2,3-Difluorophenol: A Commentary on Chemistry, Industry, and Opportunity
The Historical Path to 2,3-Difluorophenol
Few chemical stories run straight, especially with specialized fluorinated aromatics like 2,3-difluorophenol. The journey starts with the love affair chemistry has had with fluorine. Back when fluorinated compounds were exotic, research teams chased after phenols with unique substitution patterns to understand reactivity and expand the toolbox for making agrochemicals, medicines, and advanced materials. In university and industrial research libraries, you can track early syntheses to the postwar years, as halogenated phenols grabbed attention for their role in biological studies. Fast forward, this target joined the shelf as a key intermediate. The industry soon leaned on it, mostly because those two fluorines positioned next to the hydroxyl group lend the ring new physical quirks and chemical temperaments.
Getting Acquainted: What 2,3-Difluorophenol Brings to the Table
Describing 2,3-difluorophenol starts with the way it disrupts expectations. The two fluorines at the 2 and 3 positions coax out both lipophilicity and subtle electronic differences compared to plain phenol. It has a faintly sweet, sharp aroma, and its appearance doesn’t shout — clear liquid, nothing flashy — but chemists love the way this arrangement tweaks reactivity and unlocks new routes in both lab work and manufacturing. Folk who deal with delicate syntheses or design bioactive molecules see it as a reliable special effect in their toolkit, not a headline-grabber but an unsung hero.
Physical and Chemical Nature: Under the Lens
Working with 2,3-difluorophenol means paying attention to more than just the number of carbons or the presence of a hydroxyl. The molecule's boiling point sits lower than generic phenol, influenced by the electron-hungry fluorines tightening intramolecular hydrogen bonding and lowering volatility. Solubility and acidity both see a nudge; the pKa drops thanks to electron-withdrawing effects, which can push a reaction into a sweet spot that unmixed phenol leaves untouched. Its moderate volatility and flammable nature draw regulatory flags and require sturdy standard operating procedures in labs and pilot plants. The safety profile isn't as rough as with the heavier halogenated cousins, but gloves and ventilation still go hand in hand with every beaker poured.
Technical Specs and Real-World Labels
Open a chemical catalog, and 2,3-difluorophenol shows up by its CAS designation, sometimes by the shorthand DFP-23 or by longhand variants echoing its preferred IUPAC naming. Most shipments come stabilized, with purity levels that reach well over 98 percent for R&D and process development work. Labels spell out flash points and storage cautions clearly, speaking from experience—loose storage can spell gradual degradation, unpleasant surprises in yield, or purity headaches down the line. Labels on bulk containers double down on international symbols for toxicity and flammability, not just to check regulatory boxes but to remind anyone grabbing it from the shelf not to cut corners.
How Chemists Make 2,3-Difluorophenol
Making this compound usually kicks off with a precursor that carries the right halogen pattern, like 2,3-difluoronitrobenzene. Experienced chemists go for nucleophilic aromatic substitution, favoring routes where the nitro group leaves and the hydroxyl steps in cleanly. One thing I've seen in scale-ups is how a little water management in these reactions saves hours of cleanup, since uncontrolled water can hydrolyze more than intended. Once formed, the crude is typically distilled under reduced pressure to avoid charring and loss of material. Some labs push for greener chemistry with milder fluorinating agents or alternative hydroxylation, but most big players stick to the well-trodden, reproducible methods.
Reactivity and Chemical Mission-Creep
2,3-Difluorophenol doesn't just stay on the shelf waiting for one use. It plays an active role in constructing more complicated molecules, especially through etherification, esterification, or Suzuki-Miyaura cross-couplings. The electron-thirsty fluorines block some substitutions but open doors for others, making the molecule both a challenge and an opportunity for creative synthetic minds. Its use in building rung after rung of agrochemical scaffolds comes up again and again in literature and patents. By tuning electronic properties, chemists adjust the biological profile of molecules, sometimes chasing more selective pesticides or less environmentally persistent active ingredients.
More Than One Name: Synonyms and Aliases
Depending on a chemist’s background or the country of origin, 2,3-difluorophenol parades a few alternate titles. You’ll hear "m-difluorophenol," "2,3-DFP," and sometimes a trade name popping up in synthesis catalogues. These aliases track with the product’s migration into larger industry circles, where unconventional abbreviations sometimes end up on logistics manifests or import paperwork. Those working international supply chains know how important it is to double-check these terms, so product doesn't get held up at customs or swapped for a similar-sounding cousin.
Staying Safe and Getting Things Done
Good chemists know process safety doesn’t stop at checking boxes. Handling 2,3-difluorophenol draws on lessons learned from decades of hazardous molecule management. Fume hoods, proper gloves, flame arrestors, and spill kits aren’t optional. I've seen firsthand how a casual attitude toward handling leads to regrettable incidents, even with a moderate-toxicity chemical like this. Labs committed to EHS protocols make it clear that continuous monitoring is essential, especially in closed systems where vapors could build up. Responsible operations invest in comprehensive training and periodic review, not as overkill but as necessary day-to-day insurance. Regular audits, labeling reviews, and input from occupational health experts keep standards current, reflecting new scientific knowledge rather than being stuck in the past.
Areas Where 2,3-Difluorophenol Makes a Mark
Pharmaceutical research puts this molecule front and center, especially in early drug development and scaffold modification. The distinct electronics let medicinal chemists manipulate receptor binding or tweak metabolic stability. Crop science also leans on this compound—those two fluorines introduce pest resistance or weatherproof desired biological properties. Material scientists look at it for niche polymer synthesis, where unusual electronic features give rise to new plastic or resin behaviors. The compound rarely grabs the spotlight by name, but it’s hiding in plain sight inside molecules that do draw headlines. As a result, those of us in R&D quickly learn that access and purity of 2,3-difluorophenol can make or break timelines for product launches or patent submissions.
Current Research and Scientific Curiosity
Research streams exploring 2,3-difluorophenol don’t limit themselves to synthetic convenience. Teams investigate its use in late-stage functionalization, chasing after more sustainable or selective processes with less environmental loadout. I keep seeing new publications on transition-metal-catalyzed couplings and direct activation using this scaffold, hinting at broader utility than traditional substitution would predict. Combinatorial chemists value it as a building block for generating small-molecule libraries with diverse bioactivity, aligning with current interest in precision medicine and next-generation crop protection.
Understanding Toxicity and Human Interaction
Most published research pegs the compound’s toxicity as low to moderate, with levels dictated by both the phenolic group and added fluorines. Chronic exposure is not well studied; short-term effects line up with generic phenol toxicity—skin and eye irritation, respiratory discomfort at high vapor concentrations. Animal studies point to manageable acute toxicity, but regulators push for conservative thresholds to cover knowledge gaps. Those handling the material day-to-day learn to treat it with respect, not only to protect personal health but to shield co-workers and downstream users. Toxicology evolves as new endpoints are discovered, and there’s an ongoing call for well-funded, independent research into long-term environmental and biological impacts.
What Lies Ahead: Looking to the Future
2,3-Difluorophenol stands poised for more than incremental improvement. Green chemistry initiatives have started to knock on its door, with researchers rethinking both starting materials and byproduct management for cleaner synthesis. As genomics enables smarter design of agrochemicals and therapies, this compound will anchor new generations of molecules with faster development cycles. Expect continued advances in catalysis and selective reactivity, as well as pressure for better recycling or disposal solutions. Industry trends point to an era where specialization and customization call the shots, and versatile building blocks like this one will stand to benefit. In my experience, chemicals that blend established performance with potential for innovation don’t disappear—they simply pop up in the next big breakthrough.
Why Knowing Chemical Formulas Matters
Growing up around a family who spent time in various labs—everything from research at the university to testing formulations at home—I saw how chemical formulas guided every step. The formula for a compound tells you not only what it is, but also serves as your map for predicting reactions, ensuring safety, and pushing the boundaries of science. Today’s story about 2,3-Difluorophenol brings that lesson home once again.
A Look at 2,3-Difluorophenol’s Structure
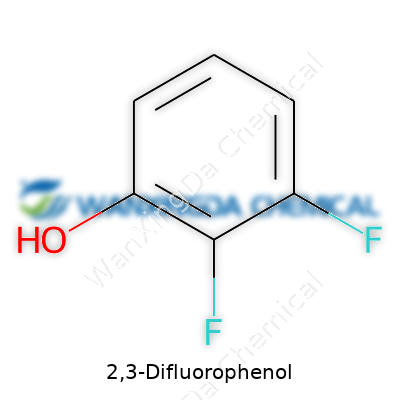
2,3-Difluorophenol has the formula C6H4F2O. This means there are six carbon atoms, four hydrogens, two fluorines, and one oxygen sitting on a phenol ring, with fluorines sitting at the 2 and 3 positions. This might look like a puzzle, but the arrangement is vital. Add or move a single atom on the ring and you get a compound with different properties, different risks, and different uses. The chemistry world is full of stories about researchers who mixed up similar compounds—sometimes with dangerous results. That’s why keeping the formula straight is not a detail to skip.
Potential Hazards and Handling
Fluorinated phenols have attracted research for good reason. Fluorine is a tricky element—hard to tame, highly electronegative, and known to tweak the toxicity and reactivity of whatever it touches. For instance, in the pesticide industry, just a small change from phenol to difluorophenol shifts environmental persistence and biological effects. If someone accidentally substitutes a similar-sounding compound for 2,3-Difluorophenol, the outcome can become a serious safety issue.
Back in my own days working with phenolic compounds, I learned to double-check labels and formulas, especially if the sample sat on the shelf for a few months. It only takes a mix-up once for everyone to appreciate the role of accurate documentation.
How the Formula Connects to Purpose
Chemists use 2,3-Difluorophenol as a building block, especially in designing pharmaceuticals and specialty materials. Fluorine atoms don’t just sit quietly; they can help drugs stay active longer or help plastic resist breakdown. Companies look for these specific compounds when developing new treatments or high-performance products, and knowing the formula means they know exactly what to order, what to test, and what to expect.
Research shows that introducing fluorine at specific sites on a phenol ring (like 2 and 3) can change everything from melting point to the way the molecule binds in biological systems. No shortcut exists—if you get the formula wrong, the new drug or material could perform wrong too.
Promoting Clear Chemical Communication
Every science student hears stories about disasters caused by confusion in the lab, but those stories stick because they’re true. Chemistry, more than many fields, rewards precision. A formula like C6H4F2O isn’t just a string of symbols; it keeps us safe, advances research, and opens the door to new discoveries.
2024 has seen chemistry taking center stage, from environmental monitoring to designing new medicines. All these advances still come back to understanding, documenting, and sharing accurate chemical formulas. That’s where progress starts.
Looking Deeper into Its Real-World Role
Every day, a stack of chemicals hides behind the scenes, working in silence to shape the medical and technological landscape. 2,3-Difluorophenol rarely appears in the spotlight, but its impact reaches beyond just being a chemical formula in a dusty lab. From my own experience following trends in pharmaceutical chemistry, I’ve seen how molecules like this quietly move the needle on innovation.
Building Blocks in Pharmaceutical Development
Drug research moves fast, and it leans hard on reliable building blocks. Chemists tinker with the arrangement of atoms, hoping to create a better medicine or treatment. 2,3-Difluorophenol carries a set of fluorine atoms in its structure, making it valuable when researchers modify compounds to fine-tune properties. Fluorinated aromatics like this often help increase the biological stability of new drug candidates. One detail I learned chatting with medicinal chemists is that adding fluorines can change how drugs behave in the body. Sometimes, it helps the new drug resist breakdown just enough to do its job longer without extra side effects.
In practice, 2,3-Difluorophenol sees use mostly as an intermediate during the early stages of drug synthesis. It's rarely the star of the show, but its role shapes how the final product turns out. In a world where drug resistance, metabolism, and safety remain challenges, every small tweak in a molecule’s structure can have a big impact.
Creating Advanced Materials and Agrochemicals
It’s not just human medicine that borrows tools from the organic chemistry toolbox. Crop science and materials research also use 2,3-Difluorophenol. Companies searching for new herbicides, fungicides, or specialty polymers add this compound to their arsenal. The unique chemistry of fluorine helps create materials that last longer or protect crops more effectively against today's pests. Over the years, I’ve watched research shift from brute-force chemical formulas to smart, targeted synthesis using building blocks like this, trying to balance impact on the environment with productivity.
Challenges with Safety and Sustainability
Working with any fluorinated compound comes with real-world headaches—cost, handling risks, and future disposal. 2,3-Difluorophenol is no exception. People deserve transparency about how these chemicals move from laboratory bench to production floor, and into the wider world. For all its usefulness, the larger challenge remains figuring out how to manage waste and protect workers, since many fluorinated chemicals linger in the environment. Industry and labs need to focus on tighter safety protocols, cleaner manufacturing techniques, and honest communication about risks.
Looking for Smarter Solutions
Instead of just pushing for more production, I believe the research community should keep asking tough questions: Can fluorinated intermediates be recycled or recovered? Are there safer alternatives with a similar impact on drug or material properties? Investment in green chemistry has shown promise by designing reactions that cut down on waste and avoid hazardous byproducts. Open access collaboration across academia, business, and regulators will help avoid the pitfalls of legacy chemicals and move toward safer, smarter science.
2,3-Difluorophenol may only be one piece of a much larger puzzle, but the lessons it brings go beyond chemistry. It’s about trading short-term fixes for long-term thinking—putting as much care into how we use chemicals as in why we use them in the first place.
Breaking Down the Numbers
2,3-Difluorophenol caught my eye during a late-night search for an answer to a fairly specific problem: what's the molecular weight of this compound, and why does such a detail even matter? The short answer is 130.09 grams per mole. It sounds like a technical tidbit out of a dusty chemistry book, but there's more behind these numbers.
The Story Behind a Small Molecule
Anyone who's worked in a chemistry lab, even if just for a college project, remembers the precision required in measuring out materials for a reaction. Calculations start with molecular weights. For 2,3-Difluorophenol, a benzene ring with two fluorines and a hydroxyl group, the weight breaks down like this: carbon atoms (12.01 g/mol each, with six of them), hydrogen atoms (1.01 g/mol times five), oxygen (16.00 g/mol), and fluorines (18.998 g/mol each, with two present). Adding those gives 130.09 g/mol.
This number isn’t just for professors or students grinding through lab reports. People in pharmaceuticals, agrochemicals, and materials science often turn to these precise calculations to make sure their processes deliver what they intend. The accuracy of such numbers can mean the difference between the right outcome and wasted effort—or worse, safety risks. Years ago, a mix-up in a summer research lab hammered that lesson home as a minor calculation led to a big headache when a yield seriously misaligned with our expectations. A simple number gained a lot of weight that day.
The Ripple Effects
Research teams use the molecular weight of compounds like 2,3-Difluorophenol to scale up production, determine how much of a reagent to use, and predict properties such as melting points and solubility. Companies building on organic synthesis must know these numbers to avoid unexpected results in their supply chains. A miscalculation can multiply errors across steps, undermining runs that cost time and money. These tiny missteps take on greater meaning when hundreds or even thousands of kilograms are in play.
The weight also feeds into environmental and toxicology assessments. Risk managers and product regulators lean heavily on these basics. If toxicity data gets linked to a substance, the exposure calculations won’t mean much if the compound’s molecular weight is off. Public safety, product compliance, and ongoing research all rely on accuracy at every stage.
Bridging Science and Usefulness
It’s easy to see this as only background data for technical specialists. But this accurate accounting of atoms forms the foundation of trustworthy science. Progress in healthcare, better crop protection products, even new coatings or electronics—a lot builds out from the careful detail caught in a molecular weight calculation.
Improving daily operations doesn’t require ultra-high-end equipment; it starts by respecting these numbers and double-checking them against reliable sources. Anyone making chemicals in a lab or at scale does well to keep basics like this close to hand, both for progress and peace of mind.
Understanding What’s on the Bench
I remember the first time I worked with a halogenated aromatic compound; a veteran chemist looked me in the eye and said, “No shortcuts here. Know what you’re handling.” That advice stuck with me, especially with chemicals like 2,3-Difluorophenol. This isn’t a substance you wave off as just another bottle in the storeroom. The phenol group alone can bite with skin and eye irritation. Toss in fluorine atoms, and you step up the challenge. Even seasoned researchers have learned all it takes is a splash to cause a long day in the emergency room.
PPE: Not Just Formality
Lab gloves, goggles, and a sturdy coat do more than satisfy checklists. Direct skin contact with 2,3-Difluorophenol can leave burns and a rash that lingers for days. Traditional latex gloves break down fast with phenolic chemicals, but nitrile or neoprene gloves show real endurance. Good safety glasses mean the difference between a bad scare and a vision problem that takes months to heal, especially since even a tiny droplet in the eye feels like being hit with vinegar and hot needles at once. In my own lab, a splash shield sits on every bench with halogenated phenols. It might add a step, but I’d rather look overly cautious than risk a colleague’s eyesight or my own.
Ventilation Shapes Results and Safety
Decent airflow does more than prevent chemical clouds from lingering. 2,3-Difluorophenol vapor is heavier than air, so it hugs surfaces, sinks into corners, and tends to stick around longer than you’d guess. Never trust your sense of smell or the absence of obvious fumes. Fume hoods do the heavy lifting here. If I’ve learned anything from years in shared academic labs, it’s that propping open the sash or skipping the hood leads to coughing, headaches, and sometimes nausea halfway through an experiment. Clean air isn’t just nice; it keeps brains sharp and hands steady while working.
Storage with Common Sense
It’s easy to let bottles multiply in drawers and forget about them between projects. That’s a shortcut to trouble. Stashing 2,3-Difluorophenol alongside acids or random oxidizers is a risky move. Phenols can turn reactive in a hurry, causing container breaches or even fires in extreme cases. Keeping incompatible chemicals far apart isn’t overkill—it’s basic safety. Investing in secondary containment, even as simple as a plastic tray or locked cabinet, stops leaks from ruining inventory and harming people. I rescued more than one project from disaster by catching a drippy bottle early, all because it was stored right and checked regularly.
Spills Demand Calm, Not Panic
Chemical spills set hearts racing, but the best response comes from practice, not panic. Soak up small spills with absorbent pads and toss them in a sealed bag for disposal with hazardous waste—not the trash. And never, ever use paper towels or sponges meant for everyday cleaning. The risk of fire or unexpected reactions is real. My routine includes reporting every spill, no matter how minor. This helps catch patterns and fix equipment or procedures before accidents escalate. The long game is safety, not speed.
Chemical Respect Builds Careers
Treating 2,3-Difluorophenol with seriousness doesn’t slow research down—it accelerates trust and good results. Every step, from knowing your gloves to using the right hood, signals a genuine commitment to health and professional standards. Labs where safety is visible keep accidents rare, and people stick around longer, building better science and stories for the next generation. A few extra moments of care at the bench keep experiments running and communities healthy. Respect in the lab doesn’t just protect the present; it shapes how well we can pass knowledge forward, bottle by bottle.
Anyone who’s spent time in a lab knows the challenge of getting a substance to dissolve just right. 2,3-Difluorophenol, with its two fluorine atoms stuck to a phenol ring, behaves differently in water compared to various organic solvents. It seems simple—add some of it to a beaker of water, give it a stir, and see what happens. In reality, the story goes deeper, especially for chemists, environmental engineers, or folks working on pharmaceutical synthesis. Every little detail—the structure, polarity, and even the number and position of those fluorine atoms—shapes how this compound interacts with water and other liquids.
Water: A Tough Crowd for 2,3-Difluorophenol
Add 2,3-Difluorophenol to water and you soon find the limits. Fluorination decreases a molecule’s overall polarity and increases hydrophobicity. The hydroxyl (OH) group still holds onto some hope of hydrogen bonding with water, but the two fluorine atoms make life tough for solubility. Sibbesen, Schindler, and other chemists publishing on halogen-substituted phenols have consistently shown that throwing more halogen atoms on a benzene ring cuts down aqueous solubility. Compared to regular phenol, 2,3-Difluorophenol dissolves far less in water. The numbers tend to hover below 3 grams per 100 mL, and this is on the generous side. Most users report needing to heat or shake like crazy just to get modest amounts into solution.
When I worked in a lab synthesizing analogs for drug discovery, this poor solubility slowed things down. Water-based reactions became a headache, and even simple washing steps needed a rethink. Environmental scientists face similar frustrations—persistence in groundwater, for instance, has less to do with actual toxicity and more to do with a molecule’s refusal to mix with anything but the stubborn organic debris in the earth below.
Organic Solvents: The Right Partners
Move from water to organic solvents, and the picture shifts. Organic solvents such as ethanol, acetone, diethyl ether, and acetonitrile welcome 2,3-Difluorophenol in much higher concentrations. In my experience with extractions and chromatography, it almost disappears into these solvents, blending in at over 20 grams per 100 mL in some cases. The phenolic ring, thanks to its aromatic structure, gets cozy with the pi-bonds of toluene, benzene, and similar solvents. Both the molecule and the solvent avoid forming strong hydrogen bonds. It’s a chemical friendship built around avoidance of water rather than outright attraction.
The catch is that not all organic solvents are created equal. Some, like methanol, spark a little hydrogen bonding with the phenolic OH, boosting solubility even more. Solvents with bulky or nonpolar structures, such as hexane, show limited success. Picking the right one depends on the task—dissolving for a reaction, moving something out of a mixture, or storing the chemical safely for later use.
What Researchers and Industry Can Do
Rather than fighting the solubility war head-on, smarter labs design synthesis schemes that either use organic solvents directly or convert 2,3-Difluorophenol into more tractable intermediates. For those in the pharmaceutical space, tweaking the molecule or using co-solvents gets results. Green chemists, aware of the environmental push to avoid organic solvents, experiment with ultrasonic agitation, phase-transfer catalysts, or novel emulsifiers to coax better dissolution in water. There’s still no easy fix—but chemists rarely expect one.
For now, a simple rule holds true: 2,3-Difluorophenol barely budges in water, but it finds its element among organic solvents. Understanding what drives this—down to the level of molecular structure and interaction—helps pick the right tools for every project that uses or studies this tricky little molecule.