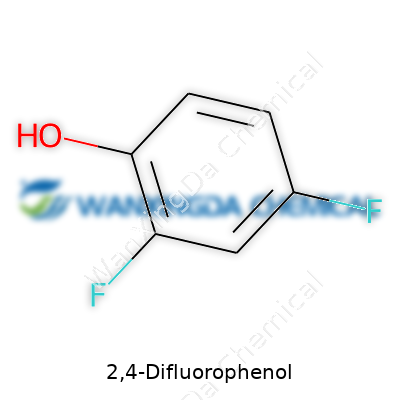
2,4-Difluorophenol: A Close Look at a Quietly Crucial Chemical
Historical Development
2,4-Difluorophenol emerged from the fertile grounds of 20th-century organic chemistry, a time when scientists were pushing boundaries by introducing new elements into aromatic rings. The interest in phenolic compounds runs deep, especially when those phenols bear halogen substitutions. Chemists began swapping out hydrogen atoms for fluorine on benzene rings, motivated by the chase for altered reactivity and biological intrigue. Adding fluorine often imparts considerable stability and changes how molecules interact with living systems, which makes fluorinated phenols worth more than a casual glance. Research dating back several decades showed that specific patterns of fluorination on phenol backbones changed chemical behavior and opened up new doors in both industrial chemistry and medical exploration.
Product Overview
2,4-Difluorophenol occupies a spot in the toolkit of anyone who works in medicinal chemistry, agrochemical research, or material synthesis. It’s not a household name, but it sits where important things happen. This compound finds use as a building block for a range of more complex molecules. Labs and pilot plants rely on its versatility, both for direct applications and as an intermediate that can anchor further chemical transformation.
Physical & Chemical Properties
This compound stands out with a pungent aroma, a clear to slightly yellow appearance, and a melting point low enough to keep it liquid under typical lab conditions. Its boiling point strikes a balance that lets chemists distill it without excessive trouble. The two fluorine atoms lying at positions 2 and 4 on the phenol ring push its reactivity away from that of regular phenol, making it less acidic and adjusting its electron density. Its solubility suits certain nonpolar organic solvents, but it doesn’t mix well with water, thanks to the influence of the fluorine atoms. These features give it a predictable but intriguing chemical personality that researchers quickly learn to recognize.
Technical Specifications & Labeling
You won’t find 2,4-Difluorophenol overflowing in standard supply catalogs, but it’s well represented in specialized chemical inventories. The labeling highlights its purity—typically upwards of 97 percent for use in synthesis or analytical processes. Technical documents point out storage instructions to avoid light and moisture, both of which can nudge the compound into unwanted side reactions or degrade its integrity. Batch-specific spectral data, including NMR and IR, help chemists confirm its exact identity before any project kicks off.
Preparation Method
Synthesizing 2,4-Difluorophenol calls for finesse and a solid understanding of aromatic substitution. Chemists often start with a suitably substituted difluorobenzene as a precursor, turning to hydroxylation to insert the phenol group at just the right position. Some opt for more intricate fluorination of phenol using selective reagents, but direct fluorination tends to overdo things due to fluorine’s aggressive nature. Catalysts, temperature control, and clever choice of reagents determine how well the reaction goes. Getting the two fluorines in the 2 and 4 positions, rather than elsewhere, matters quite a bit, and the selectivity of the route separates mediocre yields from the methods that scale for commercial synthesis.
Chemical Reactions & Modifications
Once in hand, 2,4-Difluorophenol provides an exciting platform for further transformation. Reactivity tests show that its hydroxyl group remains active for etherification or esterification, letting researchers build larger molecules atop its scaffold. The electron-withdrawing power of fluorine at the ortho and para positions tunes the ring toward specific substitutions, altering everything from rates to product distribution. Nucleophilic aromatic substitution steps in for those hoping to swap out the fluorines for bulkier groups—a trick that can spark new classes of derivatives. In my own academic work, I saw how just a few tweaks here changed everything about a compound’s effectiveness in test reactions.
Synonyms & Product Names
Common synonyms pop up in literature and labels alike. References to “2,4-DFP” or “2,4-Difluorohydroxybenzene” streamline searches. Chemists seeking clarity know to include those alternate names in database queries, reducing the frustration of missing a critical publication or data sheet during research sweeps.
Safety & Operational Standards
Working with 2,4-Difluorophenol requires the same respect you’d show any reactive organic compound, especially those with volatile phenols or halogenated rings. Safety data sheets flag its irritant properties—eye, skin, and respiratory tract all rank as potential targets if you handle it without gloves or fume extraction. I recall the memorable sting of chlorophenols during graduate work, and difluorophenol’s precautions repeat these same lessons: strong ventilation, careful pipetting, and strict avoidance of direct contact. Proper waste disposal becomes more than a checklist item—a commitment to health and environmental protection. Academic and industrial audits both stress the importance of controlled storage, spill containment, and clear labelling to block avoidable mishaps.
Application Area
You find 2,4-Difluorophenol featured in a grab bag of advanced chemistry projects. Medicinal chemists use its structural motif to create candidates for enzymes and receptor modulators, thanks to fluorine’s power to resist metabolic breakdown. Agrochemical developers explore its derivatives for pushing pest and weed management tools toward new selectivity and durability profiles. Material scientists, too, appreciate the way fluorinated phenolic compounds open pathways to high-performance polymers and coatings. My own introduction to fluorinated aromatics came when I watched a seasoned chemist win grant funding with a bunch of test tubes filled with derivatives from this very scaffold, each showing promising traits on the first screens.
Research & Development
R&D circles rarely ignore the possibilities of a new fluorinated compound, and 2,4-Difluorophenol draws serious attention in drug discovery and advanced material development. Ongoing studies morph its basic architecture to chase increased binding affinity or thermal resistance. Teams engineer it into experimental pharmaceuticals hoping to shift dose size or half-life. Academic labs publish papers on functionalized derivatives, reporting tweaks that yield new activity or physical traits. Instrumental analysis has grown sharper, allowing for minute detection and careful mechanistic studies that keep pushing the boundaries. Each time spectroscopy or chromatography evolves, researchers revisit known compounds and discover subtler applications.
Toxicity Research
Safety-conscious teams examine the toxicological implications of 2,4-Difluorophenol and its byproducts. Animal studies, in vitro cytotoxicity assays, and environmental breakdown experiments play a key role before any compound makes its way out of the lab. Early toxicity data for halogenated phenols show irritation remains a primary concern, as does the potential for longer-term organ impact if exposure occurs repeatedly. The need for caution comes not just from individual risk but also from the notion that environmental release could threaten aquatic life or soil health. Regulatory guidelines keep evolving, prompting industry to preemptively collect and publish responsible test results.
Future Prospects
Looking ahead, 2,4-Difluorophenol seems unlikely to fade from the chemist’s bench. Its basic blueprint offers enduring value as a springboard for ever more targeted chemistry. The hunger for new synthetic routes—be they cleaner, safer, or more selective—keeps interest strong. Technological improvements in fluorination or downstream modifications promise a steady march toward higher yields and lower waste. As industries keep tightening environmental and safety standards, ongoing research into non-toxic derivatives and better containment methods will mark progress worth watching. The future, if anything, calls for smarter use and sharper oversight—ensuring that the promise of novel fluorinated phenols turns into real benefits for human health and advanced technology, not new sources of risk.
Chemistry with a Purpose
Staring at the name 2,4-Difluorophenol, most people aren't going to think about its impact rolling through their daily lives or quietly influencing big changes in industry and medicine. Yet, for anyone who tunes into how chemistry shapes medicine, agriculture, and technology, understanding where this compound fits helps unravel a more complex story.
The Building Block Behind the Curtain
Talking to researchers and professionals who work with fine chemicals, I’ve found 2,4-Difluorophenol nestled into various synthetic processes. Its core appeal comes from its structure: having two fluorine atoms swapped into a phenol ring tweaks how the compound behaves and reacts. This little switch, sometimes, spells the difference for chemists needing a boost in a reaction’s selectivity or power. 2,4-Difluorophenol isn’t a blockbuster pharmaceutical or an ingredient you sprinkle on cereal. It acts as a building block—something chemists use to piece together much more complex molecules.
Why Its Value Isn’t Obvious
Some will ask why fluorinated phenols are needed at all. Here’s where it gets interesting. Fluorine rarely occurs naturally in living systems, but when smart chemists plug fluorine into organic molecules, the chemical properties can change in dramatic ways. Medical research shows that fluorinated drugs often last longer in the body or dodge enzymes that would quickly break them down. By designing a drug with a fluorine atom or two in just the right spot, researchers unlock new treatment options for diseases ranging from cancer to viral infections. 2,4-Difluorophenol enables this—furnishing a backbone that can be tailored in many directions, especially for pharmaceutical research and agrochemical development.
Connecting Research and Real Life
Having worked alongside scientists hunting for more effective drugs and better crop-protection chemicals, I’ve seen these tiny inputs make a big difference. 2,4-Difluorophenol often appears in journal articles and patent filings as an intermediate—a silent partner in the story, but a crucial step for reaching the final goal. Putting together a pain-relief medicine, for example, might lean on this compound as one of several rungs on the chemical ladder. Unlikely candidates like this shape progress quietly: without intermediates, none of the finished drugs or improved pesticides can happen.
Safety, Responsibility, and the Future
With compounds like 2,4-Difluorophenol, safety always demands attention. In the lab, chemists suit up and stick to tight safety protocols, since even a drop of phenol derivatives can irritate skin or worse. Plus, fluorinated chemicals present special disposal challenges, since some fluorine-containing molecules resist breaking down in nature. Environmental scientists and industry watchdogs are pushing for safer handling, tighter reporting, and greener routes to make and use these intermediates. As research moves forward, eco-friendlier methods for both manufacturing and disposal need more resources and support.
The Takeaway for Innovators
2,4-Difluorophenol doesn’t make headlines on its own, but for years, it’s played a subtle and essential role behind many chemical innovations. Staying informed about its uses—and the responsibilities that come with them—means respecting the science and people who carefully handle each step. By investing in more sustainable chemistry, present and future generations can tap into its power while keeping safety and stewardship at the core of progress.
Simple Molecules, Lasting Lessons
Diving into new organic molecules takes me back to dusty textbooks and glass vials bubbling away on the back bench. 2,4-Difluorophenol might sound like something you’d only mention in a quiz, but there’s more value in knowing its makeup than just acing a test. The molecular formula, C6H4F2O, gives much more than a countdown of atoms. It’s a gateway for understanding some bigger ideas that show up in labs, medicine, and even your local grocery store.
Fluorine’s Sneaky Impact
Fluorine is not just another periodic table entry. When stuck onto a phenol ring—like in 2,4-Difluorophenol—it gives the molecule new powers. This bond creates resistance to enzymes that would otherwise break down the compound fast. That’s one reason why you keep hearing about fluorinated molecules popping up among new drugs. A simple change like sticking two fluorine atoms at the 2 and 4 positions shifts the boiling point and changes how the ring interacts with other molecules. Medicinal chemists lean on formulas like C6H4F2O to build up libraries of new compounds, hoping something in the batch will fight infection or treat stubborn conditions.
Getting to the Bones of Structure
It’s easy to look at “C6H4F2O” and treat it like a secret password, but each number means something. Six carbons means a benzene core. Four hydrogens? That tells you something about how many spots have been swapped for something else—two of them, in this case, with fluorine. Oxygen marks it out as a phenol. This shorthand tells you what scientists have learned after years of trial and error. It means less guesswork in the lab, and fewer wasted materials when students set up their reactions.
The Chemistry Classroom, Without the Jargon
Teaching young chemists how to make and recognize molecular formulas helps everyone avoid mistakes. I remember a time when someone messed up an order, mixing up di- and tri-fluorophenol—one digit off and the reaction failed. It makes you respect the formula even more. Memorization counts for little unless you understand why the numbers change the molecule’s personality in a reaction or inside the body.
Better Science, Fewer Accidents
Lab safety relies on getting formulas right. Swapping a fluorinated phenol for another variant could mean unexpected fumes, tricky waste, or lost time. Industry chemists check and double-check every label, knowing that one atom’s difference means another round of safety paperwork. For students and researchers, fluency in formulas like C6H4F2O helps prevent these headaches and keeps labs less chaotic.
Supporting Better Discovery
So much comes back to simple chemical literacy. Recognizing the formula for 2,4-Difluorophenol might not get a headline, but it’s part of the unglamorous groundwork that leads to breakthroughs in greener chemistry, safer consumer products, and new medicines. Getting this step right sets the stage for responsible science and a smoother road ahead for anyone getting their hands dirty at the bench.
The moment people hear the name 2,4-difluorophenol, it rarely sparks much recognition. Compared to the toxic celebrities of the chemical world—cyanide, mercury, asbestos—this compound leads a quiet existence on paper and in labs. Yet for anyone who spends real time around chemicals, every unfamiliar name should prompt a gut-check: Do I know what I’m handling?
What Science Says About 2,4-Difluorophenol
2,4-Difluorophenol belongs to the family of fluorinated phenols, and right away, that raises some caution flags. Fluorinated organic compounds often pack serious biological punch. Take a close look at research and toxicity studies, and you’ll find that phenolic compounds tend to irritate the skin, eyes, and respiratory tract. The “difluoro-” modification just adds a twist—something chemists recognize as a potential source of both reactivity and increased absorption into biological tissues.
The Environmental Protection Agency and other regulatory agencies keep a watchful eye on phenols for a reason. These chemicals can disrupt enzymes and cause systemic toxicity. Even low-level exposure to related phenols causes issues ranging from mild irritation to fever, headache, and even kidney or liver damage after prolonged contact. Add the fluorine atoms, which make molecular breakdown in the body slower and boost bioaccumulation concerns, and you’re dealing with something that’s more than an everyday lab oddity.
Real-World Risks in Use and Handling
I’ve spent years in chemical research and manufacturing environments, and one thing stands out: nobody gets hurt when people respect the power of the chemicals they’re using. 2,4-Difluorophenol has a reputation among chemists for causing trouble if someone gets careless. Gloves, goggles, fume hoods—these aren’t just for decoration. A splash in the eye spells agony and possibly lasting injury. Skin contact leaves behind burns, not just a mild itch. Inhalation can trigger coughing fits and long-term lung problems. Accidents have happened with small spills that went ignored or when someone thought a dilute solution would pose no harm. The lesson repeats itself: treat this compound with real respect.
Environmental Impact and Disposal
Beyond the lab, the environment feels the impact of mishandled phenolic chemicals. Fluorinated organics resist breakdown—a property that lets them stick around for years, quietly building up in soil and water. This isn’t an idle worry. Even trace amounts of phenols have been tied to ecosystem disruptions, harming aquatic life and making their way through food webs. Improper disposal or accidental release can spark long-term health concerns for communities.
Responsible Action Makes the Difference
Chemical safety boils down to habit and discipline. Anyone who works with 2,4-difluorophenol needs to train for spills, storage, and emergency procedures—muscle memory prevents panic. Toolbox-ready facts matter more than technical jargon: don’t eat, drink, or touch your face around it. Use real chemical waste containers, never pour leftovers down the drain. Check labels, store it locked up, and don’t hand it to someone unless you know they understand the stakes.
Institutions, too, must push for continuous education, clear labelling, and regular audits. Lab supervisors set the tone, but staff who speak up and ask questions save lives. Every safety sheet and warning posted in break rooms exists for a reason—people only forget the rules when they haven’t felt the sting of ignoring them.
2,4-Difluorophenol won’t make headlines often. Still, its hazards remain real for those in contact with it, and so does the responsibility to treat it carefully. Chemical safety isn’t about fear; it’s about respect for what science has already proven—and learning from every close call.
Fluorinated chemicals always deserve a healthy amount of caution. Anyone who's spent time in a chemistry lab knows there’s no room for carelessness when it comes to handling materials like 2,4-Difluorophenol. Even a small distraction can cause a big mess, especially with liquids that mix issues of volatility, toxicity, and reactivity. Maybe you’ve noticed how quickly a compound without a solid storage plan can turn into a headache—both literally and figuratively. Luckily, there are some time-tested ways to sidestep trouble and keep this substance under control.
Temperature and Atmosphere Matter
Fluorinated phenols don’t love warm or humid conditions. Warmth speeds up their evaporation, turning the workplace air into something unpleasant fast. With a toxic compound like this, a leaky bottle on a sunny shelf will ruin any chemist’s day. Manufacturers around the globe warn against temperature swings and direct sunlight. Even if you work in a small research setup, a stable, cool spot—ideally in a chemical fridge or designated chemical cabinet—works best. Moisture in the air encourages slow hydrolysis, which doesn’t do anyone any favors, either.
Oxygen isn’t a friend to many specialty organics. Over time, oxygen can lead to slow degradation. Sealed glass bottles with tight-fitting caps cut down on air exchange, reduce risk, and help preserve purity. It’s important to label and date each container clearly, so you aren’t left guessing about the contents or their shelf life later. I’ve seen more than one “mystery bottle” create confusion, sometimes years after the original researcher moved on.
The Danger of Mixing and Spillage
2,4-Difluorophenol can react with strong bases and oxidizers. A splash into the wrong waste jug or an unmarked container invites disaster. Make sure every bottle lives apart from incompatible chemicals. When in doubt, separate acids from bases, oxidizers from organics, and volatile items from heat sources. I learned early on that even a humble piece of tape marking out storage zones in the lab can prevent ugly surprises.
Why Small Mistakes Add Up
So many health issues start with chronic, low-level exposure. Fluorinated aromatics have a way of sneaking into the air unnoticed. Without proper stoppers, any spill or outgassing lingers. People ignore the subtle odor at their own risk. Long-term exposure to phenols can trigger headaches, respiratory irritation, and worse. The EPA has flagged many fluorinated organic compounds for long-term monitoring due to persistence in the environment and links to chronic health impacts. This isn’t a case of being overly cautious; it’s basic respect for your own safety and that of others nearby.
Smart Practices Pay Off
Some of it is common sense: wear gloves, keep goggles handy, rethink that open bottle on the desk. Always use chemical-resistant secondary containers for extra protection, both to guard against drops and to contain leaks. If a bottle seems compromised, don’t “see how it goes.” Swap it out and update the inventory. Regular training for anyone with access—students, interns, even seasoned researchers—prevents complacency and keeps best practices fresh.
Safe storage of chemicals like 2,4-Difluorophenol doesn’t just protect productivity; it safeguards health and the environment. Personal vigilance saves lives and equipment, and thoughtful habits serve as the best defense against the unexpected. That old saying about ounce of prevention rings especially true here. Every smart move in chemical storage supports a safer, more efficient workspace for everyone who steps through the door.
Most folks never bump into 2,4-Difluorophenol during a regular day, but chemists and anyone involved in chemical manufacturing know it stands out. One look at its name tells you right off—it’s a phenol ring with two fluorine atoms at the 2 and 4 positions. This small tweak makes a big difference, especially compared to plain old phenol or even other difluoro-substituted phenols.
A Closer Look at Appearance and Behavior
In the lab, 2,4-Difluorophenol comes off as a colorless liquid or a transparent solid, depending on the temperature and purity. I’ve seen it pour with a watery consistency, much like ethanol or acetone. If you’ve worked with it, you know it gives off a sharp, biting odor—one that hints at both the phenolic core and those stubborn fluorines clinging to the ring. This is more than just trivia. The odor gives a warning about volatility and the need for good ventilation.
The boiling point provides practical insight into its handling. 2,4-Difluorophenol boils around 177 to 179 °C, which lands higher than a common solvent, but not out of range for distillation. That moderate boiling point highlights its use in organic reactions, where you want something with enough staying power to avoid vaporizing or breaking down too soon. Its melting point, sitting around 26 °C, means it slips between being a solid and liquid near room temperature, so storage in a cool, dry space makes sense.
Solubility and Chemical Attitude
I’ve watched chemists grumble about how poorly some fluoro-organics dissolve in water, but 2,4-Difluorophenol bucks that trend, at least partly. The phenol side tries to mix with water by making hydrogen bonds, while the fluorines pull in the opposite direction, trying to make it less friendly. In practice, it shows limited water solubility but dissolves much better in organic solvents like ether, alcohol, or chloroform. This duality shapes how folks in the pharma and agrochemical industries use and separate it: you pick your solvent carefully and don’t expect it to just rinse away with water.
Why Stability and Reactivity Count
Fluorine changes the rules. It pulls electron density away, making the phenol’s hydroxyl group less reactive than you’d guess by looking at phenol alone. The upshot: its stability bumps up, resisting oxidative degradation and certain chemical attacks. I’ve leaned on that kind of durability when looking for intermediates that don’t easily break down under heat or exposure to air. This chemical stubbornness means you spend less time worrying about waste or runaway decomposition, a plus for anyone managing chemical risk.
Health and Handling Considerations
Any compound bearing both phenol and fluorine needs respect in the lab. 2,4-Difluorophenol can irritate skin and mucous membranes and brings some toxicity if mishandled. Its volatility and persistent smell serve as reminders to keep the gloves on and fume hood running. In one lab I worked in, a spill turned a busy afternoon into a scramble to scrub surfaces before the odor settled into every nearby item.
Moving Forward in Industry and Research
Every time scientists or product developers turn to 2,4-Difluorophenol, they’re betting on its unique blend of physical properties—moderate volatility, selective reactivity, and robust stability under tough conditions. But with every new use, safe handling practices and careful storage sit side by side with innovation. Better ventilation systems, improved protective gear, and ongoing education about chemical hazards help protect workers while allowing this compound to play its part in progress.