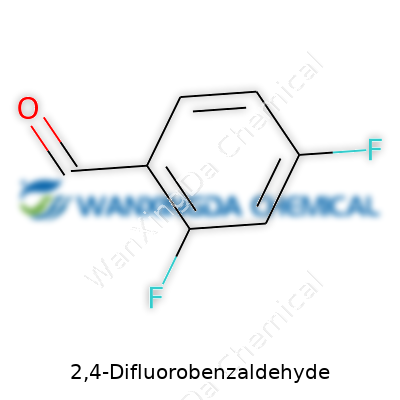
2,4-Difluorobenzaldehyde: A Closer Look at its Role in Chemistry and Industry
Historical Development
Many folks outside the world of synthetic chemistry see chemical names like 2,4-difluorobenzaldehyde and tune out fast. But a compound with two precisely placed fluorine atoms on a benzaldehyde ring doesn’t just pop up; this molecule reflects decades of intentional design and shifting needs in the chemical industry. Back in the 1950s and ’60s, as fluorination became a key strategy for tuning molecules, researchers zeroed in on aromatic aldehydes, hoping for new flavors, fragrances, pharmaceuticals, and pesticides. The rise of organofluorine chemistry meant folks tried out every combination of positions and substituents on the benzene ring, so it didn’t take long before someone reported 2,4-difluorobenzaldehyde and cooked up ways to make it part of bigger synthetic plans.
Product Overview
For anyone who’s tinkered at the bench or in the plant, 2,4-difluorobenzaldehyde carries the whiff of a working aldehyde: biting, a little sweet, and just a bit more pungent thanks to those electronegative fluorines. Chemists don’t keep it around just for its smell, though. This compound often acts as a building block, helping assemble more complex molecules in steps where precision matters. It gets picked for jobs where a regular benzaldehyde might be too reactive, or too plain, and where those two fluorines help steer the chemistry or boost stability, volatility, or other traits that come from fluorination.
Physical & Chemical Properties
Anyone buying or handling this stuff quickly learns its physical habits. 2,4-difluorobenzaldehyde appears as a clear, colorless to pale yellow liquid at room temperature. Its boiling point hovers around 84-86°C at reduced pressure, making purification manageable through distillation in a fume hood but not something to do with an open flame or a casual setup. The molecule’s structure, a benzene ring decorated with two fluorine atoms and a formyl group, shapes its reactivity. Compared to regular benzaldehyde, it’s a bit more resistant to oxidation and slightly less eager to jump into some nucleophilic addition reactions. The fluorines can pull electron density from the ring, changing how chemists can push or pull its behavior in reactions.
Technical Specifications & Labeling
Every bottle, drum, or vial of 2,4-difluorobenzaldehyde gets a detailed label because mishandling aldehydes can lead to headaches, both literal and operational. Most suppliers state a minimum purity threshold, usually not less than 97 percent, and flag the need for dry, cool storage. Labels flag the compound as harmful if inhaled or touched, warning users to handle it with gloves, eye protection, and proper ventilation. Since 2,4-difluorobenzaldehyde counts as a hazardous chemical under several global systems, folks use pictograms, hazard, and precautionary statements to stay in the clear with regulators.
Preparation Method
Old organofluorine handbooks and recent journals describe routes that aren’t too exotic for well-equipped labs. The classic way involves fluorination of a suitable benzaldehyde precursor, using select reagents like Selectfluor or elemental fluorine in tightly controlled reactions. Sometimes, chemists start with a fluorinated toluene, then use oxidation to convert the methyl group to the aldehyde—the famous use of chromium trioxide, manganese dioxide, or other oxidants. Preparation can require extra attention to regioselectivity since putting fluorines in the right spots matters more than ever with complex downstream chemistry.
Chemical Reactions & Modifications
Chemists prize 2,4-difluorobenzaldehyde for the way it reacts and the products it opens up. The fluorines change classic benzaldehyde chemistry: they slow down some additions and speed up others. Knoevenagel condensations, Wittig reactions, and Grignard additions can all use this aldehyde, and the products often keep the unique character that those fluorines stamp onto the ring. Folks working in medicinal research appreciate this, since fluorinated aryl groups offer metabolic stability and sometimes sneak drugs past enzymes that would chew up plain aromatics. This compound can head into reductive aminations, acylations, or even more elaborate cross-couplings once the aldehyde or aromatic handles get functionalized.
Synonyms & Product Names
In catalogs and progress reports, 2,4-difluorobenzaldehyde may answer to several names, including Benzaldehyde, 2,4-difluoro-, or 2,4-DFBA. The systematic IUPAC name isn’t winning any style awards, but it gets the job done and shows up on inventory lists. One thing is steady: no matter what it’s called, the key is tracking those two fluorines and the aldehyde group, because both play starring roles in whatever comes next.
Safety & Operational Standards
Dealing safely with aromatic aldehydes like 2,4-difluorobenzaldehyde calls for more attention than everyday lab solvents. The vapor can irritate eyes, nose, and throat, and skin contact tends to cause redness or worse in sensitive folks. Keeping the stuff behind closed sash hoods, with the right gloves and eye protection, matters far more than keeping things tidy. Storage requires a cool, dry shelf, away from oxidizers, acids, and bases, and everything should be tightly capped to limit exposure and evaporation. Disposal should never happen down the drain; if there’s leftover or waste, teams funnel it to hazardous waste streams, in line with both local law and best practices.
Application Area
It’s easy to overlook how many projects quietly depend on molecules like 2,4-difluorobenzaldehyde. In pharmaceuticals, chemists use it to anchor more complex syntheses or embed fluorine into drug candidates, where it can make or break metabolic stability. Agrochemical researchers reach for it to craft new herbicides or pesticides that won’t break down too quickly and resist bugs’ evolving defenses. Some folks in material science try out unusual fluorinated aromatics in polymers or liquid crystals for displays, films, and coatings where fluorine’s subtle effects shine. Its reach stretches quietly into paints, fragrances, and laboratory research, each field exploiting the unique twist those two fluorines give.
Research & Development
Over the last ten years, academic labs and big industry R&D teams keep turning to fluorinated aromatics as tools to tweak both known and new molecules. 2,4-difluorobenzaldehyde gets its share of attention, showing up in medicinal chemistry as a key intermediate in the search for better drugs. Nature rarely makes these complicated fluorinated aromatics, so humanity depends on synthetic methods. That challenge keeps pushing innovation—finding greener syntheses, skipping toxic reagents, or boosting yields. In catalysis, researchers look for ways to attach the aldehyde or aromatic to other units using modern coupling strategies, hoping to make everything faster, cheaper, and safer.
Toxicity Research
What’s known about 2,4-difluorobenzaldehyde’s toxicity tilts in the same direction as its close relatives: exposure above safe limits brings irritation, with longer exposures leading to more serious issues. So far, chronic or long-term studies are fewer, since the main user groups—industrial and laboratory chemists—meet it only under controlled circumstances. Environmental persistence comes up thanks to those stubborn fluorines; while the aldehyde group can break down in the environment, the aromatic ring resists, raising flags for waste handling and discharge. New research continues to probe environmental fate, acute and sub-chronic toxicity, and safe exposure levels, pushing regulators to update recommendations.
Future Prospects
Demand for 2,4-difluorobenzaldehyde is poised to keep growing, especially as chemists hunt for more ways to take advantage of fluorine’s unique fingerprints in everything from cancer drugs to organofluorine materials. The shift toward greener and more sustainable chemical production means that engineers and researchers continue working on less wasteful, less hazardous routes to this intermediate. Fluorination remains an expensive and tricky process, but the potential payoffs drive substantial innovation in both industry and academia. There’s little doubt that as the push for higher-performance pharmaceuticals and specialty materials continues, this once-obscure molecule will keep finding its way into new and surprising niches.
Trying to Make Sense of a Seemingly Simple Chemical
2,4-Difluorobenzaldehyde might not ring any bells for most people. It sounds like just another chemical cooked up in some distant lab, a string of numbers and syllables that chemists throw around. Dig a little deeper, though, and the stuff opens a window into the immense reach of chemistry in modern life. Even if you have never seen it on a grocery shelf or in a product label, there’s a decent chance you’ve crossed paths with something it helped create.
What Actually Happens with 2,4-Difluorobenzaldehyde
In my own brushes with chemical R&D, I’ve watched how something as seemingly minor as a benzaldehyde derivative gets handed off from one project to the next. Organic chemists use molecules like 2,4-Difluorobenzaldehyde as building blocks. Their main job: acting as intermediates for bigger, more complex molecules. Pharmaceutical companies lean on these compounds to anchor reactions that end up as medicines, or drug precursors. Agrochemical industries find value in its structure for making advanced crop protection products.
Sneaky as it sounds, adding two fluorine atoms changes what benzaldehyde can do. Fluorine’s electronegativity gives chemists control over how the molecule reacts. That might mean helping to steer transformations for making anti-cancer drugs, antibiotics, or pesticides. I’ve heard it compared to putting a stronger set of brakes and steering on a race car—you can drive reactions where you want, without spinning out. Sometimes, research papers come out describing new fluorinated benzaldehydes unlocking easier ways to make complex drugs. The attention doesn’t sit just with new medicines—there’s development in dyes, specialty polymers, and even materials science. When you talk about the supply chain for life-saving therapeutics or industrial coatings, tracing backward from the pill or paint can often loop right through chemicals like this.
Why Should Regular People Care?
The easy response would be to shrug. If something only turns up as an ingredient in research, why worry? On a broader level, though, it’s about how innovation happens. Having access to specialty chemicals like 2,4-Difluorobenzaldehyde gives researchers the tools to tinker, modify, and push new discoveries forward. Drug companies reported that over 20 percent of new small-molecule therapeutics contained at least one fluorinated component in the past decade. These tweaks improve how long a drug lasts in the body or how specifically it targets an illness. None of that happens without a deep bench of raw materials.
What Makes It Tricky, and How We Move Forward
Dealing with these chemicals reminds me of sitting around a workbench with my colleagues, swapping stories about failed syntheses and compounds turned into hazardous waste. Fluorinated aromatics require careful handling. Factories producing intermediates like 2,4-Difluorobenzaldehyde must strictly control emissions to avoid environmental and toxicity risks. Managing disposal and minimizing worker exposure are ongoing challenges. There’s a push for greener chemistry—finding ways to make and break down these molecules with less impact. Some labs focus on water-based methods or catalysts that cut out the worst solvents. Government guidelines push for safer, cleaner production, but it’s a dance between feasibility and regulatory reach.
We shouldn’t lose sight of the upsides. We expect rapid medical breakthroughs, strong new materials, and safer pesticides. Getting there depends on keeping access open to well-characterized chemicals. 2,4-Difluorobenzaldehyde doesn’t show up in the news much, but its impact touches society each day in ways that are easy to miss.
Looking Close at a Key Chemical Formula
Every chemist, from the high school classroom to the professional lab, eventually runs into a compound structure that sticks in the mind. 2,4-Difluorobenzaldehyde is one of those. The chemical formula, C7H4F2O, spells out more than a mix of letters and numbers—it tells a story about how molecules build themselves, bond, and end up mattering to so many fields.
Experience in the lab has always taught me the importance of seeing beyond the name or even the formula. When I first studied aromatic aldehydes, I didn’t pay much attention to subtle substitutions on a benzene ring. Two fluorine atoms take the usual carbon-hydrogen backbone into uncharted territory. Each substitution affects the reactivity, boiling point, and how the compound interacts—all crucial for anyone working in synthesis or pharmaceuticals.
Why 2,4-Difluorobenzaldehyde Stands Out
There’s a bigger lesson here. Adding fluorine atoms to a benzaldehyde ring isn’t just about tweaking a recipe. The fluorine atoms at positions 2 and 4 don’t just change the weight but shape how the molecule behaves. It becomes less reactive with certain agents and more reactive with others. Medicinal chemists look at tweaks like this all the time. Fluorinated aromatic rings can turn a basic molecular scaffold into a powerful drug or an important research tool.
If you ask any seasoned chemist about working with molecules like this, most will point to the challenge of selective substitution. Putting two fluorines exactly where you want isn’t trivial. In my own background, I’ve watched new students climb the learning curve in aromatic substitution: one substitution makes the next one easier or tougher, depending on the nature and position. This isn’t something a software tool or a textbook alone can explain. Getting hands-on with such compounds is the way to build that instinct.
Facts and Applications
Talking about formulas, it’s not enough to know the numbers. C7H4F2O means you’ve got a benzene ring (C6) with a CHO group (the aldehyde) and two fluorine atoms. That structure makes it valuable for several downstream uses—especially as an intermediate for pharmaceuticals, agricultural compounds, and material science projects. Recent trends in medicinal chemistry show fluorinated aromatic aldehydes popping up all over high-impact research articles. Companies are on the lookout for new, more effective ways to add fluorine to organic molecules because it often leads to more stable drugs that resist breakdown in the body.
What It Means for Research and Industry
Working with 2,4-difluorobenzaldehyde also raises important process questions. These compounds take skill to make, purify, and deploy safely. Industry and academia are always sharing better methods for synthesis—using greener reagents, improving reaction yields, and cutting back on hazardous waste. This ongoing search for better chemistry not only keeps people safe in the lab but also makes these innovations viable for industry to scale up.
The journey from simple benzaldehyde to its difluorinated cousin shows how much thought goes into what seems (at first) like a minor change. Behind each formula sits a host of decisions—about health, safety, environmental impact, and scientific progress. Learning to interpret formulas like C7H4F2O isn’t just textbook exercise. It opens up a deeper understanding of why chemists keep searching for better molecules and better methods, year after year.
Why Proper Handling Matters
Working with chemicals like 2,4-Difluorobenzaldehyde brings its own set of risks. Over the years, I've learned that giving a complex molecule the respect it deserves helps everyone go home healthy. This compound, used in pharmaceutical and organic synthesis, comes with hazards you can’t spot just by looking at a crystal or a bottle. Health risks, fire hazards, and environmental concerns all lurk if habits get sloppy.
Basic Precautions: Don’t Cut Corners
Lab veterans know that personal protective equipment counts for a lot. Safety goggles, a proper lab coat, and nitrile gloves aren’t fashion statements—they keep splashes and accidental contact from turning a minor job into a medical emergency. 2,4-Difluorobenzaldehyde can irritate the skin, eyes, and airways. Once, I saw a colleague rub his eye after handling a diluted aromatic aldehyde mixture—his day didn’t go as planned after that.
Ventilation matters just as much. Fume hoods pull vapors away from your face and lungs. Breathing in even a trace amount of this compound’s vapors can provoke coughing or worse. Keeping containers tightly closed, never sniffing directly, and avoiding open air transfers can seem like small matters, but the body remembers the times you slipped up.
Don’t Ignore Storage and Spill Risks
Chemical storage isn’t just about shelf space; it’s about making sure incompatible substances don’t mix in a way that spells disaster. 2,4-Difluorobenzaldehyde needs a dry, cool spot, away from anything combustible. Mixing up storage means risking fire or toxic byproducts. Reading those small hazard symbols on the container isn’t just bureaucracy—those pictograms are hard-won lessons from past accidents in labs and factories.
If you spill it, reaching for any cloth or towel won’t do. Absorbent pads made for chemical spills, combined with prompt cleaning, protect more than just the floor. Chemical exposure from a botched cleanup can leave long-lasting effects on skin or lungs. Whenever the lab I worked in skipped dedicated spill kits, we regretted it—standard paper towels just spread hazards around.
Know the Signs of Trouble
If splashes happen or vapors escape, you’ll know—skin reddening, watery eyes, or coughing start fast. Anyone feeling these symptoms needs clean water to rinse exposed skin or eyes, and a path to fresh air, not the typical “wait and see” approach. Rushing to a safety shower or eyewash station can feel dramatic, but dramatic beats careless.
Training and Attitude Save More Than Just Experiments
Training makes a difference. Safety practices I picked up from experienced chemists stuck with me long after university ended. Reading safety data sheets, understanding first aid steps, and practicing chemical respect can keep you out of the ER. The best labs foster a culture where watching out for coworkers and not skipping steps become habits—not just boxes checked on forms.
Looking Forward
Better labeling, ongoing training, and honest conversations about past mistakes go further than any rulebook. No one expects an accident, but preparation and attention make them rare. Locking up hazardous chemicals, wearing protective gear, and clean habits make chemistry a lot safer. Handling 2,4-Difluorobenzaldehyde isn’t about fear, but about never assuming you’re immune to the risks. Discipline keeps science moving—and people safe.
Understanding Beyond the Decimal Point
Every small detail counts in chemistry, right down to the last decimal of a molecule’s weight. The molecular weight of 2,4-Difluorobenzaldehyde is 142.1 grams per mole. No, this isn’t just a trivial figure reserved for textbooks or bored lab assistants. It stands as a cornerstone for folks involved in synthesis, analysis, or regulatory affairs. At the bench, it tells us how much of a substance we’re actually working with and helps keep scales out of the red zone. In the classroom, students can’t solve stoichiometry problems without this key number.
Daily Work, Real Impact
Organic chemists like to think in terms of reactions: reactant in, product out. I remember juggling aromatic reagents in a university lab, thinking a gram here or there wouldn’t change much. Wrong. A miscalculation, even by a tenth of a gram, means a smaller yield or a failed assay. If a researcher plans on preparing a hundred milligrams of some downstream product, they turn to the molecular weight of intermediates such as 2,4-Difluorobenzaldehyde. They calculate moles, doses, and solutions based on this number. It isn’t just a dry calculation; it guarantees reproducibility and safety.
Drug design and materials science also rely on the basics. Imagine building a Lego set and discovering some pieces don’t fit the printed instructions. The wrong molecular weight on paper can translate to the wrong amount of active ingredient in a batch, or an unexpected impurity. For regulatory compliance, accurate reporting is non-negotiable, both in the lab notebook and in the industrial QC room. Analysts expect reference compounds like 2,4-Difluorobenzaldehyde to have solid, clear data for each label and database entry.
Supporting Science with Experience and Evidence
I’ve seen junior chemists stick to “ballpark” figures when preparing their solutions. They wind up with inconsistent results, confused by the mismatch between expected and observed yields. When you honor real atomic weights, reactions show predictable results. Fluorine, for instance, isn’t a lightweight; swapping hydrogen for fluorine atoms bumps up the mass more than some realize. The extra data point is not just another entry in a spreadsheet. It’s proof of careful planning, respect for method, and foundation for every rigorous study.
Getting molecular weights right isn’t about gatekeeping or adding obstacles. It’s about building a reliable process. This doesn’t just help researchers in large facilities, but schoolteachers, hobbyists, and environmental monitors tracking what’s in water or soil. Precision in calculation prevents wasted reagents, saves money, and guards against accidental exposure. If that sounds basic, it’s because fundamentals form the backbone of discovery and safety alike.
Moving Toward Better Practice
Teaching and learning about molecular weight starts in high school labs and ends up supporting advanced research. Breaking down the reason for its accuracy leaves a lasting impression. Chemistry teachers can use simple substances like 2,4-Difluorobenzaldehyde to show why each atom matters. Lab managers can cut down on mistakes by insisting on clear, up-to-date reference tables and double-checking calculations before a pipette ever leaves the drawer.
With more open access to information and better digital tools, fewer slip through the cracks. Automating some calculations reduces human error, but understanding the importance of each number makes a real difference. Molecular weights like that of 2,4-Difluorobenzaldehyde aren’t trivia—they’re part of responsible science. Every accurate result begins with the right numbers, matched up with real-world knowledge and hands-on care.
Real Risks, Real Responsibility
Not every chemical raising eyebrows in the lab deserves respect for the same reasons. 2,4-Difluorobenzaldehyde—a compound relying on its two fluorine atoms and an aldehyde group—isn’t one that’ll bleed headlines with wild danger. Still, it teaches plenty about responsibility in science and safety.
I’ve come across plenty of chemicals that just seem to “exist” on a shelf. Yet, 2,4-Difluorobenzaldehyde stands out for more subtle reasons. It practically dares you to overlook the basics, threatening to turn convenience into an accident with just a bit of neglect. Like many organic compounds, it brings flammability, volatility, and reactivity to the table—none of which forgive carelessness.
Keep It Cool, Keep It Tight
Storing this compound gets easier when you treat it the same way you’d treat common solvents like acetone or toluene. That means a cool, dry place away from sunlight. Let the temperature creep, and vapor pressure becomes a problem—one that escalates quickly if it's anywhere near any sources of ignition.
Sealing the bottle tightly does more than keep out air; it prevents oxidation and wards off evaporation. A leaky or loose cap signals open season for contamination, and in busy labs I've watched plenty of bottles become suddenly useless or dangerous just because someone rushed clean-up or capped without checking the seal.
Respect Reactive Tendencies
At first glance, the structure doesn’t look fearsome, but that carbonyl group remains reactive. Placing this bottle anywhere close to oxidizers, acids, or bases ignores basic chemical sense. Cross-contamination turns a mundane afternoon into a frantic one. Storage in a flammable cabinet—and not with acidic or basic chemicals—provides a basic layer of safety.
In my years with chemical collections ranging from the simple to the exotic, emergencies rarely creep up from the “big scary” bottles. It’s the ignored, forgotten organic flasks—sometimes old, sometimes not labeled properly, sometimes stored at the wrong temperature—that trigger frantic scrambles for ventilation and damage control.
Labels, Dates, and Accountability
Slapping a simple label on a bottle does wonders. Include the name—written legibly—the date it entered the storage shelf, and a clear warning about its nature. I’ve seen junior scientists discard bottles simply because they felt uneasy about a half-effaced sticker or a missing date. It ends up wasting money and, more importantly, time.
Routine checks don’t just protect the people in the lab. They keep your workflow smooth and the chemical budget in check. If someone finds a bottle close to its expiration, it’s easier to dispose of than to scramble for answers after a mishap.
The Big Picture: Culture of Safety
Creating a strong culture of chemical stewardship beats any written guideline. Training goes a long way, but you only see results when everyone buys in. 2,4-Difluorobenzaldehyde doesn’t care if you’ve cut corners before and got away with it. Consistent procedure wins the day, each time.
Professional organizations, like the American Chemical Society, recommend detailed code for chemical storage because mistakes hit hardest when they’re preventable. Following these guidelines means fewer emergencies, greater confidence in the lab, and a clear sign of respect for both the science and those who practice it.
In the end, storing 2,4-Difluorobenzaldehyde properly says more about a lab’s priorities than any fancy equipment or certificate ever could.