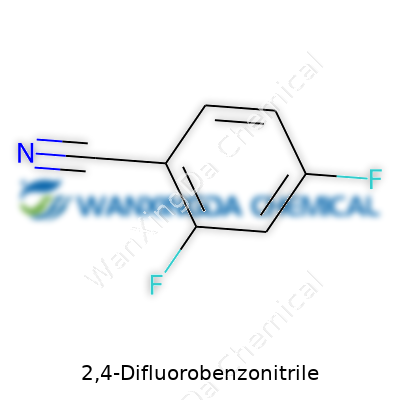
2,4-Difluorobenzonitrile: A Close Look at Progress, Challenges, and Promise
Historical Development
The arc of 2,4-difluorobenzonitrile’s story reflects broader themes across chemical history. Early on, chemists only toyed with partial fluorination of aromatic compounds out of academic curiosity. The technical hurdles loomed large. Consistent synthesis sees breakthroughs around the late twentieth century, thanks to sharper reagents and a surge in demand for custom fluorinated building blocks. I remember paging through old journals, spotting 2,4-difluorobenzonitrile mostly as a footnote. Today, it walks tall across synthetic and materials chemistry. This growth tracks the rising appetite for fluorinated molecules in both drug development and the world of advanced electronics.
Product Overview
At the heart of its popularity, 2,4-difluorobenzonitrile offers a straightforward structure: a benzene ring, a nitrile at the 1-position, fluorine atoms tagging positions 2 and 4. An experienced chemist doesn’t have to look twice. The cyano group unlocks all kinds of downstream modifications, and the fluorines shore up chemical stability. This blend isn’t common, even among other aromatic nitriles, and shapes where the compound gets used. Both the electronics sector and modern pharmaceutical research find a kind of workhorse in this molecule—robust enough for demanding processes but reactive enough to meet innovation head-on.
Physical & Chemical Properties
A sample of 2,4-difluorobenzonitrile usually appears as a crystalline solid with a pale hue. Its melting point holds steady within a moderate range, which smooths out handling. The compound isn’t especially prone to humidity or air-driven decay and stores well at room temperature. Its solubility profile acts as a practical advantage, too: reasonable in organic solvents, stubbornly low in water, a trait that tracks with much of its chemical family. From a reactivity standpoint, the activated aromatic ring couples readily with nucleophiles, while its fluorines have a kind of stubbornness that many synthetic chemists appreciate for selectivity under harsh conditions. Years of hands-on bench work bear out these details: the edge that fluorine brings continues to guide new project designs.
Technical Specifications & Labeling
Technical data on 2,4-difluorobenzonitrile isn’t just about purity or assay numbers, although such details shape performance in sensitive areas like drug synthesis. Analytical chemists tend to check for common impurities such as unreacted fluorobenzonitriles or related isomers, often through gas chromatography or HPLC. Many production batches flag a purity of over 98%, not so much out of marketing concerns as to avoid tricky downstream purifications. Accurate chemical labeling protects both the operator and the end-use—inaccurate data stalls more than one pilot run. Over the years, regulatory agencies have nudged producers to meet clearer hazard labels and harmonized standards. Despite the paperwork grind, this makes a difference for anyone who spends time on the production floor.
Preparation Method
Several industrial-scale methods for preparing 2,4-difluorobenzonitrile echo classic nitrile chemistry, using nucleophilic aromatic substitution or controlled Sandmeyer-type reactions. One common approach starts with 2,4-difluorobenzaldehyde or similar precursors, then deftly introduces a cyano group. This gets tricky—the wrong sequence loses both yield and selectivity, a fact reinforced by many years and many wasted reagents. Advances in catalysis have cleaned up a few legacy steps, swapping toxic copper salts for friendlier alternatives. Experienced process chemists don’t just chase yield. They worry about scalable safety, waste, and the unpredictable nature of large-scale exotherms. Over time, process control measures have dialed in cost and reliability, making the compound widely available even to smaller research labs.
Chemical Reactions & Modifications
2,4-Difluorobenzonitrile doesn’t just sit on a shelf—it fuels chemists’ creativity. The nitrile group acts as a launch pad, quickly transforming into amides, amines, or acids. Given fluorine’s high electronegativity, the aromatic ring resists unwanted side reactions, especially at high temperature or in strongly basic media. Medicinal chemists lean on this property: modifications keep the core structure intact while building out molecular diversity. Cross-coupling reactions using palladium or nickel catalysts have only grown more reliable, and those fluorines open up selective halogen-lithium exchange. Each new method deepens the compound’s toolbox and extends its reach into new branches of material science and drug research.
Synonyms & Product Names
Across suppliers and journals, 2,4-difluorobenzonitrile goes by several monikers: you might also see it as 2,4-difluorobenzene carbonitrile, 2,4-DFBN, or 1-cyano-2,4-difluorobenzene. Such naming diversity, while a headache for catalog searches, signals how common and well-studied the compound has become. Anybody slogging through chemistry literature runs into this tangle—picking up synonyms saves wasted time and missed connections between academic and industrial teams.
Safety & Operational Standards
Nobody wants to underestimate the risks with aromatic nitriles. Years of safety bulletins and lab anecdotes have built up a shared memory bank: always treat skin and eye contact seriously, use gloves, and make sure good ventilation keeps inhalation exposure low. Nitriles in general have a reputation for toxicity, often due to cyanide formation under metabolic stress. While 2,4-difluorobenzonitrile isn’t among the most acutely hazardous, giving it respect in storage and handling pays off. Regulatory standards in both the EU and North America require clear hazard pictograms and careful disposal guidelines, pushing academic and industrial users to keep best practices front and center. Investing in robust training made a difference in my own lab—not just for avoiding fines or review notices, but for building a genuine safety culture.
Application Area
What has unlocked the most value for 2,4-difluorobenzonitrile? Pharmaceutical discovery teams often use it as a building block for new drug candidates, especially where metabolic stability must rise beyond what classic aromatics allow. Its resistance to oxidative metabolism, driven by those fluorines, keeps molecules from breaking down too quickly in the body. Agrochemical researchers turn to it for similar reasons, as do makers of dyes and specialty materials for electronics. Experience has shown that the compound’s chemical stamina lets it weather tough processing steps where less robust analogs would falter. One of its lesser-known uses appears in liquid crystal development, where consistent alignment and thermal stability prove crucial. Each industry draws on a slightly different facet of the molecule, but its adaptability keeps opening new doors.
Research & Development
Open any recent research digest, and 2,4-difluorobenzonitrile pops up in target-oriented synthesis, especially for complex heterocycles. The field’s been exploding with cross-coupling methods and late-stage functionalizations, with this molecule often picked as a test case for robustness. Medicinal chemists value it for the way fluorination alters bioactivity—a small substitution sometimes flips a molecule’s fate in vivo. R&D groups at academic institutes and pharmaceutical firms continue publishing novel applications, often sharing data on metabolic stability, binding affinity, or physicochemical tweaks that stem directly from the benzene core. My own work touched on this area in designing enzyme-resistant inhibitors, where fluorinated nitriles led us to candidates with longer biological half-lives. Journals have documented a clear link between added fluorines and enhanced in vitro results. Every innovation cycle builds on those gains, drawing new utility out of an old scaffold.
Toxicity Research
Any widespread industrial compound eventually passes through sharp toxicological assessment—regulatory agencies, corporate safety teams, and contract labs all weigh in. Nitriles as a class demand respect due to the risk of cyanide release upon enzymatic breakdown in biological systems. Rodent studies with 2,4-difluorobenzonitrile have sketched out a profile of moderate acute toxicity, with symptoms tracking cyanosis and respiratory distress at high doses. Chronic toxicity data remain sparse, and researchers call for more work. Cell studies suggest that additional fluorines complicate metabolic pathways, sometimes helping, other times raising new questions about distribution in fatty tissues. Toxicologists keep an eye on both occupational exposure over time and downstream metabolites. Sharper analytical methods—particularly LC-MS and headspace analysis—have improved exposure monitoring. General experience cautions against casual contact. Thoughtful planning and up-to-date safety equipment matter, especially in scale-up and waste management.
Future Prospects
Markets tied to electronic materials, high-stability pharmaceuticals, and advanced polymers fuel ongoing interest in 2,4-difluorobenzonitrile. Demand continues to rise as new drug programs filter promising fluorinated scaffolds for improved metabolic profiles. At the same time, sustainability pressures push R&D teams to examine safer, greener synthesis and waste reduction. Renewed investment in catalysis could reduce the environmental burden of legacy nitrilation processes. My colleagues across both academia and industry reflect a similar optimism: careful tuning of fluorination levels promises finer control over molecular function. Improving access to greener precursors and off-the-shelf building blocks can lower barriers for small labs to innovate responsibly. The road ahead will keep challenging producers to meet tighter regulations, but the need for versatile, sturdy aromatic nitriles remains strong. Watching this molecule’s journey underscores just how much thoughtful chemistry shapes not just new materials, but safer workplaces and healthier communities.
The name 2,4-Difluorobenzonitrile might only ring a bell if you’ve spent time around chemical plants or research labs, but this small molecule can play a big role behind the scenes. This isn’t the kind of compound that makes headlines often, but its impact shows up in places the average person hardly ever thinks about. I first learned about this stuff in college during a project on how specialty chemicals lay the groundwork for entire industries, and it changed how I see things in a grocery store or at the pharmacy.
How Chemistry Shapes Solutions
2,4-Difluorobenzonitrile starts out as just another building block in the hands of chemists. Its ring-shaped structure, decorated with two fluorines and a nitrile group, offers more than clever naming. Those groups open doors for transforming it into something entirely new. Most often, this molecule ends up serving as a starting point for making more complex chemicals in fields like pharmaceuticals and crop protection.
Walk down any pharmacy aisle and you’ll probably pass by products born out of intricate synthesis routes involving chemicals just like this one. Chemists rely on 2,4-Difluorobenzonitrile to craft advanced intermediates, linking smaller molecules into treatments that end up tackling diseases or easing symptoms. In the process, the original molecule vanishes, but its role as a launchpad was crucial. It’s not flashy, but the workhorses of chemistry rarely are.
The Agricultural Connection
Farms and fields present an entirely different story. 2,4-Difluorobenzonitrile helps in the creation of herbicides and fungicides. The molecule’s reactive points make it a popular choice for constructing compounds that stubborn weeds or harmful fungi just can’t tolerate. This arms farmers with the tools they use to raise healthy crops more reliably, even when weather or pests conspire against them. Many folks don’t realize that this chain—tiny molecules in a chemist’s flask, crops in the field, food at the market—is what keeps global food supplies humming along.
Safety and Sustainability Matter
All these uses come with some responsibility. Production and handling of chemicals like 2,4-Difluorobenzonitrile require real care since it poses hazards if mismanaged. Attention to workplace safety and environmental impact isn’t just a regulatory box to check. In my experience on the safety committee at a research lab, nothing slows down a project like an accident. This underscores why chemical manufacturers keep looking for ways to minimize waste and switch to greener synthesis routes, sometimes turning to continuous-flow chemistry or biocatalysts to limit risks and waste.
Looking Forward
The growing demand for specialty chemicals pushes innovation on all fronts—not just discovery, but also how we make and handle these ingredients. New processes aim to cut down on toxic byproducts and energy use, making the field safer for workers and the planet. Researchers continue searching for even milder ways of transforming simple molecules into medicines and crop protectants. Chemistry doesn’t often feel personal, but the quiet progress shaping these processes impacts everyone—from the researchers in the lab, to the kids eating fruits and vegetables that relied on careful crop protection.
A Closer Look at the Chemical Formula
2,4-Difluorobenzonitrile goes by the formula C7H3F2N. At first glance, it’s just letters and numbers, but there’s a lot packed into those eight characters. The molecule tells its own story: a benzene ring showing off a nitrile group and a pair of fluorine atoms arranged with surgical precision at the 2 and 4 positions. That’s not just chemistry trivia—placement like this often shifts a substance’s behavior in the lab and the field. Anyone who’s done hands-on chemistry can tell you, move a substituent around and suddenly the molecule acts like a whole different creature.
The Weight Behind Fluorinated Compounds
Fluorine demands respect. Slip two of these atoms onto a benzene ring and you’re suddenly in the realm of potent agrochemicals, specialty polymers, or advanced electronics. The presence of a nitrile group (–CN) brings its own set of traits. Nitriles channel reactivity in specific ways, making them attractive for building more complicated molecules. This combo—fluorines and nitrile—shows up in targets for drug discovery, pesticide R&D, and material science.
In research labs, the drive to find better, safer, and more selective compounds pushes chemists toward structures like 2,4-difluorobenzonitrile. They look for molecules that last long enough in use, but don’t stick around where they’re not wanted. Fluorinated chemicals sometimes walk a fine line. According to the Environmental Protection Agency, some perfluorinated compounds build up in water and living tissue. It becomes crucial to design molecules with environmental fate in mind, especially as regulations tighten worldwide.
Practical Considerations
For those who work in chemical synthesis, getting reliable access to pure C7H3F2N means choosing routes that balance cost, reliability, and safety. Handling volatile organics and strong acids doesn’t get easier with time, but it pays to develop routines that safeguard health without sacrificing the bottom line. Chemists with years at the bench learn to respect every step along the path—one bad decision, and it’s not just a failed reaction, it could be an accident that costs precious time and material.
Potential Solutions and New Directions
Facing the challenges of persistent chemicals and safe manufacturing, more teams have been turning to green chemistry. This doesn’t just mean using less solvent or switching to “friendly” reagents. It’s about adapting processes to cut waste and energy use, while keeping all the crucial properties. Techniques like microwave-assisted synthesis, selective catalysis, and continuous-flow manufacturing pick up steam as chemists refuse to accept that environmental stewardship comes at a scientific cost.
Industry and academia benefit from sharing what works—and what really doesn’t. Open publication, data sharing, and honest reporting will push innovation forward, no matter where the next big breakthrough pops up. The push for safer, smarter chemistry isn’t only about ideals. It’s about practical progress, day by day, one molecule at a time.
Stepping into any lab usually takes me back to my first years of hands-on work—fumbling with glassware, navigating chemical smells, and always triple-checking labels. 2,4-Difluorobenzonitrile falls into that category of chemicals that demand respect. It's a simple-looking powder at first glance, but its properties can trip up even seasoned researchers. This is a potent irritant, and carelessness invites problems ranging from skin burns to breathing issues. These aren't scare stories; I’ve seen peer-reviewed evidence and unkind news articles when folks cut corners.
Fluorinated aromatics deserve extra scrutiny. 2,4-Difluorobenzonitrile carries a dual punch—it contains both nitrile and fluorine groups. Exposure through skin or inhalation isn't just uncomfortable; effects stick around for days if you’re unlucky. Once I brushed some of it off a countertop without gloves and spent the next few hours regretting it as the skin stung and reddened. Gloves—nitrile or neoprene, not latex—are non-negotiable. Old habits like bare-handed pipetting don’t make sense in this context. Forgetting splash goggles in a hurry can mean eye redness and pain, and there aren’t shortcuts around this.
Ventilation proves its worth time and again. On days when the fume hood gets cranky, it's tempting to proceed regardless, yet this is where problems start. 2,4-Difluorobenzonitrile releases irritant dust and fumes. Fume hoods are the real heroes; they keep any airborne threat out of your lungs and off your skin. Taking shortcuts often backfires—respiratory irritation can linger long past cleanup. Masks rated for organic vapors work too, but hoods remain the gold standard. Careless spills on benchtops can linger as invisible threats, sticking to hands, sleeves, even the odd notebook. Regular surface wipes and deliberate, measured working styles keep the environment manageable.
Friends outside chemistry sometimes assume a quick hand wash is enough if something splashes. Not true—the water alone rarely removes chemicals like this one from skin. Soap and plenty of rinsing are necessary, and still, the affected area requires monitoring. Some studies say 2,4-Difluorobenzonitrile can penetrate skin over time, which means rinsing right away is the only smart choice. For eye contact, those old wall-mounted eye washes get their time to shine. No one boasts about spending fifteen minutes flushing out their eyes, yet it's better than a chemical burn.
Disposal brings its own headaches. I’ve seen the messes that come from pouring organic compounds down a drain or letting them pile up in shared waste bottles. Lab protocols exist for a reason. Treat 2,4-Difluorobenzonitrile waste with the same discipline as live reagents. Collection in compatible, properly labeled containers remains the right way forward. Hoping the next shift will “take care of it” never ends well. In my experience, clear communication between staff and regular safety audits keep things honest.
At the end of the day, the story with 2,4-Difluorobenzonitrile is the same as countless lab hazards. Familiarity tempts people to grow sloppy, but stories from old colleagues and a few close calls of my own serve as reminders: risk never leaves the room. Respect for the chemical, respect for the process, and respect for your colleagues form the backbone of a safe lab. This is what keeps the thrill of discovery from landing anyone in the emergency room.
Understanding Chemical Temperatures Isn’t Just for the Lab
Most folks outside of chemistry circles don’t spend much time thinking about melting points. For the labs mixing up new compounds, or anyone in charge of a chemical process, the melting point of a substance like 2,4-Difluorobenzonitrile makes a real difference. This stuff melts between 44 and 48°C. Being just above room temperature, that’s warm enough you can melt it in your hand, but not so high you’ll need a massive industrial furnace.
That range matters. Not every chemical changes state at one single, crisp temperature. People in research and production need to keep an eye on that band: too wide, and you might have impurities or some issue with your sample. A sharp, clear melting point means things are pure and you’re on the right track. Impurities drag that melting point down, sometimes stretching it out over several degrees, which tells you something’s off.
Why Melting Point Matters in Everyday Chemistry
Melting points seem dry until we remember how they impact reliability and safety in real life. In pharmaceuticals, crop chemicals, electronics, or anything pushing out products, this little number on the data sheet decides storage costs and transport headaches. For anyone who’s ever stopped to worry about a warehouse heating up in the summer, knowing if your material turns to sludge at 40°C, or stays solid up to 150°C, isn’t trivia — it’s basic operations.
Let’s say you’re shipping bulk 2,4-Difluorobenzonitrile. A poorly insulated truck, or even a temporary heat wave, and that white crystalline solid could seep out of the bags. Now you’ve got a mess, lost cash, and possibly safety concerns if fumes get out. One bad batch caused by high humidity or a tainted raw material? You’ll see it as a sloppy, broad melting point.
Quality, Purity, and Trust
A trustworthy melting point also signals that the chemical you think you’ve got is truly what’s in the bag or bottle. In my own experience running undergraduate chemistry analyses, those low, smeared melting points always meant trouble. Maybe we left a bit too much solvent behind, or contamination came from careless glassware cleaning. Fixing it sharpened our data — and, more importantly, meant we understood what went wrong and how to keep it from happening again.
Manufacturers test for melting point every batch. That’s not bureaucracy — that’s protecting investments, reputations, and downstream users. If a drug maker starts with 2,4-Difluorobenzonitrile, but the melting point’s off, the chain reaction can mess up months of work.
Steps Toward Better Practice
Getting the melting point right isn’t magic. It starts with pure starting materials and equipment that’s been properly maintained and calibrated. Careful attention to solvent removal, batch-to-batch documentation, and cross-checking results builds a feedback loop. Peer review and sharing data across research groups chip away at mistakes and move the bar higher. Recording experimental details—like who ran the test, with what thermometer, in what room—helps nail down quirks before they cause bigger issues.
Melting point isn’t just another number. It’s proof that chemistry’s working as it should, and that trust, safety, and genuine know-how shape every step from the smallest academic bench to the largest chemical producer.
A Chemical With Real Risks
In the world of chemical storage, few people outside the lab pay attention to something like 2,4-Difluorobenzonitrile. For those working with it, the risk is real. This compound packs all the dangers of cyanide chemistry with the added volatility of aromatic solvents. Safety can slip through the cracks, especially in pressure-packed research environments. That’s why I find the conversation about safe handling so important.
I still remember my first year running a stockroom. We’d get shipments of all kinds—some bottles left sweating on pallets next to heating vents, names half-smeared by the elements. That moment taught me more about chemical safety than years of lectures. Even a relatively small mishap can threaten people and property, especially with volatile or toxic materials.
Why Storage Temperature and Ventilation Change Everything
The first rule in my experience with chemicals like 2,4-Difluorobenzonitrile? Keep it cool and dry. This isn’t just about “following protocol.” Excess heat and humidity spark all sorts of ugly problems. 2,4-Difluorobenzonitrile doesn’t belong anywhere near sources of heat, sunlight, or strong oxidizers. The compound holds up best under consistent, room-temperature conditions in a dark spot. Some labs spring for flammable storage cabinets with exhaust fans to keep ambient fumes at bay. I’ve seen coworkers ignore the need for good airflow, only for headaches and complaints to start piling up from the staff.
Chemicals like this sometimes waft a sharp or irritating odor. Ignore that, and you end up with a bigger problem. I once walked into a back closet where bottles sat open, flavors of solvent drifting out. Entire projects delayed because of carelessness. This is what staying alert to smell and signage does for a team—it stops accidents before they become emergencies.
Proper Containment Is Not Just a Rule—It’s Good Sense
People who don’t work with toxic organic chemicals every day might not picture just how much depends on storage. 2,4-Difluorobenzonitrile comes in tight-sealing glass or fluoropolymer bottles, never in metal or reactive plastics. Leaks might mean not just lost material but the kind of lingering contamination that’s impossible to clean. Never once did I see a professional take shortcuts in double-bagging or using secondary containment trays, especially when drips quickly corrode surfaces.
Labeling matters. A clear label, times two, with a fresh date and hazard warning, always beats a scribble on tape. I have seen near misses over ambiguous labels—what seems obvious one day becomes a puzzle the next, especially with abbreviations or a smudge from a glove.
Prevention Beats Accident Reports
Some folks think the biggest risk with chemical storage comes from big accidents—fires, explosions, hazmat suits and klaxons. In reality, the slow creep of vapor, the unseen reactions with traces of moisture or dust, those pose greater threats day to day. I’ve seen resource-strapped labs put chemical stock in old refrigerators, only for broken seals and rising temperatures to turn a manageable risk into a ticking clock.
Insurance premiums only tell half the story. The human factor makes organizational vigilance vital. Frequent checks—weekly if not daily—catch early warning signs: swelling caps, residue, fading labels. If there’s anything I learned double-checking every shelf and every bottle, it’s that nothing beats showing respect for your workspace. Good habits do what panic plans rarely can—keep people safe, and keep work moving forward.