less expensive grades that carry more baggage. But if your work heads for pharmaceuticals, electronics, or agrochemicals, that extra 1-2% of unknowns in a lower grade batch can threaten product safety or regulatory approval.
Labs and manufacturers that demand tight control go for higher-purity 2,3-difluoroaniline, often over 98% or even 99%. That choice isn’t about looking fancy—it can mean the difference between reproducible results and frustrating reruns. In the pharmaceutical world, regulators like the FDA expect all starting materials to pass high standards for quality and traceability. A shortcut with an impure precursor is the kind of risk no responsible company wants to take.
Quality Assurance: More Than Just a Specification
Anybody in charge of buying chemicals has a tough job. Budgets can be tight, and it’s tempting to grab whatever fits the spreadsheet. The trouble appears when subtle impurities in 2,3-difluoroaniline lead to new reaction pathways or carry through to the end product. I’ve seen production runs ruined and deadlines missed for want of lab-grade material, turning what seemed like a good deal into a costly lesson.
Quality doesn’t stop at purity. Reputable suppliers run their materials through tests like NMR and HPLC, then supply certificates of analysis that give a real picture of what’s in each drum or flask. Transparency is crucial, especially as supply chains become more complex and regulation ramps up. Labs and companies who tie themselves to reliable sources—ones willing to put their specs in writing—stay out of trouble more often.
Searching for Solutions Beyond the Label
For those just starting out or running small operations, pooling resources with trusted labs or negotiating for larger, batch-tested quantities helps keep costs down. Communication between procurement and R&D makes sure everyone stays realistic about when top-shelf purity is a necessity and where it’s possible to save.
I’ve learned, sometimes the hard way, that guessing at purity or hoping a lower grade will “do the job” just brings setbacks. In the real world, getting 2,3-difluoroaniline in the right purity for the purpose isn’t a luxury—it’s what keeps processes running smoothly and reputations intact. If more folks in the supply chain shared their struggles and strategies, maybe others could avoid a few missteps along the way.