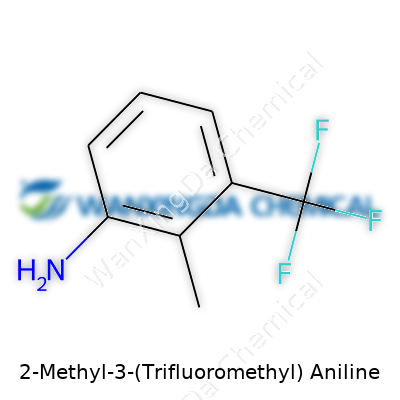
2-Methyl-3-(Trifluoromethyl) Aniline: A Deep Dive into Its Chemical Journey and Industrial Relevance
Historical Development
Chemistry thrives on both innovation and refinement, but some stories don’t get told outside the lab. 2-Methyl-3-(Trifluoromethyl) Aniline didn’t become a fixture in the toolbox just by accident. Looking back, you find its roots tangled up with the rise of fluoroaromatics right after chemists started chasing stability perks and unique reactivity in pharmaceuticals and agrochemicals. These aren’t just minor tweaks. Adding the trifluoromethyl group flipped the script completely on what simple anilines could do. Through the late twentieth century, improved analytical tools and more efficient fluorination methods carried this compound out of hazy patent literature and into the mainstream, showing up whenever someone wanted molecules with more punch and less unpredictability. People in synthesis started to pay attention, realizing those modifications opened up a wider range of chemical possibilities.
Product Overview
2-Methyl-3-(Trifluoromethyl) Aniline doesn’t get the same spotlight as blockbuster chemicals, but it does a steady job in complex settings. This compound stands out because the trifluoromethyl group reshapes both its electron density and its physical characteristics. In practice, that small molecular tweak pays off where other anilines fall flat, especially in the design of active ingredients. You won’t find it on the shelves of everyday consumer chemicals, but step inside a research lab or an industrial pilot plant and you’ll realize its popularity. Chemists looking to experiment with electron-withdrawing effects gravitate here for those non-intuitive results. Tracing its buying patterns over the years, demand tells a quiet story of specialty needs rather than mass-market appeal, yet its influence sprawls much wider when you track the number of patents and technical papers linking back to this aromatic building block.
Physical & Chemical Properties
Anyone who's worked with anilines knows subtle shifts in their structure can change the whole experience. With 2-Methyl-3-(Trifluoromethyl) Aniline, that trifluoromethyl group isn’t just about heft. Stability and volatility change, the boiling point climbs higher, and the air smells sharper and more medicinal—hard to mistake on a bench crowded with simpler aromatic amines. This compound brings a denser, more persistent vapor, making ventilation a priority even before handling begins. Its reactivity owes much to that fluorine load, making it less prone to unchecked side reactions compared to close cousins. Chemical durability under tough conditions means more options during multi-step synthesis. You get less risk when scaling reactions, since both solubility and stability track more predictably in solvents popular with fine chemical makers. Color, odor, solubility, and melting point aren’t always dramatic, but the mix lands just right for chemists needing a reliable, easily manipulated intermediate.
Technical Specifications & Labeling
Experience with regulatory paperwork and container labeling for specialty chemicals tells you that 2-Methyl-3-(Trifluoromethyl) Aniline often shows up in high-purity grades, tailored for nuanced synthetic work. Specification sheets certainly demand clarity about percentage purity, water content, presence of by-products, and trace metals, since labs depend on reproducible results. Its labeling practices mirror those of other hazardous aromatic amines, with attention drawn to potential health risks and storage rules. On the supply side, you see ever-tighter tolerances demanded by pharmaceutical customers, leading to a chase for batch-to-batch consistency. Documentation in this field starts to resemble quality control proceedings rather than basic product blurbs. In practice, this attention to detail pays dividends during scale-up, where minor contaminants would scuttle expensive syntheses.
Preparation Method
Early routes for making 2-Methyl-3-(Trifluoromethyl) Aniline forced chemists to wrestle with cumbersome reagents, but persistence and new thinking changed that game. Today, the most trusted preparation leverages nucleophilic aromatic substitution or selective halogen exchange—especially once direct trifluoromethylation methods picked up precision. It’s easy to forget the labor that once went into working up these reactions, handling aggressive reagents, or coaxing reluctant intermediates along the way. Improvements in catalyst design, and the move toward greener fluorination strategies, helped bring both safety and yield under control. In academic settings, students learn quickly that reaction optimization for this compound means more than just playing with temperature or solvent choice; they juggle reactivity, logistics, and thorough purification, since even small impurities affect every downstream reaction. From my own time in process development, the margin for error shrinks as you move from flask to reactor, meaning that subtle improvements in synthesis make or break the case for using this building block in scale-up.
Chemical Reactions & Modifications
Working with 2-Methyl-3-(Trifluoromethyl) Aniline, chemists step into a world where small changes flip expected outcomes. The amino group likes to be modified, but the trifluoromethyl group resists attack, protecting the ring from substitutions that would cripple less robust anilines. Coupling reactions, diazotization, and acylations all go forward, often with cleaner profiles, giving medicinal chemists a playground for new analogs. My years in chemical development taught me that this compound lets in creativity—one can attach sulfonates, heterocycles, or other substituents, building structures that keep both chemical and biological properties in tight control. That trifluoromethyl handle keeps side reactions at bay, while the methyl stick changes the steric profile, offering a range of outcomes that fuel invention in lead optimization or material science.
Synonyms & Product Names
Depending on context, this compound’s name shifts among its synonyms. Some call it 2-methyl-3-(trifluoromethyl)aniline, others prefer 3-(trifluoromethyl)-o-toluidine. In the literature, you might find it as 2-methyl-3-(trifluoromethyl)benzenamine. Catalogs and procurement systems rarely stick to a single preferred naming convention, and chemists developing new processes double-check CAS numbers to stay out of trouble. Anyone who’s ordered the wrong fluorinated aniline knows that even small naming differences can stall a project for weeks.
Safety & Operational Standards
Labs working with 2-Methyl-3-(Trifluoromethyl) Aniline don’t just rely on generic precautions. By drawing on both regulatory guidance and years of practical experience, researchers adopt splash protection, fume hoods, and tight inventory control. The compound’s volatility means that closed systems and active ventilation are standard, and experienced chemists deal with it as they would any potentially toxic aromatic amine. Standard operating procedures put spill containment and rapid cleanup on the checklist, not only for compliance but also because a single mistake can disrupt weeks of preparation or threaten workplace health. Waste disposal tracks with rules for halogenated organics, requiring more planning. My own view sharply favors routine exposure monitoring, since underestimating cumulative hazard catches even experienced teams by surprise.
Application Area
Pharmaceuticals, agrochemicals, and specialty polymers all build from 2-Methyl-3-(Trifluoromethyl) Aniline as a starting point. In drug development, it boosts metabolic stability, helping molecules survive bioactive environments. Agrochemical developers count on its resilience in field conditions. Materials scientists incorporate it into advanced polymers and dye precursors—these applications benefit from its unusual combination of reactivity and inertness. Research into this compound fuels patent filings and new candidates for clinical trials. The trifluoromethyl group brings both hydrophobicity and metabolic resistance, which helps products hang on longer and perform consistently. This isn’t just theoretical: actual finished goods in the market draw from this chemistry, and behind every trial batch are process steps relying on its reliability.
Research & Development
Innovation often starts with a few grams measured out on a well-worn balance, and chemists push the limits of 2-Methyl-3-(Trifluoromethyl) Aniline in a bid to build better medicines or protective materials. Current projects spotlight site-selective modifications and scalable fluorination methods. Interdisciplinary teams—combining organic, analytical, and process chemists—work the details, digging through data to spot improvements in yield and selectivity. A big push in both academia and industry focuses on sustainable synthesis, trying to bypass hazardous reagents and shrink energy requirements. Every year, a flood of papers and patent filings expands what we know about this compound’s uses, and cross-sector collaborations accelerate the pace, feeding back into further process refinements and greener approaches.
Toxicity Research
Long-term handling brings an appreciation for hidden risks, and 2-Methyl-3-(Trifluoromethyl) Aniline is no stranger to close scrutiny. Researchers run tests to measure direct toxicity and metabolic byproducts, as regulators watch for any sign of bioaccumulation or carcinogenicity characteristic of other aromatic amines. Most labs insist on additional hazard communication, building from animal studies and occupational exposure data. The priority is always early identification of potential risks—changes to protective equipment or disposal protocols follow each round of findings. Personal experience underlines that even with best practices, unexpected exposures or minor spills need fast response and thorough documentation.
Future Prospects
Looking ahead, opportunities grow as more industries realize the benefits of installing the trifluoromethyl group on flexible aromatic frameworks. Regulatory constraints keep process chemists scanning for greener synthesis, and demand for stable, high-performance molecules drives every investigation into both biological and material applications. Improvements in direct functionalization and catalyst design make more sustainable chemistry seem less like wishful thinking and more like realistic progress. Continued work into structure-activity relationships and metabolism will shape how this compound fits into both old and new product pipelines. Ultimately, the lessons learned from decades of incremental improvement sharpen the focus on safety and sustainability, ensuring 2-Methyl-3-(Trifluoromethyl) Aniline keeps its foothold in the shifting landscape of specialty chemicals and modern manufacturing.
Odd-sounding chemicals fill up plenty of shelves in labs, many of them rarely mentioned outside those fluorescent-lit rooms. One that keeps popping up for chemists and industry folks is 2-Methyl-3-(Trifluoromethyl) Aniline. This mouthful of a name belongs to a compound that packs a punch both in its chemistry and its relevance for today’s research and development.
A Building Block in Modern Chemistry
Molecules like 2-Methyl-3-(Trifluoromethyl) Aniline show up as starting points in bigger projects. In my own experience bouncing between university research and conversations with people in pharmaceutical labs, these compounds matter because they help create new drugs. Drug discovery doesn’t just come from thin air—it’s a slow grind of testing, breaking, and rebuilding molecules. Chemists take compounds like this one and tweak the structure to search for the next big breakthrough in medicine. The trifluoromethyl group attached changes the way the molecule interacts inside the body, sometimes making a drug more effective or less likely to break down quickly.
Fluorine’s reputation in chemistry stands out for a reason. Adding those three fluorines, as seen here, can turn a basic molecule into something very different. In medicines, that shift changes how a drug travels through the body or how it latches onto its target. Research has shown that pharmaceuticals with trifluoromethyl groups tend to have better stability and can sometimes avoid being chewed up right away by the liver. By slipping 2-Methyl-3-(Trifluoromethyl) Aniline into new molecular recipes, scientists explore new ground in treating disease.
Beyond Pharmaceuticals
Some people only think of chemistry in terms of pills and tablets, but this compound stretches out beyond medicine. In agrochemicals, that same tricky group of atoms gives pesticides or herbicides a longer life in the environment—not necessarily good news to everyone, but sometimes necessary for protecting crops. Researchers also look at this compound for dyes and specialty polymers. Even electronics and advanced materials draw from the unique traits that the trifluoromethyl group can give.
This raises real questions about safety and environmental impact. The same traits that let a pesticide stick around in the soil or water may bring headaches for conservationists or people living near farms. The industry keeps an eye on persistence and bioaccumulation—how long a compound sits in the environment and whether it climbs up the food chain. The conversation here should not be just about clever chemistry but about responsible use and disposal. Strong oversight and smarter engineering at the early design stage can make all the difference.
Learning from Experience and Looking Forward
As someone who spent weekends in dusty old university labs, I’ve seen students and seasoned researchers wrestle with molecules like this. Sometimes, one small tweak unlocks a whole new set of possibilities. Other times, it brings up new risks, or solves one problem only to create another. The story of 2-Methyl-3-(Trifluoromethyl) Aniline isn’t just about new discoveries. It's also about careful thought on how chemicals shape the world around us—and making sure science serves people, without leaving a mess behind. Good science pays attention to both the promise and the pitfalls.
Getting to the Root of Chemical Structure
Chemists, students, and even folks in industries outside chemistry stumble across names like 2-Methyl-3-(Trifluoromethyl) aniline, then pause. Nomenclature in organic chemistry stacks up terms and numbers in a way that gives precise hints about molecular structure. Reading the name breaks down not just the arrangement of atoms but often the purpose and quirks of the compound. Familiarity with chemical structures is almost like reading a street address—each element tells you something about where to find what you're after.
The Formula Fingerprint
2-Methyl-3-(Trifluoromethyl) aniline comes from a family of compounds known as substituted anilines. Aniline itself means a benzene ring sprouting an amino group (–NH2). The rest of the name adds side streets off the main avenue—methyl at the 2-position, trifluoromethyl at the 3-position. The chemical formula sums up all the atoms in the molecule: C8H8F3N. Look at it closely—eight carbons, eight hydrogens, three fluorines, and a nitrogen. It’s not magic; it’s the result of consistently applying IUPAC naming standards that let anyone reconstruct the skeleton just by reading the name.
Why Formula Literacy Matters
Chemical formulas turn abstract chemistry into something you can grab with your hands—at least in your imagination. A simple glance reveals the presence of a trifluoromethyl group, a flag for chemists because it brings in a strong electron-withdrawing dynamic. These kinds of groups often show up in drug development, materials science, and even in the search for new pesticides. Trifluoromethyl groups resist metabolic breakdown and push molecules to behave in uncommon ways, sometimes boosting a medicine’s performance or helping an industrial material stand up to harsh environments. The methyl group, meanwhile, packs a five-atom punch that can modify physical and biological properties.
From Structure to Impact
Stepping past the formula, it’s clear that small changes in structure have ripple effects out in the real world. In medicinal chemistry, switching out a hydrogen for a trifluoromethyl group or sliding a methyl to the ortho position on a ring tweaks activity. Drug researchers wrestle with these tiny changes to hunt down new treatments for everything from infections to chronic diseases. Fluorinated aromatics also feature in agrochemicals, dyes, and more. You don’t need to be in a chemical lab to appreciate why understanding the structure and formula matters. The choices chemists make on paper can shift economies, affect health, and even shape future technologies.
Solutions for Better Understanding
Many high school and undergraduate classrooms barely scratch the surface when it comes to training students to decode chemical formulas and names. Yet, this skill opens doors. Adding more practice with nomenclature, using modeling kits to build molecules by hand, and teaching with examples that show real-world consequences underscores why these abstract-sounding names are far from meaningless. For professionals, continuing education and exposure to case studies remind us how formulas shape the backbone of innovation. No one needs to memorize every formula, but learning how to break them down pays off every time a chemical challenge crops up.
Understanding the Real Risks
Chemicals like 2-Methyl-3-(Trifluoromethyl) Aniline rarely appear in mainstream headlines, but plenty of us cross paths with them if we work in labs, manufacturing, or even art restoration. The concern around toxicity always lingers where chemicals play a role, and I've seen how ignoring the specifics invites real trouble.
This compound slips quietly into many industrial processes—pharmaceutical research and agrochemical production come to mind. Most people, even in technical circles, don’t walk into the details unless they must. But the trifluoromethyl group alone gets my attention. Fluorinated compounds often stick around in the environment and resist breaking down; that persistence means any toxicity doesn’t just fade out of the picture.
What We Know About Its Toxicity
Finding clear-cut public data about 2-Methyl-3-(Trifluoromethyl) Aniline toxicity proves tricky. Official lists, like those from the Globally Harmonized System (GHS) or the European Chemicals Agency, sometimes classify these related anilines as “harmful if swallowed” or “harmful in contact with skin.” Breathing in dust or vapors could irritate the respiratory tract. Many aniline derivatives show clear risks of damage to blood and nervous systems.
Anecdotally, older colleagues tell stories about minor skin exposure leading to blue-tinged fingernails—methemoglobinemia, where the blood’s ability to carry oxygen drops. That’s not a risk I shrug off. Ingesting or inhaling even small amounts of aromatic amines over long periods causes problems in lab animals, too.
Let’s not forget—the trifluoromethyl group boosts the lipophilicity, which often means the chemical sneaks into biological systems more easily. Some research on related chemicals suggests organ toxicity with enough exposure. It’s not wise to treat any of these solvents or intermediates as benign, no matter how routine they seem in a synthetic route.
Why It Matters on the Ground
Skimming over safety with chemicals feels tempting for the sake of speed, but over my years in academic and industry labs, I’ve seen people pay for shortcuts—rashes, headaches, weeks off due to inhalation injuries. In sectors where oversight gets less attention or budgets run thin, the real-world impact grows.
On another front, environmental regulations now draw more attention to fluorinated emissions. PFAS headlines underscore the challenge: once released, these molecules drift into groundwater, refuse to degrade, and turn up far downstream from where they started. 2-Methyl-3-(Trifluoromethyl) Aniline fits into that same web of chemicals that demand careful handling both in the lab and throughout disposal.
Paths Toward Safer Use
Safer work with chemicals like this demands more than eyewash stations and gloves tossed on at the door. I’ve learned that proper training—showing people firsthand what spills mean or walking through close calls—has a longer-lasting effect than a stack of Material Safety Data Sheets nobody reads. Facilities tracking their chemical use, updating protocols, and welcoming feedback from the bench level build healthier habits. Engineers who design with closed systems, efficient scrubbers, and good waste management create workplaces where risk shrinks.
There’s real value in substituting less dangerous substances when the chemistry allows; green chemistry isn’t just about buzzwords. Where substitution won’t work, transparency about the real hazards helps everyone see why vigilance matters. Regulators, managers, and frontline researchers share that responsibility. The right culture causes the risks to drop, not just on paper, but out in the real world.
Why Storage Isn’t Just a Box-Ticking Task
Too often, chemical storage looks like it’s only about following a list of rules—keep it cool, keep it dry, keep it sealed. Anyone working in a lab or at a small industrial site knows storing 2-Methyl-3-(Trifluoromethyl) aniline safely means a whole lot more than that. You’ve got a compound with a trifluoromethyl group, which calls for particular care. Ignore the risk of moisture and you invite slow degradation, contamination, and possibly even safety issues. Ignore the right type of container and you face leaching, spills, or dangerous odors. I’ve seen more than one workspace learn this lesson the hard way.
What Proper Storage Looks Like
I’ve stored compounds like this in university research labs and small scale pharma settings. The same things make sense wherever you’re working. This chemical goes in a tightly sealed amber glass vial, not plastic—those reactive amines can chew through or stain. Sunlight breaks down organics faster than most realize, so a dark cabinet or a dedicated refrigerator is a must, away from windows and heat sources. Temperatures should stay on the cool side, below room temp if possible, though you don’t want it so cold it crystallizes on you. Moisture control isn’t just preference, it keeps hydrolysis in check, so I keep desiccants in the cabinet.
No matter how harmless it might look, this isn’t a compound to toss in with open bags of sodium sulfate or beakers left out for air-drying. Volatile organics, especially those with fluorine, pose inhalation hazards and strong odors. A dedicated chemical hood gives ventilation if you’re working regularly with the stuff, but for pure storage, double-layer containment is the way to go—use a secondary container to catch leaks.
What Happens If You Don’t Take Care?
A chemical like this, stored poorly, leads to real headaches. Traces of water or oxygen trigger surface discoloration and build-up of impurities. One missed leak leads to sticky shelves and a smell nobody wants to live with. The bigger issue comes during disposal or handling—one splash, due to a brittle aging bottle or cracked cap, sets off skin or eye irritation. I learned early to keep a spill kit on hand, because some labs let bottles sit for years, quietly degrading.
OSHA notes that aromatic amines can sometimes turn up with unexpected hazardous byproducts due to slow side reactions with light and air over time, so letting your stock oxidize or evaporate is not just wasteful, it invites trouble. No researcher likes tossing half their chemicals because humidity or excess heat turned them unreliable.
Getting It Right: Solutions for Everyday Labs
Better storage relies on good routines and clear labeling. I date each bottle and record when it gets opened. If humidity spikes, silica gel packs go in the cabinet for backup. Regularly checking for sticky residue, unusual smells, or any signs of bottle damage prevents a disaster before it starts. Training every technician or student in your group on this process costs almost nothing but saves plenty in the long run.
If budgets are tight, focus on basics: glass containers, dark storage, and secondary containment. Don’t mix incompatible chemicals in one spot—a dedicated bin avoids cross-contamination. For higher volumes or critical quality, investing in a temperature-controlled cabinet pays back in longer shelf life and fewer headaches. Documentation, though tedious, keeps everyone on the same page and helps spot trouble early.
Taking these steps always beats the cost of wasted batches or the scramble that follows a minor spill gone wrong. Respecting the quirks of each compound isn’t just good lab practice—it keeps colleagues safe and projects moving.
Understanding What’s At Stake
Many chemicals out there raise some eyebrows, but 2-Methyl-3-(Trifluoromethyl) Aniline isn’t the sort of thing anyone picks up at the corner store. In my own experience visiting labs and talking with chemists, sourcing a substance like this brings up more questions than answers. Most regular people probably haven’t even heard of it, and for good reason—this compound finds its home in pharmaceutical research, agricultural chemistry, and maybe the odd piece of advanced materials science.
Walking into a chemistry supply catalog, you won’t catch this on the shelf next to cleaning solvents or gardening fertilizers. Businesses that handle specialty chemicals check buyers from top to bottom. Suppliers ask for professional credentials and paperwork, and some go as far as demanding institutional backing. There’s tight control here—not to annoy the buyer, but for clear, safety-driven reasons. Synthetic intermediates like this one deserve respect, both for their handling risks and for their potential misuse.
Finding A Reliable Source Means More Than Searching Online
People comb the web looking for distribution channels, and it’s tempting to reach out to the first result with an order form. Experience tells me: real, reputable sellers do not work in the shadows. Providers with a good track record show proof of supply chain transparency, and their licenses check out with chemical regulatory agencies. In some countries, I’ve seen how tight restrictions block shipping altogether, especially for buyers without formal research affiliations.
Legitimate chemical suppliers prefer dealing with businesses, universities, or government-backed labs. They keep compliance paperwork close at hand and walk buyers through questions about intended use and storage. A casual shopper trying to place an order—no matter how well-meaning—rarely gets past the first screening.
The Role of Regulation in Shaping Access
Across the United States, Europe, and much of Asia, chemical registration laws keep tabs on both creators and buyers. As I dug through regulatory frameworks, it struck me that the point isn’t just tracing who buys what. The industry wants to limit accidents and illegal activities, keeping toxic exposures and environmental risks in check. Audits come regularly, and fines for sidestepping these rules can ruin a business’s reputation overnight.
On one hand, tight regulation slows rogue actors looking for shortcuts. On the other, it creates headaches for genuine researchers. I’ve heard from colleagues who needed months to process a single chemical order just because a line item looked unfamiliar to customs. In this environment, patience and preparation mean everything.
Supporting Safer Access Without Losing Security
Researchers often talk about easier, safer, and faster routes to the chemicals they need. But shaping a perfect system remains tricky. Many suggest updates to the way governments and suppliers verify credentials. Adding online credential databases or blockchain-backed records could make the vetting process smoother, but these efforts take time to roll out. Extra training for staff who verify buyers could help too, cutting back on confusion or mistakes.
From my seat, the balance between progress and safety keeps the chemical industry on its toes. Transparency, up-to-date regulations, and responsible supply chains help everybody—producers, buyers, and the wider public—avoid trouble without grinding valuable research to a halt. Getting 2-Methyl-3-(Trifluoromethyl) Aniline may seem like a simple search, but in reality, only trust and thoroughness keep the process on the right track.