Understanding 2,4,6-Trifluorophenol: From Roots to Future
Historical Development
Chemists have chased after modified phenol derivatives for over a century, driven by a search for materials with new electronic and structural qualities. Fluorinated aromatics made headlines in the post-war decades, once folks realized that introducing fluorine atoms into basic organic scaffolds produced chemicals with striking changes in reactivity and stability. 2,4,6-Trifluorophenol emerged as a point of special interest during the late 20th century, as pharmaceutical and agrochemical companies dug deep into molecular fine-tuning. The substitution pattern, with fluorine atoms tucked in at three separate ring positions, set the stage for materials with unusual acidity, resistance to metabolic breakdown, and unique roles in coupling reactions.
Product Overview
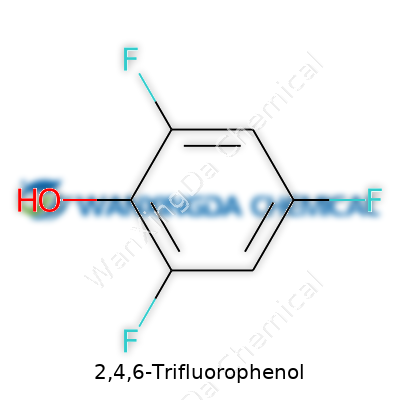
2,4,6-Trifluorophenol looks simple on paper but punches above its weight in specialty chemistry. Three fluorines placed symmetrically around a phenolic ring bring a charisma that isn't found in regular phenol. This chemical helps build new compounds, influences how drugs behave in the body, and serves as a touchstone in both basic and applied chemical research. If a synthesis calls for a phenolic group that resists oxidation and delivers higher acidity, trifluorophenol stands out as a strong candidate.
Physical and Chemical Properties
Solid at room temperature, 2,4,6-Trifluorophenol forms colorless to faintly off-white crystals, with a subtle odor that betrays its phenolic roots. Its melting point lands a bit higher than its non-fluorinated cousins, since those tough carbon-fluorine bonds stiffen up the structure. The molecular weight tips the scales at 148.08 g/mol, packing more heft than plain phenol thanks to the extra fluorine. Its solubility in water drops below that of phenol, as the fluorines strip down hydrogen bonding. In organic solvents—like ether and acetone—it dissolves far more readily. Anyone working with 2,4,6-derivatives comes to appreciate how these minor tweaks at the atomic level produce such a radical difference in properties.
Technical Specifications & Labeling
Buyers expect 2,4,6-Trifluorophenol to meet exacting technical specs, especially when the final product could wind up in pharmaceuticals, diagnostics, or materials for electronics. Purity typically runs above 98 percent for research and industrial use, and trace metal analysis matters if the compound will see service in catalysis or electronics fabrication. Labeling follows established conventions: the CAS registry number, hazard pictograms, and signal words are non-negotiable, especially for international transit. Accurate labeling protects workers, researchers, and anyone handling the material during storage and transport.
Preparation Method
Synthesizing 2,4,6-Trifluorophenol forces organic chemists to get creative, as direct fluorination of phenol can run amok and produce all kinds of messy byproducts. Most routes start with pre-halogenated aromatic building blocks. Nuts-and-bolts fluorination methods swap halogens for fluorine using reagents like potassium fluoride or cesium fluoride under high temperatures, often with polar aprotic solvents. Fancier approaches leverage catalytic fluorinating agents or use nucleophilic aromatic substitution strategies, making it possible to get to the 2,4,6 pattern with minimal side reactions. The ideal method delivers high yield, few impurities, and less environmental load from solvent waste and byproducts.
Chemical Reactions & Modifications
What gets researchers excited about 2,4,6-Trifluorophenol is how those fluorine atoms tune the reactivity of the phenol ring. That makes the molecule a playground for reaction mechanism studies and custom syntheses. Its phenolic proton, made even more acidic by the electron-hungry fluorines, opens doors in nucleophilic aromatic substitutions. Chemists reach for it when they want to add a tough, electron-withdrawing group onto a new material or mimic metabolic transformations found in natural product biosynthesis. It’s also come to play a role as an intermediate in creating new ligands for transition metal catalysts, as well as customized blocking groups that withstand intense reaction conditions.
Synonyms & Product Names
The molecule travels under a few different aliases: 2,4,6-Trifluorophenol, Benzol-2,4,6-trifluorol, Phenol, 2,4,6-trifluoro-, and even sometimes as TFP. Each name points back to the same core structure; the preferred IUPAC name keeps the field on the same page in publications. Clarity in naming cuts down on errors when ordering, handling, or discussing the compound in research and regulatory circles.
Safety & Operational Standards
Working with 2,4,6-Trifluorophenol means taking exposure risks and reactivity into account. Like many phenols, it can irritate skin, eyes, and mucous membranes, so labs keep it in tightly capped containers, work behind fume hoods, and stress the use of nitrile or neoprene gloves. Safety data sheets point out its moderate toxicity and recommend keeping absorption and inhalation to a minimum. Storage calls for cool, dry, dark areas, away from incompatible materials like strong bases or oxidizers. Over a decade in the lab taught me that the best safety routines depend on both preparation and vigilance. Double-checking labeling, never storing strong acids and bases together, and running small-scale test reactions before ramping up remain best practice.
Application Area
2,4,6-Trifluorophenol’s biggest payoff comes from its ability to shape the behavior of complex molecules. Its use ranges from pharmaceutical building blocks—where it helps craft molecules that dodge enzymatic breakdown or change bioavailability—to advanced materials known for resilience and unusual electronic traits. Agrochemical makers mix it into new pesticides and herbicides that stick around long enough to do their job without excessive breakdown. Specialty polymer manufacturers add trifluorophenol as a monomer, building advanced plastics for electronics and protective coatings. On a smaller scale, it serves in research labs that look to understand and manipulate the subtle effects of fluorine on molecular properties.
Research & Development
For chemists, 2,4,6-Trifluorophenol pulls double duty: it both serves as a scaffold for molecule building and as a test case for new synthetic techniques. I’ve seen labs use it as a model when tweaking nucleophilic aromatic substitution protocols, since those three fluorines ramp up the reactivity and make mechanistic studies clearer. Drug discovery researchers lean on it to build libraries of analogs—just a change in the position or number of fluorine atoms can turn a weak drug candidate into a standout performer with altered metabolic fate. In material science circles, its electronic quirks invite new research. Teams have explored its use in self-assembling films, proton-conducting membranes, and designer catalysts.
Toxicity Research
The field treats all fluorinated aromatics with caution, because animal studies show that certain derivatives can stick around in organisms and disrupt regular biochemistry. While the acute toxicity of 2,4,6-Trifluorophenol hasn’t hit the red flag levels of more notorious fluorinated compounds, safety groups warn against long-term or high-dose exposure. Researchers follow strict guidelines for disposal and use, since breakdown products in waste streams may pose risks for aquatic life or disrupt microbial communities. Newer research pushes to understand not just gross toxicity, but also the subtle impacts on enzyme function, signaling pathways, and the environment, in line with rising expectations about chemical stewardship.
Future Prospects
Looking ahead, the place for 2,4,6-Trifluorophenol seems secure, though shaped by changing regulations, sustainability demands, and the hunt for greener chemistry. The push for high-performance but less persistent chemicals motivates researchers to engineer better routes for synthesis, cut solvent waste, and improve recovery and recycling. Advances in catalysis and substitution chemistry should make it easier for large-scale processes to run cleaner and cheaper. The ongoing search for pharmaceuticals and industrial materials that stand up to harsh conditions without stubbornly lingering in the environment will keep attention focused on the balance between performance and biodegradability. My own experience suggests that regulatory scrutiny will tighten, especially as communities face the consequences of per- and polyfluorinated compounds showing up in water sources and the food chain. In every field touched by fluorinated phenols, the next decade calls for creativity, responsibility, and open dialogue between chemists, policymakers, and the public.
Understanding What It’s For
2,4,6-Trifluorophenol doesn’t get much attention outside labs and specialty chemical circles. Most people have never heard the name, but this compound plays a quiet, important role in building complex molecules for medicine, materials, and research. The world depends on chemical building blocks like this. Whether we realize it or not, these substances shape the medicines we take and the electronics we use daily.
Building Better Medicines
People working in pharmaceutical research use 2,4,6-Trifluorophenol because it’s good at helping attach certain chemical groups to molecules. Proteins in our bodies can react with a huge range of substances, but scientists have learned that tweaking structures with fluorine—like what comes from this trifluorinated molecule—often helps new drugs resist breakdown and work longer. This small difference sometimes means the difference between a drug that fails and one that succeeds in the body.
My first job in college was in a pharmaceutical lab, helping the team run tiny-scale experiments testing reactions. We’d use trifluorophenol derivatives because they offered stability, even under harsh reaction conditions. That meant experiments would run longer before they “died,” so we’d get more reliable results—something every chemist wants.
Modern Manufacturing Relies on Specialty Chemicals
I’ve noticed that tech companies and materials science researchers also work with fluorinated building blocks for next-generation plastics, resins, and coatings. These advanced materials show up in circuit boards, fiber optics, and even specialized filters. Trifluorophenol plays a part in making some of these materials more durable, giving them flexibility or helping them resist harsh industrial environments. When your phone survives a drop or your laptop lasts for years, these advances feel invisible—but the chemistry matters.
Research published in journals like the Journal of Fluorine Chemistry highlights how even small changes, like using trifluorinated phenols in a polymer, can lead to greater heat resistance and mechanical strength. Engineers count on those gains because nobody wants products that fail in the field. Reliable chemistry is central to solving that problem.
Environmental and Safety Considerations
Whenever I hear about a new chemical showing up in research articles, I think of waste and disposal. The world has learned some tough lessons with persistent pollutants—think PFAS or other “forever chemicals.” While 2,4,6-Trifluorophenol isn’t in the same class as those super-stable pollutants, any compound with multiple fluorines deserves careful handling. Chemists who use this compound need proper training, and companies producing it should invest in safe waste management.
Countries like the United States and members of the European Union enforce strict rules to track, store, and dispose of specialty chemicals. That matters when we scale up from tiny lab amounts to full-scale industrial use. Good housekeeping in the lab or plant prevents problems in water systems and neighborhoods later on.
Looking Ahead
As industries push for greener practices, finding safer, more sustainable ways to build molecules remains a priority. Some chemists turn to renewable sources, others work on better recycling or safer degradable alternatives. Demand for molecules like 2,4,6-Trifluorophenol will stick around for now, with ongoing research keeping an eye on toxicity and environmental impact. Upholding strict safety measures and continued investment in green chemistry will balance the needs of innovation with the health of our communities and environment.
Understanding the Formula and Weight
2,4,6-Trifluorophenol, at first glance, doesn’t grab much attention. It’s a small molecule with a scientific name that may scare off anyone who hasn’t spent their evenings poring over organic chemistry textbooks. Strip away the jargon, and what you have is a molecule made up of carbon, hydrogen, fluorine, and oxygen—nothing exotic, just the building blocks that keep showing up in nearly every pharmacy shelf. Its molecular formula is C6H3F3O, and its molecular weight hovers around 148.08 g/mol. Fact is, those numbers get tossed around so much in research circles, it’s easy to overlook what makes this simple-sounding compound so significant.
Why These Numbers Matter in Real Life
Snagging the accurate formula and weight for compounds like 2,4,6-Trifluorophenol isn’t just a paperwork obsession. Every milligram counts when you’re in a lab, even more so when the product is headed for something as sensitive as a drug trial. Dig out the wrong molecular weight, and an entire batch of research or production might head for the trash. Precise math saves money and cuts down risk—missing the mark even by a hair could mean someone ends up with an ineffective or unsafe product. I’ve watched researchers double-check the same numbers, over and over, just to make sure the process runs without a hitch. They do it for a reason.
The Chemistry Behind the Use
What really stands out about 2,4,6-Trifluorophenol isn’t just the numbers, but what those three fluorine atoms do. Each position on the benzene ring influences not only physical properties like boiling point and solubility but also the reaction patterns. Introducing fluorine changes the molecule’s electron distribution. Some might see this as a dry detail, but it impacts how reactive the compound becomes, influences medicinal chemistry strategies, and can enhance a drug’s metabolic stability. The presence of the three fluorines, especially in those precise locations, is more deliberate than many non-chemists realize. It’s about tuning the molecule for a purpose, almost like a chef fine-tuning the seasoning in a signature dish.
Where the Trouble Comes In
Working with fluorinated phenols isn’t always a walk in the park. Safety regulations are tight for a reason—fluorine’s benefits come with baggage. I remember stories from colleagues about accidentally inhaling vapors from similar compounds and spending hours huddled around a fume hood cursing their own mistakes. Waste management gets tricky. Incinerators built to handle standard organics might not break down fluorinated stuff completely, and the world has enough trouble with persistent pollutants. Everyone talks about sustainability, but handling fluorinated molecules highlights how much further there is to go. Scientists have to balance innovation with environmental responsibility, and that conversation gets louder every year.
Looking for Better Solutions
The simple facts—like the formula C6H3F3O and a weight of 148.08 g/mol—anchor bigger decisions. Innovation doesn’t just stop at designing new molecules. It branches out: finding ways to use greener chemistry, recycling spent materials, and updating safety practices based on the latest health data. Some researchers look for alternatives to traditional fluorinated phenols, hunting compounds that offer similar benefits with fewer drawbacks. Funding often follows those who can pair smart science with practical safety and sustainability. There’s no shortcut; it takes persistence, curiosity, and a willingness to keep pushing for progress, even with something as deceptively simple as 2,4,6-Trifluorophenol.
Looking Beyond the Label
People who work in labs or industries where chemicals play a role face more than equations and glassware; they're also responsible for what's behind the locked doors and labeled bottles. Take 2,4,6-Trifluorophenol, for example. On paper, it’s just another specialty compound, but experts and workers know it does more than just sit quietly on a shelf. Safe storage and proper handling don’t just tick boxes for compliance—they protect people, projects, and the bottom line.
No Room for Guesswork
Straight talk: 2,4,6-Trifluorophenol isn’t a household item. The liquid form, clear as water, gives a false sense of security. Inhaling its fumes or letting it touch your skin means risking burns, blisters, and coughing fits, based on real case reports from industrial hygiene studies. The chemical's toxicity draws concern in both research papers and workplace incidents, once even prompting a temporary shutdown at a small specialty plant.
I’ve watched chemists learn the hard way that storing it like generic lab alcohol doesn’t cut it. One summer, a colleague left a poorly sealed bottle near a heat source. The result wasn’t dramatic, but the fumes made the room unworkable for hours, and a report later said the exposure approached hazardous limits. That tells me a metal cabinet with proper ventilation isn’t bureaucracy; it’s basic respect for health and safety.
Best Practices Grow from Hard Lessons
Experience, not just rules, teaches respect for details. Direct sunlight or rising summer heat can alter the compound. Some bottles end up stuck to shelves from seepage, a mess no one wants to handle. Keeping it in a cool, dry space means more than comfort—it reduces evaporation, leaks, and accident risk. Flammable liquid safes and chemical storage refrigerators keep volatility in check.
I’ve also seen how small lapses with 2,4,6-Trifluorophenol stack up over time. A spill that isn’t properly contained can threaten not only air quality but also lab morale. Regular lab audits helped spot the telltale, musty odor that lingers after a minor leak, and after that, eye-level storage of clear labels and secondary containment became the rule.
PPE: Not Just a Rule, a Routine
Stories spread fast in labs—someone gets a rash, another calls in sick. Strong chemical gloves, goggles, and lab coats sound routine, but for chemicals like this, a mask or respirator helps, too. One slip with contaminated gloves touching skin can lead to hours of discomfort, as any senior lab tech will tell the newcomers. Strict hand-washing after handling seems hardly worth repeating, but those who skip it, unfortunately, end up learning why it matters.
Thinking Wider: Training and Culture
The explanation always circles back to training and a culture of collective responsibility. Annual refreshers, mock spill drills, or simply pointing out a loose cap save headaches and costs down the road. A safety culture isn’t just a slogan—people remember the stinging eyes and bitter taste in the air more vividly than a stern memo.
Smart handling and storage of chemicals like 2,4,6-Trifluorophenol demand more than just checking off requirements. They call for listening to those who’ve dealt with real-life mishaps, learning from close calls, and treating every bottle with caution and respect. It’s the investment in awareness and care—not just hardware or policy—that keeps people safe and lets science move forward.
Understanding What’s in the Bottle
Ask any lab technician or synthetic chemist about purity and sooner or later you’ll hear a story about a reaction gone astray. In the world of specialty chemicals, purity isn’t a luxury—it lets you trust your work. 2,4,6-Trifluorophenol, a building block in pharmaceuticals and materials science, is no exception.
It’s hard to forget the moment a batch of material slipped through quality control and ruined a crucial experiment. I’ve watched friends lose weeks because a supplier sold them phenol littered with mystery peaks on the NMR. For 2,4,6-Trifluorophenol, the label almost always says ≥98% or ≥99% purity. These numbers aren’t marketing fluff; they mean fewer headaches downstream. If you’re chasing a new drug target or developing functional polymers, an impurity can mask your results or spark safety concerns. Purity translates to dependable performance.
Packaging Options Tell a Bigger Story
I remember my first industrial order: I only needed a few grams, but the only option online was a 250-gram drum. The lesson is simple—packaging matters. For 2,4,6-Trifluorophenol, suppliers usually offer bottles starting at 1 gram or 5 grams, suitable for research. Scale jumps to 25 grams, 100 grams, or even 500 grams. Large quantities often come in industrial packaging, though those who need them tend to have established supply chain relationships.
These sizes reflect the chemical’s use. In drug discovery, you rarely need more than a handful of grams to run assays and synthesis. On the other hand, a pilot plant or production line eats up bigger batches. It’s never just about the price per gram—it’s about waste, storage stability, and escaping the nightmare of cross-contamination. Smaller bottles mean someone upstream is thinking about shelf life and safe handling.
Transparency as a Pillar
Trust in chemical supply hinges on transparency. The best suppliers back their purity claims with detailed certificates of analysis. Labs need to see GC or HPLC traces, not just numbers on a sheet. Even a one percent unknown can cause endless troubleshooting when scaling a process. Supply chain reliability, clear documentation, and traceable batch information all play into the E-E-A-T framework Google emphasizes—demonstrating real-world experience, expertise, and a solid foundation of trust.
A close colleague once caught a supplier quietly switching formulations to cut costs, leading to subtle impurities sneaking into the workflow. The damage showed up weeks later in inconsistent results. Reliable suppliers prioritize data transparency to keep their reputation intact. It isn’t just about ticking compliance boxes; chemists rely on that consistency to do their best work.
Pushing for Better Solutions
Instead of generic packaging and mystery specs, suppliers have an opportunity—and a responsibility—to engage with their users. That means feedback loops about what packaging sizes work best, honest conversations about shelf-life, and willingness to adjust offerings when research trends shift. Digital tools like QR-coded batch data, on-demand lot histories, and clear communication channels can help prevent costly mistakes. The labs themselves can push for these changes by demanding higher standards from their partners instead of settling for the status quo.
Purity and packaging shape the work scientists do every day. Addressing these two factors head-on won’t just make research more efficient—it will save time, money, and trust for everyone involved.
Digging Into the Problem
Anyone who has spent time around chemicals in any lab—research, quality control, or industrial production—knows one fact: some substances don’t announce their hazards loudly, but that doesn't mean they're safe. 2,4,6-Trifluorophenol finds a spot in several synthesis routes in pharma and materials labs, yet rarely does anyone outside those circles know its name. This compound doesn’t land itself on TV or in public controversy, and because of that, most of us forget it sits on shelves with real risk attached.
Hazard Story, Unwrapped
The basic picture: 2,4,6-Trifluorophenol packs both toxicity and environmental persistence. Its sharp, acrid smell hints you shouldn’t inhale it. If a splash lands on bare skin, you realize the burning isn’t just from your imagination. Acute symptoms after exposure include irritation to eyes, nose, throat, and lungs. Anyone unfortunate enough to inhale its dust or vapors can deal with headaches, dizziness, or even respiratory tract discomfort. There isn’t ample epidemiology on chronic effects, but experience tells most chemists—persistent exposure to phenolic compounds rarely brings anything good. It pays to treat trifluorophenol with the sort of respect you’d give any corrosive organics. Skin contact, ingestion, or direct inhalation should never happen without sturdy protection.
Safety Data Sheet Red Flags
Every manufacturer expects labs to keep a Safety Data Sheet (SDS) on hand, and for good reason. An SDS for this compound lists gloves and goggles as absolute musts. Chemical-resistant gloves such as nitrile work best, because trifluorinated aromatics can fight their way through thinner latex or vinyl. Chemical goggles, not just safety glasses, work to cover all angles when the risk of mist or splashes exists. Many standard fume hoods can handle low-level releases, but a crowded hood and clumsy handling multiply the risk. Ventilation systems need regular checks to prevent vapor buildup. Most labs direct waste containing 2,4,6-Trifluorophenol into labeled organics, keeping it away from regular trash or gray water drains—because this compound resists biodegradation and raises issues if it enters soil or waterways.
Regulatory Situation: Gaps and Gray Zones
This is where things get messy. In the United States, 2,4,6-Trifluorophenol doesn’t pop up on every regulated chemical list, although some states or countries classify it under hazardous waste because of its toxicity. The Toxic Substances Control Act catalogs it, so its use falls under basic reporting and responsible handling protocols. In Europe, REACH registers it as a substance that demands careful tracking in advanced manufacturing. No one in chemical safety expects to see this compound handled by non-professionals or left unsecured in schools or homes. This lack of clear, specific controls means responsibility falls heavily on employers, lab managers, and individuals—often outpacing enforcement by government inspectors.
Ground-Level Solutions
Institutions and companies can’t afford to treat trifluorophenol like inert solvents or minor lab chemicals. The answer starts with thorough in-house training: new staff must learn not just how to protect themselves, but what separates this compound from less hazardous options. Chemical hygiene plans should include regular drills for spills or accidental exposure—no one should have to improvise cleanup in the heat of a real incident. Most crucially, purchasing officers can drive change by choosing greener alternative reagents if the synthesis allows. While some processes demand trifluorophenol’s chemistry, exploring alternatives eliminates risk at the source. If the compound stays in use, clear inventory controls and proper waste management keep it—and everyone nearby—accountable.
Labs work best when everyone knows what danger looks like, even if it comes in a plain, unassuming bottle. Chemicals like 2,4,6-Trifluorophenol deserve both respect and a sense of responsibility backed by policy, not just paperwork filed and forgotten.