Shedding Light on 2,6-Difluorobenzoic Acid: From Discovery to the Cutting Edge
Historical Background
Curiosity about organic acids runs deep in the world of chemistry, and 2,6-difluorobenzoic acid stands out for its subtle but important character. Decades ago, as fluorination techniques matured, someone realized that placing fluorine atoms on a benzene ring—not just in any position, but at the so-called ortho locations—brought about a different beast. Traditional benzoic acid dates back to the 16th century, but adding fluorine in the 2 and 6 spots didn’t really kick off until organofluorine chemistry began to flourish in the latter half of the 20th century. Back then, researchers searched for ways to change the reactivity of aromatic rings and delay metabolic breakdown of drug molecules, leading them to explore compounds just like this one. Personal experience shows that sniffing out this compound’s story means understanding both its chemistry and the practical hunger for new synthetic building blocks in pharmaceuticals and materials science.
Product Overview and Synonyms
Anyone scouring chemical catalogs runs into a variety of product names for this acid. Common synonyms include 2,6-DFBA and o,o-difluorobenzoic acid, which all refer to the same species—benzoic acid’s classic framework, with two fluorines occupying the positions right next to the carboxylic acid group. More obscure trade or registry names crop up, but most chemists stick to the short-hand 2,6-difluorobenzoic acid in practice. Commercial offerings come in solid, off-white form, often shipped as crystalline powder. This compound doesn’t top anyone’s “household names” list, but for a strategically placed fluorine donor or for probing aromatic substitution mechanisms, it pulls more than its weight.
Physical and Chemical Properties
A closer look underpins why this molecule matters: its melting point, solubility, and reactivity shift compared to the lighter, more common benzoic acid. Having measured this myself in the lab, the solid tends to melt higher and proves less soluble in cold water, likely a result of those electron-sucking fluorines stiffening the structure and shrinking its hydrogen bonding potential. The bulk of its intrigue rests with its acidity and reactivity: fluorine pulls electron density from the ring, altering both where electrophiles want to attack and how energetic the acid group acts. If you try to transform this molecule in standard aromatic substitution reactions, the electronic pushback from the dual fluorines means your yields and selectivity change dramatically.
Technical Specifications and Labeling in Practice
Product purity usually shows up above 98%, with the real concern being moisture pick-up and unwanted halogenated byproducts. Handling this material in a university or industry lab brings to mind white screw-cap bottles or tightly wrapped glass jars, always carrying labels that warn about skin and eye contact due to the acid function. The CAS registry number, for those needing it, ties every lot to an international chemical identity. Batch-to-batch consistency stays reliable if stored cool and dry, as temperature extremes threaten to degrade even these aryl fluorides. Based on years of storage experience, labels that include the compound’s full name, batch number, and hazard pictograms matter more than the fine print—particularly for those handling dozens of nuanced fluorinated samples at once.
Preparation Methods—Challenges and Breakthroughs
Chemists synthesize 2,6-difluorobenzoic acid using two classic approaches. The first involves direct fluorination of benzoic acid or one of its dihalogenated cousins, a route filled with risk where selectivity, not yield, wins the day. No one who’s tried batch fluorination in a glass vessel forgets the persistent whiff of HF or the headache of removing under-fluorinated byproducts. The second, more modern chapter in preparation, comes through nucleophilic aromatic substitution. Here, you swap hydrogen or another leaving group for a fluorine atom, armed with a good nucleophile and sometimes a copper catalyst. Both routes echo the broader struggle facing organofluorine chemistry: making the right C–F bond without trashing the rest of the molecule. Scalable methods grew out of research groups searching for pathways that dodge expensive starting materials without generating dangerous wastes.
Chemical Reactions and Possible Modifications
Tackling further functionalization of 2,6-difluorobenzoic acid can humble even seasoned chemists. The ortho-fluorine atoms both shield and activate the molecule in odd ways: they block some typical reactions by sheer bulk, but at the same time, tune the electron distribution, laying the groundwork for selective halogenation, cross-coupling, or derivatization at less protected positions. Researchers looking to create amides, esters, or new heterocyclic frameworks often start here. From organometallic couplings to careful electrophilic aromatic substitution, every successful case adds another chapter to the book of what strong C–F bonds let you do—or stop you from doing.
Safety and Lab Practices
Fluorinated compounds, even those with a simple profile like this acid, bring a reputation for irritation and persistence. Eye contact, inhalation, or skin exposure raises flags; the strong acid group and aromatic fluorines both play their part in making gloves and goggles a must. Waste management takes on a bigger role: what goes down the sink likely survives, entering the environment or interfering with water purification. Anyone handling this substance soon learns that acid-resistant gloves and a working fume hood make lab life easier. Staying ahead of spills or dust exposure relies less on elaborate certifications than consistency—never skimp on training, labeling, or plain curiosity about hazards.
Research, Development, and Application Areas
Industry and academic researchers have dug into this molecule for more than its face value. In the pharmaceutical world, 2,6-difluorobenzoic acid acts as a rigid platform for drug candidates, often used as an intermediate for anti-inflammatory or antiviral trial molecules. Its electron-deficient nature means it resists some metabolism, potentially lengthening the life of drugs built upon it. In materials science, derivatives unlock special properties, from dielectric layers in electronics to surface modifiers for polymers. The work I’ve seen in catalysis shows that attaching this acid to ligands or supports can control how fast or slow other chemicals react. Each breakthrough points to the same simple truth: adding even a couple fluorine atoms in the right spots alters the entire landscape of performance in the final product.
Toxicology and Environmental Risks
A reflex to associate fluorinated chemicals with danger exists for good reason. There’s evidence that benzoic acids in general break down in the environment, but boosting the number of fluorines often stiffens their resistance to biodegradation. Toxicity research for this particular acid suggests a low order of acute toxicity—no immediate threat from brief skin or eye contact, but caution ramps up for chronic exposure or ingestion. The bigger picture ties into the persistence of organofluorine compounds: they last, resisting breakdown in nature, and easily escape traditional wastewater treatments. Environmental chemists recommend tight waste handling, careful inventory tracking, and research into greener synthesis or degradation routes so future generations won’t inherit a buildup of “forever chemicals” where they're unneeded.
Looking Forward: Future Prospects
Interest in 2,6-difluorobenzoic acid likely won't fade soon. Growth in green chemistry pushes for new ways to make and degrade fluorinated acids with less impact. Advances in drug discovery mean more labs turn to unique building blocks like this to create longer-lasting, more selective drugs—while regulatory agencies pay closer attention to any persistent organic pollutants. Expect to see increased research into catalytic systems that recycle or benignly destroy these acids. Creative folks are already working on process intensification and biocatalysis to lower the energy and resource costs tied to old-school synthesis. The push to balance innovation with sustainability means every use case gets challenged for its necessity and impact.
Chemistry at the Core: Breaking Down 2,6-Difluorobenzoic Acid
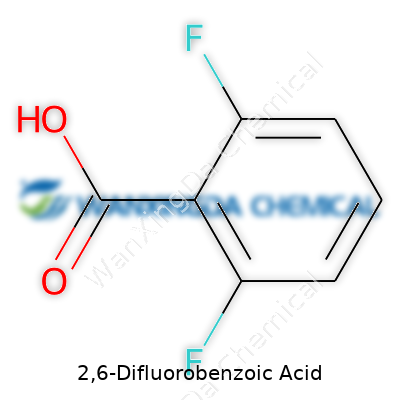
Think about a benzene ring—it’s a six-carbon loop, and chemists know it as a building block for all kinds of molecules. Now, stick a carboxylic acid group on that ring, and you’ve got benzoic acid, a compound you’ll find in many preservatives. The twist with 2,6-difluorobenzoic acid comes in with two fluorine atoms, each sitting on the second and sixth positions around the ring. Their presence looks minor on paper, but it’s game-changing for the molecule’s behavior.
Nothing in nature—or in a lab—happens by accident. Changing a hydrogen for fluorine transforms how electrons move through the structure. That carboxyl group still pulls weight with its double-bonded oxygen, jutting off the ring, but the added fluorines reshape the molecule’s personality. Fluorine’s pull is much stronger than hydrogen’s; it grabs electrons and holds on tight. With two of them staring each other down across the ring, the benzoic acid isn’t quite as reactive as it used to be. This change has real consequences for chemistry labs and manufacturers who rely on specific reactivity in their recipes.
Why Detail Matters: Everyday Experience in Research
Over the years, I’ve learned that small substitutions have outsized effects. In undergraduate labs, students struggle with synthesis reactions, frustrated when the yield drops. More often than not, it takes someone to point out a halogen sitting on the benzene ring, quietly shifting the acidity and tweaking the molecule’s relationships with solvents. 2,6-difluorobenzoic acid doesn’t act like its cousins—adding those two fluorines gives it both a sharper edge and a stubborn lower solubility in water. Extraction gets tougher, and crystallization drags out. I’ve seen teams spend hours tinkering with solvents and temperature, just to coax out a clean sample.
It’s not just about organic chemistry labs. Pharmaceutical researchers track every subtle difference in molecules. Adding a pair of fluorines, especially in these particular slots, can extend shelf life or completely block an enzyme from doing its work. That means safer drugs in some cases, or longer-lasting formulations for industries trying to avoid chemical spoilage. On the other hand, making new compounds from difluorinated versions is trickier. These stubborn molecules resist transformations that work fine on other benzoic acids.
What the Chemistry World Can Do
Rather than run into the same roadblocks with stubborn compounds, teams can take a cue from polymer chemists and improve predictability. Up-front computational modeling offers a good roadmap. The way fluorines tweak electron distribution often shows up clearly in simulation, saving hours of guesswork at the bench. Adjusting standard protocols—changing solvents, rethinking purification processes or using catalysts that specifically handle electron-poor aromatic rings—builds better solutions. The more shared protocols circulate, the less time new researchers will waste on the same trial-and-error cycles.
All details count. Understanding why 2,6-difluorobenzoic acid stands out—right down to the placement of those tiny fluorine atoms—translates to stronger research and safer products. Progress in chemistry comes down to listening to what molecules tell us, and respecting the ways a simple substitution can shape everything downstream.
Some chemicals stay out of the spotlight even though they play crucial roles in the world around us. 2,6-Difluorobenzoic acid fits this mold. Few people outside specialized labs have even heard the name, but without it, key industrial and pharmaceutical advances would stall out.
Why the Chemical Structure Matters Beyond the Lab Bench
Take a look at this molecule’s structure. Adding fluorine atoms changes how it behaves in reactions, making it far more valuable than plain benzoic acid. The two fluorines aren’t just for show—chemists count on them to influence how the molecule reacts and how it slots into chemical chains. In organic synthesis, little tweaks can lead to big changes in performance and stability.
From Benchtop to Industry: Main Applications
As someone who’s waded through more technical papers than I care to admit, I’ve learned that 2,6-difluorobenzoic acid often shows up in pharmaceutical research. Drug developers look for molecules that can both stick tightly to target proteins and resist breaking down too quickly in the body. The difluoro tweak delivers on both fronts. Medicinal chemists use this acid as a building block—a finished drug may not look much like it, but the backbone owes something to 2,6-difluorobenzoic acid’s role early in synthesis.
Beyond medicines, agricultural chemicals also get a boost. Herbicides and fungicides made from fluorinated benzoic acids tend to last longer in the environment, which boosts yields but sets off debates about environmental safety and bioaccumulation. Tough trade-offs, no easy answers—I’ve watched researchers wrestle with those firsthand.
Materials science leans on this compound as well. When you’re designing new liquid crystals for high-end displays or searching for customized polymers with unusual resistance to heat or chemicals, you often find yourself reaching for specialty acids. The electron-withdrawing punch packed by those two fluorines makes it easier for chemists to build up complexity or bond to metals, which matters in both electronics and coatings for medical devices.
Balancing Benefits and Risks
It’s impossible to talk chemistry today without nodding to environmental and safety concerns. Fluorinated aromatics like this one don’t break down easily. Industrial runoff can linger, and while most facilities now trap and process chemical waste, enforcement remains patchy across the globe. Policymakers argue—sometimes endlessly—about acceptable thresholds and regulations. I remember following news about persistent organic pollutants and seeing how slow real change can be once profits are on the line.
Solutions and Moving Forward
Green chemistry isn’t a buzzword anymore; it’s a necessity. Researchers have started developing milder, cleaner methods to produce specialty acids, including fluorinated ones. Catalysts, for example, now often use fewer rare minerals, cutting costs and the environmental toll. Some teams test biodegradable analogues, but so far, nothing matches the unique blend of stability and reactivity found here.
I’ve seen serious progress when academia, industry, and government collaborate. Funding basic research opens up smarter ways to assemble complex molecules and handle residues responsibly. Regulations can foster competition by rewarding companies for safer practices, not just higher profit margins. These shifts don’t happen overnight, but steady work pays off.
2,6-Difluorobenzoic acid embodies both the promise and puzzle of modern chemistry—a tool with profound potential, so long as we handle it thoughtfully and with eyes wide open.
Small Details, Big Impact
Every chemist has felt the frustration that comes from unreliable ingredients. 2,6-Difluorobenzoic acid, used frequently in pharmaceutical synthesis and specialty chemical development, reminds me how much hinges on these small details. Purity, especially, has a real grip on the quality of your results.
A quick look through chemical supplier catalogs shows most reputable sources offer 2,6-difluorobenzoic acid with a minimum purity around 98%. Some push this higher, to 99% or above, targeting researchers who face tight regulatory checks or those chasing cleaner yields in synthesis. The difference between high and slightly lower purity might not always register in initial reaction screens, but trace contaminants can become a nightmare in scale-up or in regulated industries.
Knowing what else lurks in your bottle matters because even a small amount of impurity can throw off reaction pathways or complicate downstream purification. I once watched a straight-forward coupling turn messy because a prior batch of 2,6-difluorobenzoic acid included a few percent of water and unreacted starting material. Extra column washes, extra verification runs—these take more time and money. Trust in reported purity saves all those headaches.
How Much Should You Get?
Some labs need grams, others need kilograms. For academic research, 5-gram and 25-gram bottles cover a lot of experiments without stretching grant budgets. Lots of suppliers offer these sizes, but prep-scale and industry groups push for bigger options, such as 100 grams, 250 grams, or even 1-kilogram containers.
The bigger sizes fit manufacturing and pilot-scale testing. Researchers working on scale-up can’t afford to chase down half-empty tiny bottles, and the consistency between bottles goes a long way during method validation or regulatory review. I’ve seen the peace of mind that comes from having one large, properly sealed container—no batch-to-batch surprises when each synthesis step counts.
The Importance of Good Sourcing
Current regulations and quality policies demand clear documentation. Labs and production lines now expect chemical traceability to reduce risks and keep results consistent. Purity specs on a label reflect deep work upstream—with carefully chosen starting materials, tightly controlled processes, and strong analysis. I’ve found that being picky about who you buy from pays off. Suppliers who back up their claims with certificates of analysis and clear paperwork help you pass inspections without last-minute scrambling.
It’s hard to overstate how much time is lost if you need to double-check purity yourself each time you open a new container. Reliable suppliers make that extra testing the exception and not the rule.
Solutions Beyond the Label
Building lasting supplier relationships goes beyond comparing prices and purity grades. Ask questions, request sample documentation, and learn what’s in that bottle before it hits your bench. For scale-up or regulatory filings, request lot-specific analyses. Push for packaging that matches your true consumption—less waste, safer storage, fewer surprises.
As demand grows for specialized chemicals like 2,6-difluorobenzoic acid, smart sourcing habits and attention to purity and packaging will continue to separate efficient, successful projects from ones bogged down by uncertainty. A little careful sourcing today spares a surprising number of headaches tomorrow.
Few people wake up thinking about 2,6-Difluorobenzoic acid, yet anyone working around chemicals knows this compound deserves respect. Just looking at the name, you can almost hear the clink of glass bottles in a lab. Many chemists and lab workers have stories about what happens when someone gets careless with strong acids or sensitive powders, and those stories don't end well. Safe handling doesn't just protect you, it keeps your workplace from turning into a costly mess.
Clear Labeling and Good Organization
Most accidents start when someone grabs the wrong bottle. I once watched a colleague mistake a benzoic acid derivative for a simple buffer, leading to hours of wasted work and a small scare. Label every container clearly and don’t trust faded markers or sticky notes. Store chemicals in a way that keeps similar hazards together, but not mixed with things they can react with. Separate your acids from your bases and never toss unknown powders together — that's just asking for trouble.
Choosing the Right Storage Spot
2,6-Difluorobenzoic acid doesn’t belong near a sunny window or next to some whirring heat source. Most labs I’ve seen use cool, dark cabinets for sensitive organics. Fluctuating temperatures encourage degradation and sometimes even dangerous gas formation. Glass and high-quality plastic containers hold up best, since this acid can corrode metal over time. If you’ve worked in older labs, you know what a corroded shelf looks like — rusty holes aren’t just ugly, they signal years of neglect.
Personal Protection Can’t Be Skipped
Even if you’re careful, accidents happen. I always grab my goggles and gloves before opening any bottle labeled “acid,” fluorinated or not. 2,6-Difluorobenzoic acid isn’t the most threatening chemical on the shelf, but skin and eyes won’t thank you for splashes. Gloves and lined lab coats save you from many small mishaps. Good ventilation keeps fumes at bay, especially in crowded settings where several people may open chemicals at once.
Thinking Ahead About Waste and Cleanup
Safe storage only works if planned alongside safe disposal. Don’t dump this acid down the drain; it harms water systems and violates regulations. Local rules might be strict, so set aside a marked container for collecting waste and train anyone who works nearby on what goes into it. A simple chart kept near the chemical shelves can make the right choice second nature. I’ve seen labs fined because an intern didn’t know the difference between clean and dirty disposal, so communication really does matter.
Learning From Others' Experience
Trust grows in a well-organized lab. Respect starts with the basics: good labeling, safe storage, and proper handling. These steps make it less likely for someone to cut corners. Audits and regular training refresh habits and turn rules into muscle memory. If management treats chemical safety like an afterthought, mistakes pile up. If everyone feels responsible, fewer accidents happen. The goal isn’t to make life harder — it’s to make sure nobody gets hurt, and nothing gets ruined.
Dealing with chemicals like 2,6-Difluorobenzoic Acid in any setting—whether it’s a university lab bench, a pharmaceutical research office, or a chemical manufacturing site—brings its share of risks and responsibilities. The first step to safe handling goes beyond just knowing the name and formula. It’s about having a solid grasp on Certificates of Analysis (COA) and Safety Data Sheets (SDS) for every batch and bottle. That need feels personal for anyone who’s ever had to answer for what happens when accidents happen or results come back funny.
What a COA and SDS Cover That the Label Never Will
You pick up a jar with a printed label: 2,6-Difluorobenzoic Acid, CAS number, maybe a purity value, and the weight. That information barely scratches the surface. The COA provides batch-specific proof of quality. You see documented values for purity, moisture content, melting point, and sometimes trace metals or residual solvents. Those numbers may seem fussy, but in the world of fine chemicals, a half-percent impurity can send an experiment or production batch off course—no joke for anyone chasing consistent results.
The SDS is about safety—real, practical, everyday safety. It covers toxicity, safe handling, storage, disposal, and emergency steps. I remember a time a student thought “organic acid” meant mild, and splashed benzoic acid in an open lab. The right SDS on hand helped everyone respond fast and right. With 2,6-Difluorobenzoic Acid, there’s potential for skin, eye, or respiratory irritation. The SDS lays out where to find the right gloves and goggles, how to vent a spill, and what to expect if someone gets exposed.
COA and SDS: Not Just Bureaucratic Paperwork
Some see these documents as just part of red tape, checked boxes at the receiving dock or lost in digital filing systems nobody looks at again. But, skipping this step can cost more than a failed assay or flagged audit. Chemists, lab techs, and purchasing managers lean on these sheets to troubleshoot issues, verify compliance with local and international regulations, and protect workers from exposures that wreck lives.
A COA helps answer the tough questions from the quality team or regulatory inspectors: Why did this reaction yield drop? What changed between batches? The SDS goes hand in hand with right-to-know laws and GHS standards adopted across the globe. Inspectors expect to see records available and training based on real, up-to-date data, not just generic warnings.
Where the System Breaks Down—And How to Fix It
Sometimes the trouble isn’t about the documentation itself, but about who’s keeping it current or even checking it. Paper copies can go missing, digital links can break, and language barriers show up in multinational settings. Some suppliers only provide generic or minimal information. That’s not good enough when real hazards or trace contaminants are on the table.
One solution lies in clearly written procurement contracts demanding up-to-date COA and SDS documents for every purchase. Another fix comes from onsite training—not just about reading these documents, but acting on the information inside. We can’t assume everyone on site knows chemical names or hazard codes offhand. Making COA and SDS access and training routine helps everyone work smarter and safer.
Trust, Science, and the Lab Bench
People working with chemicals like 2,6-Difluorobenzoic Acid rely on good information. Trust forms when suppliers share clear, batch-specific data, and when every person stepping up to a workbench understands both the science and the risks behind what that data means. Safety, reproducibility, and compliance ride on having those facts in hand—not buried in a filing cabinet or sitting in someone’s unread inbox.