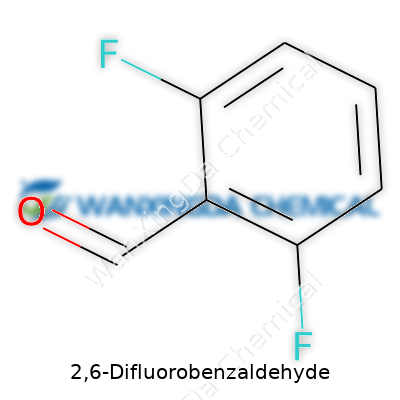
2,6-Difluorobenzaldehyde: Unpacking Its Role in Modern Chemistry
Historical Development
2,6-Difluorobenzaldehyde entered the spotlight as the toolbox of organic chemists expanded through the twentieth century. The drive to create more selective, efficient building blocks opened up fresh routes for chemical transformation. Fluorinated aromatics, often overlooked for decades, became increasingly valuable as researchers learned how a single atom swap changes an entire molecule’s behavior. The addition of two fluorines on a benzaldehyde ring, nestled in the 2 and 6 positions, steered this compound into labs working on both academic and industrial synthesis. Some of this history comes from the wider recognition that tweaking aromatic rings with electronegative atoms brings new biological and material properties within reach. As the pharmaceutical and materials scenes demanded more precise molecular tailoring, compounds like 2,6-difluorobenzaldehyde quietly earned a permanent spot on the shelf.
Product Overview
2,6-Difluorobenzaldehyde isn’t the kind of compound you’ll see on store shelves, but it’s a building block behind the scenes for countless industries. Chemists reach for this molecule when a specific, directional reactivity is needed—often to introduce a handle on a benzene ring that can steer future reactions. Its aldehyde group creates opportunities for further transformations, especially when other parts of a target molecule aren’t easy to modify. The two fluorines set the stage for properties like increased metabolic stability in pharmaceuticals and altered polarity in liquid crystals, giving those who understand how to use them a strategic leg up.
Physical & Chemical Properties
The impact of fluorine atoms on a benzaldehyde ring cannot be overstated. 2,6-Difluorobenzaldehyde appears as a colorless crystalline solid or pale liquid at room temperature, carrying a sharp aromatic scent. The presence of two fluorine atoms, both strongly electron-withdrawing, drops both melting and boiling points compared to its non-fluorinated cousin. These same atoms create resistance to certain oxidative or nucleophilic attacks, making the aldehyde less reactive in ways that a chemist might not expect from non-fluorinated aromatics. Its moderate volatility and solubility profile in common organic solvents help during extractions and purifications, letting practitioners incorporate it into a wide span of synthetic routes.
Technical Specifications & Labeling
Purity takes on real significance here, especially for those working in electronics or pharmaceutical research. With applications demanding low residuals of related compounds, buyers look for products above 98 percent purity, often confirmed by gas chromatography. Labeling provides clear articulation of hazards—corrosive properties to skin and eyes, the need for good ventilation, and warnings about storage away from sunlight or strong bases. Proper batch tracking and allergy statements remain standard practice for labs that keep up with best practices in documentation and traceability.
Preparation Method
Whether in large-scale plants or smaller synthetic labs, producing 2,6-difluorobenzaldehyde is not a rookie task. Classic methods draw on the selective oxidation of the corresponding difluorotoluene, often with chromate or permanganate salts under controlled conditions. Others look to halogen exchange or even directed ortho-metalation strategies, making the process a matter of chemoselectivity and yield. Each approach carries pros and cons: some emphasize straightforward workup, while others lean on clever protecting groups or low-temperature controls. Waste management and reagent recovery factor into setup decisions, especially under ever-tightening environmental regulations.
Chemical Reactions & Modifications
Those who’ve spent time in the lab know just how potent a single substituent can be in shifting the reactivity of a molecule. 2,6-Difluorobenzaldehyde’s unique structure allows it to act both as a substrate for condensation reactions and as a stepping stone toward more elaborate fluorinated aromatics. I’ve seen researchers use it in the synthesis of Schiff bases, oximes, and hydrazones—reactions that hinge on the aldehyde’s reactivity. Nucleophilic aromatic substitutions become possible thanks to the electron-poor ring activated by the two fluorines. Chemists looking to string together larger architectures sometimes exploit these positions to stitch on new side chains or expand on heterocyclic frameworks, especially for drug candidates or polymer precursors.
Synonyms & Product Names
Different labels can be confusing. The full IUPAC name is 2,6-difluorobenzaldehyde, but catalogs often list it alongside identifiers like 2,6-DFBA, alpha,alpha-difluoro-o-tolualdehyde, or CAS registry numbers to cut down confusion with similar-sounding chemicals. Some vendors might highlight it under “fluorinated benzaldehydes” or “aromatic aldehydes”—being able to navigate these synonyms helps ensure the right material ends up on the bench.
Safety & Operational Standards
Nothing derails a research project faster than a safety mishap. Handling 2,6-difluorobenzaldehyde requires respect for both its chemical irritancy and the potential toxicity of fluorinated aromatics. Proper personal protective equipment—gloves, goggles, fume hoods—remains non-negotiable. Storage guidelines emphasize segregating it from bases, oxidizers, or acids. Air monitoring and proper waste stream management aren’t just legal requirements—they’re best practices that keep people healthy and labs working smoothly. Regular safety training keeps new hazards from slipping through the cracks, even for staff familiar with less reactive aldehydes.
Application Area
Aromatic aldehydes serve as molecular keystones in pharmaceutical, agrochemical, and materials science domains. 2,6-Difluorobenzaldehyde plays a subtle but vital role in synthesizing specific bioactive molecules, where controlling metabolic breakdown with fluorines can keep drugs active in the body longer. In display technologies, the electronic tweaks from fluorine atoms shape the performance of liquid crystal materials in touch screens and monitors. The molecule also pops up in diagnostic chemistry, where its distinct reactivity creates new pathways for covalent modification. Real power comes not from standing alone but from how it opens doors to more complex, functionalized molecules.
Research & Development
Academic and industrial teams continue to probe how small tweaks on the benzene ring can ripple out to influence everything from electrical conductivity to enzyme inhibition. For those tracking the next generations of targeted drugs or specialized polymers, 2,6-difluorobenzaldehyde often acts as an entry point for further functionalization. Publications describe emerging catalytic methods that lower reaction temperatures, cut waste, and give access to isomeric products not feasible with bulkier fluorinated rings. Collaboration between synthetic chemists and computational modelers adds another layer, pushing forward the understanding of substituent effects at the quantum level. Adapting established reactions to green chemistry principles shapes the future of how this compound is made and used.
Toxicity Research
Fluorinated aromatics raise important questions about environmental persistence and chronic toxicity. While 2,6-difluorobenzaldehyde hasn’t sparked the same level of controversy as perfluorinated alkyl substances, ongoing studies assess its behavior in soil, water, and biological tissues. Researchers track breakdown pathways and look for metabolites that may linger or accumulate. Data so far points to low acute toxicity, but concerns about bioaccumulation keep this molecule under a microscope, especially for those developing products intended for large-scale or long-term use. Risk assessment frameworks continue to evolve, demanding robust data about both short-term and chronic exposures.
Future Prospects
Looking ahead, 2,6-difluorobenzaldehyde seems poised to remain a workhorse for cutting-edge molecule building. The thirst for new pharmaceuticals, green pesticides, and advanced materials only grows, and every one of these fields benefits from the reliability and tunability that this compound offers. Technical advances in greener synthetic routes, biocatalysis, and digital synthesis planning may soon make its production cleaner and more efficient. Meanwhile, tighter scrutiny of environmental impact pushes researchers to chart safer, more responsible uses. In my experience, chemists thrive on small changes that unlock big advantages—2,6-difluorobenzaldehyde is a prime example of how a smartly positioned molecule can bridge current needs and future ambitions in modern chemistry.
I first came across 2,6-Difluorobenzaldehyde during my university chemistry days. It’s a name that will trip you up if you’re not used to Chemical Abstracts, but in simple terms, it’s a derivative of benzaldehyde with two fluorine atoms attached. Even though it looks like a tongue-twister, this compound plays a real part in the world of synthetic chemistry, pharmaceuticals, and even a few household products. It rarely gets the headlines, but it’s quietly doing some of the behind-the-scenes work to help bring new medicines and materials to life.
What Makes 2,6-Difluorobenzaldehyde Special?
The punchline is in its structure. Substituting benzaldehyde with fluorine atoms at specific points on the ring changes the way the molecule behaves. Fluorine has a reputation for tweaking the properties of organic compounds—such as making them more resistant to breakdown by the body or more efficient during chemical reactions. Because of this, researchers have found that adding fluorine to benzaldehyde opens doors for all kinds of new applications.
In laboratories and factories, chemists use 2,6-Difluorobenzaldehyde as a building block. Say you’re working on a new drug or agrochemical—you need starting materials that will survive a tough synthesis and still give you exactly the structure you want. That’s where this compound comes in. People often use it to make pharmaceuticals that handle inflammation, allergies, or neurological problems because the fluorine atoms help the final molecules last longer in the body and bind better to their targets.
Real-World Uses and Impact
Walk down the aisles of any pharmacy, and some of the drugs on the shelf trace their origins to building blocks like this one. Industry uses 2,6-Difluorobenzaldehyde for the production of intermediates—steps along the way to turning simpler molecules into life-saving drugs or high-value pesticides. These building blocks help give drugs their strength and reliability.
Researchers also turn to this compound for developing new diagnostics and imaging agents. The added stability and unique chemical behavior from fluorine atoms lets scientists fine-tune molecules so that they stay visible longer in the body during scans. As a bonus, these modifications can reduce side effects or the risk of unwanted reactions—something both doctors and patients care deeply about.
Concerns and Possible Solutions
One issue with specialty chemicals like this involves the environmental footprint. Fluorinated compounds sometimes linger in the environment, and not every manufacturer handles production responsibly. Evidence has shown that some perfluorinated and polyfluorinated substances persist in water and soil, causing headaches for regulators and communities. While 2,6-Difluorobenzaldehyde doesn’t belong to the most notorious group, it’s smart to keep a close watch on how waste streams get managed.
Making improvements depends on strong oversight and cleaner chemistry. Many companies have started investing in new catalytic systems and better recycling methods to capture waste and reduce emissions. I’ve visited plants where processes recover nearly all the fluorine, allowing for re-use instead of discharge. It takes investment and willpower, but progress is happening.
Why This Matters Now
As drug discovery keeps advancing and as farmers and doctors rely more on specific and efficient molecules, chemical building blocks like 2,6-Difluorobenzaldehyde help keep that engine running. Whether it’s the medicine for a chronic condition or the imaging agents during a diagnostic scan, behind the scenes, small details in chemistry put the right tools in the right hands. By keeping an eye on production practices and encouraging responsible innovation, the benefits can keep flowing without leaving a mess behind.
Breaking Down the Name
Chemistry never hesitates to pack a story into a name. 2,6-Difluorobenzaldehyde sounds like a mouthful, but its formula, C7H4F2O, actually reflects the logic of its structure. Each part of the name reveals clues: “di-” for two, “fluoro” for fluoro substituents, and “benzaldehyde” signals the core benzene ring attached to an aldehyde group. I remember sitting in organic chemistry classes, learning how one tweak to a molecule’s framework can create a completely different substance. It illustrates how even slight shifts at the atomic level can steer chemical behavior in a new direction.
Why the Formula Matters
Having the correct formula for 2,6-Difluorobenzaldehyde, C7H4F2O, isn’t just academic nitpicking. In research, pharmaceutical synthesis, and industrial chemistry, accuracy means safety, efficacy, and compliance. Misidentifying a compound’s makeup can lead to wasted time, unintended reactions, or even dangerous situations.
To build C7H4F2O, chemists start with a benzene ring, swap two hydrogens for fluorines at the 2 and 6 positions, and tag on an aldehyde group at the first position. This seemingly simple adjustment changes how the molecule interacts with enzymes, solvents, and other molecules. In labs, even minor errors in chemical structure can torpedo a whole set of experiments or throw safety protocols off track.
From Lab Bench to Real World Use
2,6-Difluorobenzaldehyde doesn’t just stay in textbooks. It plays a role in pharmaceutical development and materials science. These worlds demand precision and consistency. A correct formula ensures regulatory compliance, fair pricing, and honest reporting. Getting it wrong could spell setbacks—not just in development, but in the trust that underpins modern science.
I have seen how a misread formula can set off a cascade of problems in both academic and industrial labs. Bottles mislabeled or paperwork that goes unchecked have ended with halted research or even hazardous waste mishandling. Know the exact chemical formula, and a lot of hassle disappears. It becomes easier to track environmental impact, manage waste, and design safe handling protocols. With substances like this, knowledge becomes a tool for protection as much as for innovation.
Addressing Challenges
Chemistry can open doors to valuable medicines and new technologies, but the foundation has to be solid. Keeping formulas front and center—something as straightforward as C7H4F2O—helps everyone from researchers to regulators. Scientific journals and industry databases carry a responsibility to double check and clearly present these details, closing the door to confusion or mistakes.
Education also plays a big part. Encouraging students and early-career scientists to respect notation and review their references can cut down on simple but costly errors. Even in my own work, a habit of double-checking formulas has saved projects and protected my colleagues more than once. Responsible chemistry goes well beyond the lab—it anchors safety, innovation, and trust.
Respect in the Lab Goes Beyond Clean Coats
Some chemicals remind you they’re nothing to mess around with after a single whiff or splash. 2,6-Difluorobenzaldehyde belongs on that list. The name feels complicated, but the real trouble comes from what it can do if someone skips basic safety. It’s funny how chemical stories end up just like stories about sharp tools: carelessness never works out well. That’s why respect for every step matters, even on familiar ground.
More Concrete Risk Than the Usual Lab Hype
This compound doesn’t explode or put on a show shouting danger. It sneaks up quietly, biting through skin or lungs before anyone realizes. You touch it with bare fingers, you might get burned. A little on the bench turns into fumes, and your throat knows something’s off right away. To some people, this sounds like typical over-caution—just like students rolling their eyes at goggles and gloves. But the facts line up: the material irritates eyes, skin, and respiratory systems. Some literature mentions lasting damage, and no chemical worth respect ‘might’ cause harm; it does harm when ignored.
Small Steps That Save a Lot of Trouble
My early lab days saw folks rushing to finish late experiments. Some worked bare-handed or grabbed a bottle without checking the label. The biggest lesson: real safety relies on habits, not just emergency gear. I keep a habit of gloves before everything, lab coat always buttoned, goggles at the ready, and a proper mask when dealing with volatile solvents or solids. There’s no shame in double-checking everything before opening a bottle labeled like 2,6-Difluorobenzaldehyde.
Well-ventilated workspaces matter more than folks think. Fume hoods aren’t decorations—they’re the best line of defense short of avoidance. Sinks or eye-wash stations shouldn’t become the only backup plan. This blends knowledge and experience—if you respect chemical volatility, you prevent most accidents.
Truth in Preparedness
The chemical industry reports plenty of minor injuries, and most trace back to skipped steps. If you ask me, chemical burns and inhalation incidents don’t start with bad luck—they start when people try shortcuts. Double-seal containers after use, and never trust a mystery residue. If a spill happens, folks panic less with a solid cleanup plan and spill kit nearby. Clear pathways and uncluttered surfaces save lives when something goes sideways. These aren’t just teacher’s pet routines; these are hard-earned lessons.
Better Habits Start Early
Some people get their training in an undergraduate lab and think they know enough. No manual covers every scenario, but firsthand mistakes teach fast. The chemical safety culture needs practicality. Nobody enjoys paperwork or lectures, but everyone likes going home with their health. Peer checks, honest reminders, and accountability culture help turn rules into reflexes. I tell newcomers: take every small danger seriously, or one day you’ll regret not doing it.
So next time someone holds a bottle of 2,6-Difluorobenzaldehyde, I hope they remember it’s not just another chemical name. There’s real risk in that vial, but respect for safety habits keeps everyone out of the kind of trouble nobody ever wants to see.
Why Chemists Sweat Storage Details
Few topics spark more hasty double-checks in the lab than handling aromatic aldehydes like 2,6-Difluorobenzaldehyde. Working as a lab assistant early on, I watched an experienced chemist pause mid-procedure to eyeball our bottle. He cared less about the fancy label and more about a simple fact: even a slight slip in storing this stuff brings big headaches later.
The Risks Hiding on the Shelf
A lot of folks encounter 2,6-Difluorobenzaldehyde in the hunt for new pharmaceutical ingredients. Its double fluorine punch adds useful reactivity, but those same atoms push up its volatility. Open the bottle too often and the room fills with a sharp, choking smell nobody quickly forgets. Even at small scales, improper storage ramps up health dangers and spoils carefully measured reactions. Stories float around college research groups—open air contact leading straight to yellow crud in the container or worse, resin gunk coating glassware.
Simple Storage, Fewer Worries
A dry, cool spot is the secret. That sounds almost obvious until you remember how often glassware sits rinsing, drawers stay damp, or sunlight hits a forgotten bench. Ultraviolet light speeds up breakdown in aromatic aldehydes. Room temperature storage works, but heat starts trouble—light, air, and warmth team up to degrade the compound faster than many realize.
The best spot sits away from sunlight, in a sealed glass bottle. Not plastic. Even a tiny bit of moisture clouds the liquid and triggers slow polymerization. Silica gel packets or tight-sealing desiccators help block out humidity. These steps keep the benzaldehyde clear, colorless, and ready for actual chemical work.
Health Concerns That Don’t Get Enough Attention
No one likes surprise headaches or irritated throat and skin. 2,6-Difluorobenzaldehyde vapors mean business. On a busy day, a cracked lid or loose cap turns a safe working space into a coughing zone. Glove use seems obvious but enforcement falters in fast-paced setups. Strict storage habits reduce these risks before they start.
I once saw a colleague brush off the bottle, thinking 'just another organic.' She couldn’t work near it a week later, symptoms too nasty to ignore. Industry reports back this up: a single incident can spiral into workplace reviews or medical checkups.
Practical Steps for Every Lab
People trust written protocols but real safety comes from habit. Label containers clearly with both name and date received. Keep away from acids, bases or oxidizers—these common reagents don’t play nice with aromatic aldehydes. Regular inventory checks flag old or decomposed material before it causes harm.
Large facilities try automation or chemical management systems, yet small teams benefit most from a shared sense of responsibility. Teaching people what goes wrong, not just what works, matters just as much as the right shelf or the right bottle.
Better Storage, Better Outcomes
Careful storage earns its keep by cutting accidents and keeping research on track. If more teams focused on airtight bottles, low humidity, and away-from-sunlight shelves, safety talks could shrink without loss of vigilance. Small daily decisions pack long-term punch—especially in labs chasing the next discovery.
Why Molecular Weight Matters in Everyday Chemistry
Most chemistry students encounter a moment of truth in the lab, staring down a substance’s label, desperately hoping they’ve remembered the right way to figure out molecular weight. Ask about 2,6-Difluorobenzaldehyde, and some people might draw a blank. Put simply, this compound has the formula C7H4F2O, and its molecular weight clocks in around 142.10 grams per mole. Far from a trivial number, this figure unlocks a lot for anyone working in chemistry, pharma, or even just tinkering at a home bench.
Small Compounds, Big Impact
Back in my university days, a teaching assistant asked us to calculate the amount of a certain chemical needed for a reaction. A few of us flubbed the calculation because we glossed over the molecular weight. Precision matters. In drug discovery or environmental monitoring, even a tiny miscalculation can throw off results, waste valuable resources, or skew data that guides big decisions. For 2,6-Difluorobenzaldehyde, this matters just as much. Its mixture of fluorine atoms packaged onto a benzaldehyde frame changes not just its reactivity but also its metabolic fate in biological systems.
Facts Around Fluorination
Fluorinated organic chemicals like this one turn up all over, from crop protection agents to advanced pharmaceuticals. Those two fluorine atoms aren't just window dressing. They raise the molecular weight, shift the electron density, delay metabolic breakdown, and resist microbial attack. This property is why so many researchers lean toward these molecules in the search for more stable or potent drugs. Reliable knowledge of the exact mass informs not only dosage calculations but also environmental studies—tracking how much ends up where, and how long it might stick around.
Stumbling Blocks and Solutions in Lab Life
We’ve all watched the digital scale flicker, hoping the tiny crystals are the right amount for a reaction. The real world doesn’t hand out do-overs when molecular weight is miscalculated. For 2,6-Difluorobenzaldehyde, inaccurate numbers mess up yields, product purity, and waste money. Using verified chemical references solves a slice of that challenge, but new chemists can get tangled up between molar mass versus molecular mass, or between theoretical and real sample purity.
This highlights a need for better access to digital tools and reliable data sources in teaching labs—not just old textbooks. Modern software and chemical databases offer a way out, guiding early-career scientists to avoid common pitfalls.
Working Toward More Transparency
Many labs still run with hand-me-down procedures and illegible notes. It pays off to share accurate data, precise calculations, and sources among teams. Greater openness ensures fewer mistakes—even for well understood compounds like 2,6-Difluorobenzaldehyde. Whether it’s a pharma start-up or a local university class, building solid habits around chemical data cuts errors and deepens know-how.
Understanding molecular weight almost feels humble, but it’s the bedrock for safe experiments, cost-effective processes, and credible research. So next time a new student asks, “How much do I need?”—it’s worth remembering these numbers back up every step, no matter how small the vial.