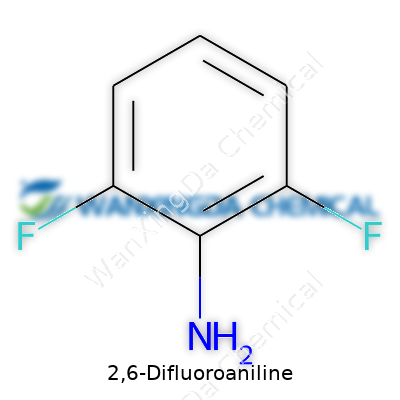
2,6-Difluoroaniline: Meeting Chemistry’s Demands Through Innovation and Responsibility
Historical Development of 2,6-Difluoroaniline
The development of difluorinated aniline derivatives has often mirrored the broader story of industrial chemistry. Researchers and manufacturers sought molecules with specific traits, looking for ways to stretch the capabilities of organic synthesis. 2,6-Difluoroaniline first caught the attention of chemists during the mid-20th century, a period marked by advancements in halogenation techniques and the growing relevance of aromatic amine derivatives in dyes, agriculture, and later pharmaceuticals. The product grew up in the age of fine chemicals, where nuanced differences in atom position made monumental shifts in reactivity and application. Early journals from this era discuss painstaking methods for fluorinating aromatic rings, often yielding low quantities with much effort. As modern catalytic and electrophilic fluorination procedures took off, yields improved, and purity no longer remained such a hurdle. It became easier for scientists and industries to work with this fluorinated aniline, opening new doors for research and technology.
Product Overview and Real-World Demand
2,6-Difluoroaniline brings something unique to the table among aniline derivatives. The molecule, shaped by two fluorine atoms ortho to the amino group, stands apart for its chemical stubbornness where needed, yet offers enough reactivity for innovative synthesis. This compound shows value wherever precise chemical behavior translates into product performance—especially in areas like pharmaceuticals, fine chemicals, polymer modification, and crop protection. It doesn’t flood the market as a bulk commodity but holds steady ground in specialty and research-focused applications. In practice, working with this chemical has highlighted not only its versatility but also the rigor required in safely handling halogenated aromatics.
Physical and Chemical Properties
Anyone who’s mixed, heated, or stored 2,6-difluoroaniline can attest that this isn’t a compound to overlook. Its pale yellow-to-colorless crystalline form hints at purity when freshly synthesized. A melting point around 25°C gives this product a peculiar liquid-solid balance in the lab, and its boiling point near 170°C allows manageable distillation under controlled conditions. Solubility trends among aromatics continue with 2,6-difluoroaniline: it prefers organic solvents—nothing surprising for those familiar with substituted anilines. Still, introducing fluorine atoms at the 2 and 6 positions dials back electron density, making the ring less eager to jump into further electrophilic substitution. Those fluorines also add some steric congestion, shaping the molecule’s profile in reactivity and separation. These nuances shape everything from process hazards to chromatographic separation.
Technical Specifications and Labeling Practice
For those buying or selling 2,6-difluoroaniline, purity targets often align with the intended application. In research settings, a minimum purity of 98% sets the baseline, with trace impurities (especially other positional isomers or unreacted aniline) scrutinized closely. Documentation usually highlights water content, residue on ignition, and spectral traces—key details for those chasing reproducibility or scaled synthesis. There’s little room for error in labeling with chemicals like this. Over the years, I’ve come to respect the discipline demanded by chemical labeling and documentation; one wrong digit or unlisted impurity can sidetrack an entire project, and with a fluorinated aromatic, even a single percent of unknown impurity means extra analytical work and repeated purification.
Preparation Method: A Challenge in Precision
Synthesizing 2,6-difluoroaniline doesn’t follow a simple pour-and-stir routine. Traditional routes often start with difluoronitrobenzenes—specifically, 2,6-difluoronitrobenzene—using nucleophilic aromatic substitution or catalytic hydrogenation to produce the target amine. At bench scale, reactions march on under an inert atmosphere, heating often needed for effective hydrogenation and careful attention paid to side products. The introduction of fluorine atoms onto an aromatic ring remains nontrivial, and historic methods required either direct fluorination under harsh conditions or indirect substitution patterns. I’ve seen experienced chemists run entire weeks’ worth of reactions only to isolate barely a gram, proof that the fluorination game hasn’t lost its challenges, even with modern reagents.
Chemical Reactions and Modifications
The chemistry toolbox for modifying 2,6-difluoroaniline revolves around its amine function and its halogen-substituted ring. The two fluorines, both electronically and sterically, slow down some classic aromatic substitution reactions, but the amino group still anchors the molecule as a launching pad for further reactions. Acylations, diazotizations, or condensed product formation work best under tightly controlled conditions. In my own lab work, I’ve noticed that failed reactions often trace back to underestimating the resilience added by those two ortho-fluorines. This resistance can be an asset—offering metabolic stability in medicinal compounds, for instance—but it also means patience and creativity from a synthetic chemist hoping to coax the ring into playing along with complex schemes.
Synonyms and Product Nomenclature
2,6-Difluoroaniline appears under a series of names, each marking a different context—the IUPAC name, 2,6-difluorobenzenamine, dominates in regulatory documents. CAS Number 5509-65-9 gathers all relevant literature and safety records for easy cross-checking. Those of us who spend years immersed in chemical catalogs, research papers, and regulatory guides know the importance of tracking synonyms to avoid confusion or even costly procurement mistakes, especially in multi-lingual or multi-disciplinary environments.
Safety and Operational Standards: Realities of Handling
Lab safety around aromatic amines goes beyond gloves and goggles, especially when working with difluorinated variants. 2,6-Difluoroaniline, like its relatives, brings the usual suspects: irritation to skin, eyes, and respiratory tract, and possible sensitization. Researchers also need to remain aware of organ-specific toxicity risks associated with aromatic amines, referencing established workplace exposure limits and local PPE guidelines. A moment’s lapse—leaky pipette, unventilated workspace—can turn routine synthesis into a trip to the doctor. Disposal requires respect, with halogenated waste streams often held for specialized treatment. In my experience, every safe experiment with difluoroaniline builds habits for broader chemical vigilance.
Application Area and Practical Utility
Certain molecules carve out a niche, and 2,6-difluoroaniline found its place in building more complex structures for drugs, agrochemicals, and specialty coatings. In the pharmaceutical sector, the increased metabolic resistance imparted by ortho-fluorines makes this compound a powerful intermediate for potential therapies. I’ve seen research teams invest a year of work using this molecule as an anchor, incorporating it into kinase inhibitors and enzyme-targeting scaffolds. Agricultural chemists tap into similar properties, designing crop protection tools that stand up to environmental stress. Coating formulators appreciate the altered solubility and bond strength that fluorine brings, translating to products resisting harsh environments in electronics and infrastructure.
Research and Development: Driving Innovation
The story isn’t just about where 2,6-difluoroaniline goes—it also represents the relentless push in R&D circles to tailor molecules for tomorrow’s problems. Chemists keep returning to this compound’s unique substitution pattern, exploring new derivatives for activity screening, coupling with polymer backbones, or building sensor arrays. It stands as a frequent subject in patents related to kinase inhibition, imaging agents, and electronic materials. Those working at the research frontier know that every substitution drives different behavior, and a nimble synthesis route developed for 2,6-difluoroaniline can spur follow-on projects in larger compound classes.
Toxicity Research: Learning From Caution
Toxicologists and environmental scientists don’t regard any aromatic amine lightly, and 2,6-difluoroaniline keeps the tradition going—demonstrating acute and chronic toxicity features, including methemoglobinemia risks through metabolic biotransformation. Animal models, in vitro screening, and regulatory monitoring all contribute to cataloging hazards. It’s easy to point to published LD50 numbers, but as someone who’s navigated risk assessment and lab ventilation upgrades, it’s clear that “below threshold limits” doesn’t always mean risk-free. Even trace contamination can complicate waste handling and downstream product safety. Regulatory guidelines suggest careful monitoring for workplace exposure and rigorous handling protocols, adapting as new toxicity data rolls in. Safer handling, improved detection methods, and clear labeling aren’t just bureaucracy—they keep accidents rare.
Future Prospects: Sustainable Chemistry and Technology’s Reach
What comes next for 2,6-difluoroaniline depends on both science and industry’s appetite for ever-refined molecules. With growing attention paid to green chemistry, fluorination strategies continue evolving—pushing for safer starting materials, lower emissions, and higher yield. Advanced catalytic techniques promise more efficient insertion of fluorine, aiming to cut the environmental burden that historically followed aromatic fluorination. Chemists remain eager to harness the stability and reactivity balance of fluorinated anilines in next-generation drugs, smart coatings, and molecular electronics. The conversation around sustainable manufacturing, improved toxicity mitigation, and expanded analytical monitoring will frame the future use of this compound. For seasoned chemists and future innovators, working with compounds like 2,6-difluoroaniline remains a test of skill, discipline, and vision.
Seeing Chemistry at Work in Everyday Products
In a world packed with specialty chemicals, certain building blocks shape whole industries. 2,6-Difluoroaniline might not sound familiar to most people, but it hides behind the scenes in the manufacture of many products that touch daily life. At its core, this chemical forms the backbone for complex molecules, especially in pharmaceuticals and crop protection.
Crafting Tomorrow’s Medicines
Pharmaceutical research often needs starting materials that can be turned into more complicated structures. 2,6-Difluoroaniline stands out thanks to the reactivity of its fluorine atoms. Medicinal chemists turn to compounds like this to develop new medicines, especially those used to tackle stubborn diseases. Fluorinated anilines often appear in drug molecules because they improve the drug's ability to last longer in the body and block harmful enzymes or signals more efficiently.
Drugs for cancer, inflammation, infections — all these areas benefit when chemists have access to new fluorinated scaffolds. The industry rarely sits still, always searching for molecules that can slip past drug resistance or avoid troublesome side effects. 2,6-Difluoroaniline gives research teams a reliable jumping-off point thanks to its unique properties.
Defending Crops, Feeding the World
Food security drives a huge chunk of chemical innovation. 2,6-Difluoroaniline finds a second life in crop protection products. Pesticides and herbicides need sturdy chemical structures to withstand sunlight, rain, and aggressive pests. The fluorine atoms in this molecule not only bring stability but also affect the way these chemicals interact with insect and weed physiology. Stronger molecules mean longer-lasting protection for crops, helping farmers keep yield losses to a minimum.
There’s a balance to strike here. Public concern keeps growing about chemical residues entering the food chain. Regulators and manufacturers face pressure to make plant protection safer, cleaner, and targeted. Chemicals like 2,6-Difluoroaniline require careful evaluation, both for their benefits and their risks. My own experience with community gardening has shown how even backyard growers pay close attention to trace chemicals in soil or water, bringing a genuine demand for innovation with more sustainable footprints.
Other Specialized Applications
Material science and electronics also tap into the unique features of 2,6-Difluoroaniline. New dyes and pigments use substituted anilines for increased brightness and stability, meaning longer-lasting colors in everyday objects. Research into high-performance polymers and specialty resins leans on molecules like this for added chemical resistance or thermal strength.
Seeking Safer and Smarter Chemistry
The story of 2,6-Difluoroaniline shows both innovation and responsibility. Chemists must keep pushing for safer manufacturing steps and better waste handling. In many labs, green chemistry is no longer optional. Solvent recycling, cleaner production lines, and tighter oversight all need to stand alongside innovation. That pressure isn’t just academic; worries over chemical spills or pollution enter every conversation about this and similar chemicals. The best future for any specialty chemical means maximizing its benefits while trimming as much risk and waste as possible.
Everyone in this space has a stake in making sure that the chemicals fueling progress do not leave a lasting problem. Whether it’s in medication, agriculture, or high-performance materials, 2,6-Difluoroaniline’s story underlines how important it is to mix creative science with common-sense stewardship.
Why Chemistry Details Like This Matter
You don’t need to work in a lab to feel the impact of little changes at the molecular level. Take 2,6-difluoroaniline as an example. It shows how a couple of tweaks—just two fluorine atoms—can make a world of difference in the way a molecule interacts with the world. The molecular formula for 2,6-difluoroaniline is C6H5F2N. That means six carbon atoms, five hydrogens, two fluorines, and one nitrogen all linked up in a chemical arrangement that’s more than the sum of its parts.
Bringing Structure and Function Together
I remember my first organic chemistry class feeling intimidating. The mess of hexagons, lines, and dots scattered across the board felt remote from everyday life. Over time, I learned to appreciate these details. For example, in 2,6-difluoroaniline, the placement of the fluorine atoms on the benzene ring—both sitting next to the amine group—shifts everything from electronic distribution to reactivity. This isn't just a trivial point. It explains why fluorine substitution can make molecules better at resisting breakdown in drugs or more selective in industrial reactions. The basic structure—a benzene ring with an amino group and two fluorines attached—becomes a blueprint that shapes how the molecule behaves.
Impacts on Research and Industry
There’s a specific reason chemists seek out variations like this one. Fluorinated aromatics play a big part in pharmaceuticals and agriculture. A small formula like C6H5F2N opens up possibilities for developing new treatments or building blocks. The presence of the –NH2 group, paired with those fluorines, allows for different reactions than you’d get from plain aniline or non-fluorinated versions. These details matter in synthetic pathways. You see it in research papers where swapping a single atom spells the difference between an effective medicine and an ineffective one.
Health, Environment, and Responsibility
Responsible handling becomes everything when dealing with fluorinated compounds. Fluorine atoms make organic chemicals more persistent in the environment. Companies and labs have to pay attention to waste streams and safety measures because molecules like 2,6-difluoroaniline don’t break down easily. Regulators and health authorities keep a close eye on potential toxicity. While this specific compound might not feature in headlines, trends in environmental chemistry stress the need for careful management of fluorinated organics. It’s not just about theoretical risk—fluorinated residues can build up in water and soil if folks ignore proper disposal.
Solutions and Forward Steps
Low impact chemistry deserves more backing. Scientists have started leaning into green chemistry principles—reducing hazardous waste, using milder conditions, and designing safer molecules. Open communication between chemists, manufacturers, and regulators helps shape a world where beneficial compounds don’t unintentionally harm health or ecosystems. For anyone working with or developing molecules like 2,6-difluoroaniline, these principles aren’t just checkboxes—they’re habits that ripple outward.
Real-World Relevance
It helps to remember: behind every chemical formula sits a network of choices and consequences. A single molecule like C6H5F2N stands at a crossroads. On one side, it’s a foundation for medical or material advances. On the other, it pushes us to respect the details and consider what comes next, inside the lab and in the world outside.
Getting Real About Chemical Hazards
The first time I handled 2,6-Difluoroaniline, its sharp, chemical odor hit me. That smell was a reminder: some chemicals mean business. Not every bottle with a long name on the shelf draws attention, but this one deserves respect. Handling it without proper precautions can bring its own set of health risks, including skin burns, eye injuries, and breathing problems. One can never take shortcuts with safety in a chemistry lab.
Simple Steps: Gloves, Goggles, and Good Sense
You slip into a pair of thick nitrile gloves, you check that your goggles fit, and you make sure your coat sleeves reach your wrists. Lab coats sometimes feel like a nuisance until chemicals splash—then you’re grateful. Safety gear comes before curiosity or convenience.
It only takes a tiny spill for 2,6-Difluoroaniline to burn your skin or eyes. Its vapors may not be as notorious as some other anilines, but any inhalation can leave you with a sore throat, nausea, or worse. Splash-proof goggles feel bulky until you picture a hospital visit. I learned early on to never skip that step.
Let The Fume Hood Do Its Job
Good ventilation is more than a suggestion. Fume hoods exist for a reason. Their fans draw away invisible vapors, keeping the air you breathe cleaner. You slide the sash down so only your hands enter. It’s not paranoia; it’s protection. I once watched a classmate casually open a cap outside the hood and cough instantly—lesson learned by all of us watching.
Waste Goes Where It Belongs
Proper disposal can seem tedious, but dumping 2,6-Difluoroaniline down the sink brings risks to both people and the environment. It can harm waterways and animals if ignored. Specialized waste containers safely lock away leftovers and contaminated gloves. Putting in the extra effort helps keep you and your colleagues safe, while also reducing chemical footprints outside the lab gates.
Why Labeling Matters
Clear labeling avoids a world of confusion. Unmarked bottles, mystery residues—these accidents are waiting to happen. Bold, waterproof labels with dates and hazards keep everyone honest and safe. Misplaced trust in memory has caused enough scares for me and teammates to always double-check.
Training Makes All The Difference
No one wakes up ready to handle toxic chemicals. Good training sticks with you: learn the symptoms of exposure, understand the layout of your emergency showers, and always know where the eyewash stations sit. Inexperienced workers often fumble under pressure. A few minutes spent drilling emergency responses make sure you act fast without thinking twice.
The Culture of Caution
Labs thrive only as long as everyone respects the rules. I’ve seen seasoned chemists and young students alike reminded by near-misses that chemical safety never gets old. Real safety comes from habits—checking bottles, double-gloving, covering eyes, breathing clean air. A few extra minutes with each step prevent days lost to injury or illness.
2,6-Difluoroaniline isn’t unique in demanding this attention, but its risks paint a clear picture. Chemical safety, built from practice and vigilance, lets the science move forward without personal cost. That matters more than shaving off time or cutting corners. Always suit up, work aware, and leave nothing to chance.
Chemistry in the Everyday World
Walking through high school chemistry feels far away, but lessons from those days often echo in surprising places. The common question “What’s the boiling point?” comes up for more than just kitchen water. It decides how we handle substances, design experiments, and manufacture everything from pharmaceuticals to plastics. Take 2,6-Difluoroaniline for example—a tongue-twister of a name, but behind it sits a molecule vital in research circles.
Why Boiling Points Matter for Chemicals Like This
Ask anyone who’s spent time over a lab bench: the boiling point often rules the choices we make. With 2,6-Difluoroaniline, the figure lands around 183°C according to several chemical handbooks and peer-reviewed sources. Knowing this isn’t a throwaway detail. Set the temperature too low and nothing happens, set it too high and you’re reaching for the fire extinguisher—or worse, losing your sample and patience. For folks involved in organic synthesis, separation, or purification, this single number draws the line between clean results and a messy bench.
Boiling Points as a Marker of Structure
Boiling points tell us a lot about the structure of a compound. I remember in university, we’d compare similar chemicals, swap a few atoms, and watch the boiling point change. Swap just one hydrogen for fluorine, the boiling point sometimes jumps; nature plays with us like that. Here, the difluoro substitution on the aniline ring forces a change in how molecules pile up in the liquid. Fluorine, being a bit antisocial, keeps molecules apart, but the pattern of substitution—here, the 2 and 6 positions—adds just enough complexity that the boiling point holds higher than unsubstituted aniline.
Industry Depends on These Details
This is not some dusty academic exercise, either. Chemical plants need exact temperatures for safe, efficient reactions. Miss by even a few degrees, and costly mistakes happen. I’ve seen stories of industrial leaks or failed runs because someone relied on a generic database instead of checking a trusted source. Precise physical data, like for 2,6-Difluoroaniline, drives decisions on distillation, solvent choice, and even how companies dispose of waste. Safety lives or dies by accurate boiling points, not rough guesses.
Improving Access to Data
It’s easy to overlook how tough it can be finding physical constants for obscure molecules. Many chemists still find themselves flipping through old handbooks or paying for access to digital databases. The solution sounds simple: push for open-access chemical databases maintained collaboratively by universities, government, and industry. Peer review and transparency can catch mistakes, update figures, and help everyone work from the same trustworthy sheet. Accurate, verified boiling points trickling down from publications into free resources would save time, reduce accidents, and level the playing field between large firms and small labs or classrooms.
Fact-Driven Science Serves Us All
I’ve found that respect for accurate data—like boiling points—connects directly to scientific trust. Skipping, guessing, or rounding off can sink good ideas and put people at risk. So whether you’re running experiments or drafting safety guidelines, remembering that 2,6-Difluoroaniline boils near 183°C isn’t just trivia. Genuine attention to fact lifts up the whole field, makes science real to those outside of it, and keeps both researchers and the public safer at the end of the day.
A Chemical Many Don’t Talk About
Most folks have never heard of 2,6-difluoroaniline unless they’ve spent time in a chemistry lab or the chemical industry. The name itself draws out visions of complex molecular diagrams and a lingering sense of “should I be worried?” As someone who’s handled aromatic amines and seen firsthand what makes a chemical “water soluble,” I think it’s worth breaking down how this particular compound behaves once someone tries to mix it with plain old H2O.
The Reality of Its Chemistry
Solubility comes down to what the molecules want. Water likes to bond with other molecules that can form hydrogen bonds or at least cling on through some kind of polarity. 2,6-difluoroaniline, with its aromatic ring and two fluorine atoms sitting on those corners, doesn’t quite fit the bill. Fluorine atoms pull electron density away, making the molecule a little less friendly toward water. Aniline itself—the building block here—already resists dissolving in water beyond a certain point, and fluorine substitution only makes matters trickier.
Why Water Solubility Matters Beyond the Lab
Not every chemical should go down the drain. Low water solubility means any accidental spill, leak, or improper waste handling keeps the chemical hanging around in the environment a lot longer. It doesn’t just “wash away.” Living near a facility that handles these substances, I've seen conversations shift quickly from curiosity to concern when the topic of groundwater contamination pops up. Regulators often have to dig deep—literally and figuratively—into how these compounds can affect local water tables. Scientists and environmental groups keep asking tough questions about persistence and how low-soluble chemicals interact with ecosystems.
Putting Numbers to Words
Technically, 2,6-difluoroaniline offers very limited water solubility, usually reported at just a few grams per liter or less under standard conditions. To put it another way, dropping a spoonful into a small glass of water won’t leave it looking like sweet iced tea. Most of it floats, sinks, or clumps at the bottom. You need organic solvents if you really want to break it apart at any significant concentration. This behavior isn’t rare among halogenated aromatic amines, and the fluorine in this case keeps things even more stubborn.
Handling Real-World Situations
My own experience says care is always the better policy. If you work where these chemicals appear, or even live nearby, knowing about their solubility—or lack of it—should guide your decision-making. Facilities should double-check spill protocols and waste treatment processes, making sure chemicals don’t reach the wrong places. Water treatment plants face challenges here because low solubility often means regular filtering won’t pull the compound out. Instead, activated carbon filters or advanced oxidative processes do a better job removing traces for good.
Looking for Smarter Solutions
Organizations can lower the risk by minimizing open transfers and keeping tight control on waste. Research into more biodegradable or less persistent aromatic amine alternatives grows every year. On the regulatory front, staying transparent with local communities builds trust when sites handle low-solubility chemicals.
Why We Should Keep Talking
Understanding chemicals like 2,6-difluoroaniline’s reluctance to dissolve in water shifts the conversation around chemical safety. It’s less about scare tactics and more about staying informed, preparing for accidents, and protecting shared resources. That’s something everyone, whether in a lab coat or just living nearby, should think about.