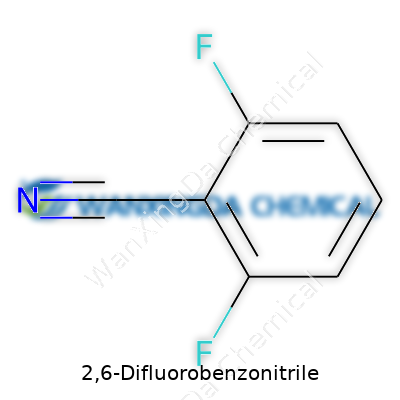
2,6-Difluorobenzonitrile: A Chemist's Perspective on Value, Challenge, and Possibility
Historical Development
Folks who’ve tinkered in the organic chemistry lab long enough will tell you that the story of 2,6-difluorobenzonitrile isn’t about eye-catching headlines but about deliberate progress. The demand for halogenated aromatics spiked after World War II, as new pharmaceuticals and exotic plastics called for molecules with very specific behaviors. By the mid-20th century, chemical companies realized the power of small tweaks—swap out some hydrogens for fluorines, add a nitrile group, and you wind up with compounds like 2,6-difluorobenzonitrile. Chemists appreciated how the two fluorine atoms and the nitrile shifted both reactivity and stability, opening doors to new syntheses. Decades later, this seemingly unremarkable chemical found its way into the foundation of specialty intermediates for pesticides, dyes, and even some advanced materials, riding on the reputation of the cyanide group’s versatility and the electronegativity of fluorine. Chemical innovation rarely comes from grandstanding; it happens quietly at the bench, one functional group at a time.
Product Overview
Every time I handle a bottle of 2,6-difluorobenzonitrile, I notice how niche chemistry stands the test of time. This compound lands in a strange spot—it’s neither as common as simple benzonitrile nor as exotic as heavily fluorinated aromatics used in cutting-edge electronics. Instead, it fills gaps in synthetic pathways for drugs, herbicides, and specialty monomers. Not flashy, but indispensable to certain researchers. If you’re mapping out new molecular scaffolds, this molecule can save days, maybe weeks, of synthetic effort. Its commercial value hinges on demand from downstream chemicals, often as a building block where the placement of both fluorines matters.
Physical & Chemical Properties
2,6-Difluorobenzonitrile comes as a solid, usually a white powder that packs a noticeable, sometimes pungent odor. The fluoro substituents and nitrile boost its stability and help resist some of the usual degradation tricks. Its structure sets the tone for many of its physical properties. The melting point, usually in the range of 49–52°C, means it doesn’t stick around as a liquid for long under normal lab conditions. Solubility in organic solvents like acetone or dichloromethane sits about where you’d expect for this family, and water solubility remains low, which can frustrate attempts to clean glassware thoroughly after experiments. Chemists lean on its stability and low volatility when developing multistep syntheses, and the strong electron-withdrawing effect of both fluorines and the nitrile affects reactions in predictable, sometimes stubbornly selective ways.
Technical Specifications & Labeling
Lab work doesn’t fudge numbers. Purity usually sits north of 98 percent for synthetic work, and most suppliers print chemical identifiers like CAS numbers, molecular weights, and accepted hazard statements on the container. A bottle will often show a warning for toxicity and flammability. I check for reddish tinting—that means potential impurity problems, often from improper storage or careless transfer. You learn to trust suppliers sparingly, so I still run a quick melting point check and confirm by NMR if the upcoming reaction doesn’t invite surprises. Sturdy containers stand up to accidental bumps; no one enjoys scooping up a few grams off lab benches after a spill.
Preparation Method
Back in grad school, my advisor pressed the importance of strategic thinking around preparation. You can start from 2,6-difluorotoluene or its related anilines, using oxidative steps, Sandmeyer reactions, or copper-catalyzed cyanation. The pathways can shift based on lab budget, equipment, and safety guidelines. The process often starts with halogenation before introducing the nitrile group through copper-catalyzed reactions involving copper cyanide or similar reagents. Industrial makers favor robust, scalable processes—think fewer purification steps, cheaper reagents, and tighter controls on waste by-products. Yield and purity fight each other, and experienced chemists stare at their TLC plates or chromatograms for hours, coaxing reaction conditions into a sweet spot that minimizes labor during isolation.
Chemical Reactions & Modifications
What grabbed my attention about this molecule was how those paired fluorines put a stranglehold on reaction sites along the aromatic ring. Electrophilic aromatic substitution turns sluggish. If you’re doing nucleophilic aromatic substitution, though, those fluorines become convenient leaving groups, providing controlled handles for introducing new substituents, especially on the ortho or para positions. Reduction of the nitrile group opens a path to amines, while cross-coupling reactions let researchers link new groups to the core structure. You figure out quickly that each alteration shifts both chemical behavior and possible applications. Seeing this compound link to new drug candidates or high-performance polymers in the literature confirms the kind of flexibility synthetic chemists hope for.
Synonyms & Product Names
In catalogs and research articles, 2,6-difluorobenzonitrile sometimes shows up under aliases like o,o-Difluorobenzonitrile or 2,6-bisfluorobenzonitrile. If you leaf through old chemical catalogs, you’ll find the German trade names, and in regulatory registries you’ll find its CAS number attached to alternate labels, but it’s the molecular shorthand—C7H3F2N—that lab workers scribble on fume hood sashes. I’ve never seen it sold in bulk at hardware stores or consumer outlets for good reason.
Safety & Operational Standards
Every time someone uncaps a container of 2,6-difluorobenzonitrile, they’re reminded of the need for caution. Skin contact or inhalation isn’t something you take lightly. Nitrile compounds carry well-deserved reputations for toxicity, and the organofluorine content ups the ante. Wearing nitrile gloves and working in a well-ventilated fume hood matter more than fancy credentials. I’ve seen improper storage lead to spoiled product—and lab visits from safety officers. Waste management for fluorinated aromatic compounds drags on sustainability goals, which means every lab notebook entry needs to match real efforts to minimize environmental release. Clear labeling, accessible safety data sheets, and tight controls on access show up everywhere in reputable labs.
Application Area
In industry and research, 2,6-difluorobenzonitrile rarely becomes the star of marketing slides, but its presence lurks in plenty of downstream products. Herbicide developers reach for it when working on next-generation molecules aiming to boost efficiency or tackle resistant weeds. Pharmaceutical teams use it in fragment-based approaches to drug development, where fluorines can tune receptor interactions or limit metabolic breakdown. Anyone looking to make specialty coatings, dyes, or resin hardeners may find a use for its unique reactivity. The basic structure sets off possibilities for both fine-tuning of properties and structural novelty.
Research & Development
From an R&D perspective, much of the action revolves around expanding reactions that build on the difluorinated core. Publications on new ligands, catalysts, or synthetic intermediates often refer back to 2,6-difluorobenzonitrile as a starting point. AI-assisted retrosynthesis tools flag it as an attractive intermediate for novel nodes in drug scaffolding and advanced functional materials. There's growing interest in greener manufacturing routes—people look for less hazardous cyanation steps, recyclable catalysts, and lower energy processes. Teams sprinkle computational chemistry into the mix, aiming to predict unusual reactivity before spending precious days in the lab.
Toxicity Research
Putting aside the bland warnings on safety data sheets, real-world toxicity research digs deeper. Nitrile compounds catch attention for their potential to release cyanide, and even if not acutely hazardous at microgram levels, repeated exposure or improper handling racks up health problems over time. I’ve watched debates at conferences over exactly how persistent these fluoroaromatics become in groundwater and soil, especially where herbicide use connects to agricultural runoff. Regulations continue to tighten, and research follows. Monographs document both acute toxicity, like effects on respiratory and nervous systems, and persistent, low-level exposures. Risk assessments emerge from animal studies, computational toxicology, and environmental monitoring, guiding users in refining both containment and disposal procedures.
Future Prospects
Big shifts in chemical industries challenge all staple intermediates, and 2,6-difluorobenzonitrile isn’t immune. Calls for sustainable, waste-minimizing synthesis—and stricter limits on persistent organic pollutants—push researchers to develop friendlier alternatives or greener synthetic routes. Specialty applications for advanced polymers and pharmaceuticals keep this molecule in demand. More powerful computational tools, smarter automation, and a greater willingness to collaborate across disciplines could mean new breakthroughs in reactivity, catalysis, or recycling. My own outlook expects gradual, context-driven advances—no dramatic reinvention, but instead steady improvement in both utility and safety. Whether in a start-up’s R&D pipeline or an academic group looking to break into new chemical territory, the practical value of 2,6-difluorobenzonitrile shows up right where careful chemistry meets necessity.
2,6-Difluorobenzonitrile doesn’t show up in headlines, but it quietly shapes a big part of how we make things, especially in the chemical world. I’ve met chemists who could talk for hours about ingredients and never mention this compound, yet they’d be lost in the lab without it. To most folks, it sounds like a tongue-twister. To researchers, it’s a critical building block, especially for developing products that touch everyday life and health.
Where 2,6-Difluorobenzonitrile Fits In
This compound stands out for one simple reason: it helps spark new chemical reactions. People make use of it as an intermediate — that means it forms a bridge between raw materials and finished products. Its main role? Supporting the creation of more complex materials, most commonly in making pharmaceuticals and specialty chemicals.
Ask someone who’s worked with agrochemicals, and they’ll tell you 2,6-difluorobenzonitrile helps build molecules that keep crops free of pests. Quite a few herbicides and fungicides owe their punch to precursors like this one. In medicine, it provides a key steppingstone for crafting certain drugs. This kind of chemistry lets drug developers put just the right pieces together, increasing the chances that a compound will work as designed. These custom touches make new therapies possible, particularly where tweaking one part of a drug’s structure changes how it tackles disease in the body.
Challenges and Responsibility
Handling 2,6-difluorobenzonitrile brings its own hurdles. Most chemists will confirm that just because a chemical is useful doesn’t mean it’s simple to work with. This one calls for care — contact with skin or getting it in the eyes leads to irritation. Fumes aren’t friendly either. Solid safety protocols, training, and real respect for protective gear are non-negotiable in labs working with it.
Production also creates environmental questions. Chemicals that build up in air, soil, or water demand thoughtful management. Too many stories in the news track back to misplaced chemicals causing harm far outside the lab: contaminated waterways, sick fish, and worried communities. Tight controls, clear waste routes, and regular reviews of disposal habits can limit that harm. Smart policy isn’t about stifling innovation. It’s about protecting the future while letting science move forward.
What Could Change?
Everyone in chemistry circles knows innovation needs stewardship. Greener chemistry practices deserve more support, not just lip service. That means funding alternatives, setting clear emissions targets, and inviting more voices — from workers on the ground to people living near industrial plants — into the conversation. Some companies already lean into plant-based chemistry or retooled processes, seeking out ways to trim down dangerous byproducts. Progress takes time, but pressure from scientists, regulators, and the public keeps the needle moving.
It’s tempting to overlook the humble intermediates, but molecules like 2,6-difluorobenzonitrile show how science works in the background, crafting the building blocks for medicine, farming, and materials technology. Making room for honest discussion, careful stewardship, and continued innovation remains essential if we want to see these benefits last and reach everyone safely.
2,6-Difluorobenzonitrile Packs More Than Just a Chemical Punch
I remember walking into a lab for the first time, looking at row after row of reagents lined up on the shelf. One label caught my eye—2,6-Difluorobenzonitrile. To most, the name means little, sounding like one of the thousands of bench-top compounds. To those familiar with synthetic chemistry, this one calls for sharp focus. Its toxicity isn’t legendary, but its risk sneaks up on the careless.
Chemical safety isn’t about paranoia. It grows out of real stories: accidents, near-misses, unexpected spills. I’ve seen nitriles handled without care and watched coworkers fumble through glove changes, thinking little could go wrong. That kind of daydreaming is where mishaps begin. 2,6-Difluorobenzonitrile brings respiratory hazards, skin and eye irritation, and might even cause longer-term health problems if used outside a fume hood or with bare hands. The lesson here: treat it with respect, not routine.
No Room for Shortcuts
Most organic chemists reach for nitrile gloves as a first line of defense. Standard latex ones won't hold up, given how organic solvents sometimes leech through. You work with materials like 2,6-Difluorobenzonitrile by pulling on a tested pair, sometimes even double-gloving if the process calls for it. Aprons and splash goggles matter too. Safety glasses won't cover the side gaps, especially if there’s a risk of drops or mist. Contact with this compound creates more than a temporary sting.
One part that frustrates me is how easy it is to overlook the basics. Ventilation. No one wants to spend their afternoon in a fume hood, especially when it’s crowded or claustrophobic. Contact with nitrile vapors, though, has consequences. Acute exposure triggers coughing and shortness of breath. Chronic inhalation can lead to more complications. I once skipped the hood during a rushed reaction setup and came away with a nasty headache. Lesson learned—airflow matters.
Fact-Driven Safety Isn’t Overkill
A compound like this should always come with a Safety Data Sheet stapled to the wall or posted in your digital library. Read it—don’t just wave the paperwork off. Regulations ask for thorough labeling, correct storage, and strict waste disposal. These rules don’t come from thin air. Environmental agencies track rates of chemical exposure and their findings influence workplace limits. Germany’s MAK commission, for example, looks at safe limits for compounds such as 2,6-Difluorobenzonitrile, and these guidelines help set habits in labs worldwide.
With nitriles, waste shouldn’t go down the drain or in regular trash. Special containers store spent solvents and contaminated gear. Risk doesn’t end once you leave the bench. If a fire breaks out, one can’t get cavalier—nitriles burn hot, and the fumes get toxic fast. Local authorities recommend CO2, foam, or dry chemical extinguishers, since water doesn’t always cut it.
Real Solutions Are Rooted in Real Habits
People sometimes focus only on personal protection and overlook the training side. Seasoned chemists know that walking a new researcher through the ropes—pointing out proper disposal, correct labeling, careful storage—can prevent months of headaches or worse. I try to leave room for questions, never brushing off those “What if?” moments. That attitude goes farther than signs on the wall ever will.
Each time someone reaches for 2,6-Difluorobenzonitrile, a mental pause should follow. Not fear, just routine clarity. Wear the right gear, use the right hood, and keep your eyes open. Safety isn’t a box checked at the start of a shift. It’s a practice that grows, grounded in daily decisions and shared knowledge. That’s the real barrier against laboratory regret.
Digging Into the Formula
Plenty of folks feel a little uneasy scanning complicated names like 2,6-Difluorobenzonitrile. It doesn’t exactly sound friendly, but this compound comes up often in labs and on shelves for a reason. The molecular formula is simple enough: C7H3F2N. Picture a benzene ring with two fluorine atoms hanging off the second and sixth carbon positions, and a nitrile group attached at the first. That’s the full chemical structure behind the long name.
Why Structure Holds Weight
I’ve spent time in both teaching labs and industry settings, and I can say from first-hand experience that the placement of those two fluorines changes everything. Add just one fluorine and the chemical behaves differently in reactions. Stick two in and it shifts response even more. Chemists pay attention to these tweaks because it often makes or breaks a project. The nitrile group, which is a carbon triple-bonded to nitrogen, turns the molecule into a reactive intermediate—a building block for making dyes, pharmaceuticals, and agrochemicals.
Synthetic chemists love these benzonitrile compounds. They often wind up as the “parent” structure for many tricky organic molecules. For instance, doctors rely on medicines that start with benzonitriles. Crop scientists use them as building blocks in pesticides that protect food. The fluorine atoms in particular not only resist breakdown in harsh conditions but can also change how drugs interact in the body, sometimes making medicine last a bit longer or pass a certain barrier.
Risks and Hazards Nobody Can Ignore
Fluorinated chemicals catch a lot of heat lately, mainly because some of them stick around in water and soil for decades. Not every compound with fluorine creates problems as extreme as headline-making “forever chemicals,” but it does raise an eyebrow. Workers handling 2,6-Difluorobenzonitrile use gloves, goggles, and good ventilation for a reason. Nitriles, especially aromatic ones, can be toxic if you’re careless. Breathing in vapors or getting them on your skin sets you up for trouble. I’ve watched colleagues develop nasty headaches or skin irritation after exposure to similar compounds, so the safety advice isn’t just overcautious.
Science and Responsibility
What we do with compounds like 2,6-Difluorobenzonitrile matters. Chemical manufacturers and labs need to stay vigilant about how these materials move through drainage and air. Following strict waste protocols helps keep the compound from ending up where it doesn’t belong. There’s also a push in the field to design molecules that work as well as these fluorinated benzonitriles but break down more easily in the wild, cutting down long-term risks.
Chemistry rewards folks who look closely at small details. The fluorine atoms on a benzonitrile ring may seem minor, but they steer everything from reactivity in a beaker to safety in a workplace and environmental footprint years down the line. The more time I spend in this field, the more I appreciate the careful balance between invention and responsibility. We need both if the work is going to count for something greater than a name on a bottle.
Most people don’t think about chemicals like 2,6-Difluorobenzonitrile until there’s a problem. Chemists and lab techs handle this compound carefully for a reason. It isn’t some household item you keep on the shelf above the sink. This stuff calls for respect—because it can cause trouble if ignored.
Experience has taught me that chemicals with cyano and fluorine groups carry real hazards. Vapors sneak past leaky lids. Direct skin contact leaves burns or, worse, sets the stage for long-term health headaches. Some compounds react with air or water and 2,6-Difluorobenzonitrile falls in that zone—volatile, sensitive, unforgiving if mishandled. So, forget the casual approach; this one needs thoughtful storage.
Temperature and Containment Matter
Leaving this compound at room temperature in a warm lab means increased vapor release and possible decomposition. Refrigerators or cool, dry cabinets provide a better home. Yet, not every fridge is suitable—food fridges are out of the question. Every chemical storage expert I’ve known insists on a separate, explosion-proof unit for volatile organics. This isn’t paranoia; refrigeration slows down harmful reactions, reduces exhalation of fumes, and contains accidental releases. Cool and dry storage saves a lot of grief down the line.
Airtight Sealing Reduces Risk
Bad lids are almost as big a problem as lousy cabinets. I’ve witnessed more than one incident where “just tight enough” lids let fumes escape or—worse still—allowed moisture to sneak in. The cyanide part of this molecule will not play nice with water or acids, leading to release of toxic gases. That’s why solid, screw-top containers made from materials with proven resistance to organics make sense. Glass works, but only if you trust your shelving and don’t stack containers like soup cans. Some labs invest in high-grade plastics, which stand up well over time, provided the chemical compatibility checks out.
Clear Labeling and Segregation Count
Even smart folks make mistakes during the rush of a busy day. A clear, bold label with the chemical’s name and hazard info spares everyone from that sickening “what’s in this bottle?” moment. Mixing up chemicals—or worse, knocking 2,6-Difluorobenzonitrile off a shelf onto something reactive—serves up real trouble. Segregation from incompatible substances such as strong bases, acids, or oxidizers is not an optional step; it’s crucial. Storage plans that organize by hazard, not alphabet, give the best line of defense.
Why It All Comes Back to Health and Accountability
Proper storage isn’t just about avoiding fines or ticking off standard operating procedures. I’ve seen too many stories where poor chemical practices led to respiratory injuries or long-term disease, all of which could have been prevented. Nobody likes taking time out for exhaustive inventory and labeling duties, but the alternative will cost much more than time. Following best practices for containment, refrigeration, airtight sealing, labeling, and segregation keeps the workplace—and everyone in it—safe. That’s not bureaucratic overkill; it’s simple respect for science and those who work with it.
Getting to Know Its Personality
If you’ve ever worked in a lab or found yourself curious about specialty chemicals, you might already know that some molecules develop reputations for their quirks. 2,6-Difluorobenzonitrile falls squarely into that category. This compound doesn’t just sit quietly in a bottle. It brings a certain attitude to the workspace, thanks to how its structure influences what you notice right away: a sharp smell, a low melting point, and a form that’s pretty easy to recognize.
Crystals or Liquid: Catch It If You Can
At room temperature, you won’t find this compound masquerading as a powder or sticking around as a solid for long. Instead, it tends to show up as a colorless to pale yellow liquid. Its melting point sits below room temperature, usually hovering somewhere around the upper single digits in Celsius. Put the bottle in a cool spot, though, and it solidifies in delicate crystals.
I remember the first time I cracked open a bottle of this compound: the long, needle-like formation that appeared along the edge of a beaker stuck with me. These crystals look clean and sparkly, which can trick you into thinking they’re easy to handle, but the volatility means you don’t leave the cap off for long. Evaporation can sneak up quickly and leave a lingering, acrid aroma, the kind that signals you’d better check your fume hood.
Solubility: Where Does It Go?
One practical thing about 2,6-Difluorobenzonitrile is how it likes to keep to itself around water. If you mix it with water, it mostly parts ways and settles out, thanks to its low solubility. On the other hand, bring out some organic solvents—things like ethanol, acetone, or ether—and it blends right in. Organic chemists often rely on this knack for dissolving in non-polar environments when they’re synthesizing pharmaceuticals or developing complex intermediates.
Density, Boiling Point, and Real-World Effects
You can tell a lot about a compound from its density. This one tilts slightly heavier than water, with a pretty typical range for aromatic nitriles. When it comes time to heat things up, the boiling point stays modest—high enough to work with in the lab without causing too much drama but not so high that a careless hand on the Bunsen burner spells trouble.
The practical upshot: in industries where precision matters, these seemingly mundane details make a difference. Improper temperature control leads to rapid evaporation, wasted chemicals, and sometimes safety issues. Add in the sharp odor and you’ll see why good ventilation isn’t up for negotiation. For teams scaling reactions, calculating how much will actually make it through a procedure becomes an everyday math lesson.
Handling Challenges and Safer Labs
Every material comes with trade-offs. The low boiling point and volatile nature mean even seasoned chemists keep a watchful eye. Splash a little on your skin or inhale that vapor and you’ll know right away that lab coats and gloves aren’t optional. Yet, these physical quirks also play to its strengths. The volatility and solubility profile make this molecule useful in fine-tuned synthesis.
Practical experience tells you what the textbooks gloss over. If you treat 2,6-Difluorobenzonitrile with the respect it’s earned—from clean glassware to diligent air exchange—there’s no reason it can’t be a useful partner at the bench. Understanding these small details takes practice. It’s one thing to memorize numbers on a chart, quite another to see how this compound responds to the air, the heat, and your own care behind the bench. That’s where chemistry becomes not just a science, but a craft.