Reflecting on 2-(Perfluorobutyl)Ethyl Acrylate: Stories from Lab and Industry
Historical Development
No one working with specialty acrylates these days can ignore the silent shift that fluorine chemistry has brought to surface coatings and advanced materials. 2-(Perfluorobutyl)ethyl acrylate, often simply referenced as PFBA acrylate among researchers, didn’t appear out of thin air. Decades ago, chemists dreamt about new monomers that could fight both water and oil, and their work grew out of the larger story of fluorinated organics. As a young chemist in a university lab some years back, I saw that earlier attempts with longer perfluoroalkyl chains raised valid toxicity and bioaccumulation fears. The push toward shorter chains like perfluorobutyl was not about riding trends but about taking responsibility—shrinking environmental footprints while keeping the same powerful surface traits. The shift away from larger PFAS molecules is no small part of this history, and it signaled a better direction for performance and environmental health together.
Product Overview
Looking at a bottle labeled with 2-(Perfluorobutyl)ethyl acrylate, a seasoned polymer chemist immediately recognizes both promise and caution. This monomer wears many hats—it anchors itself in the world of high-performance coatings, specialty adhesives, and demanding textiles. The simple act of co-polymerizing it with other acrylates can lead to surfaces that laugh off stains and shrug at both water-based and oily challenges. These qualities come from the marriage of a robust acrylate group with a perfluorinated tail, creating properties that stay rare among other traditional monomers. It rarely works alone; most often, it joins larger molecular teams to deliver those non-stick, stain-resistant, low surface energy traits that mature industrial designers crave.
Physical & Chemical Properties
From my days putting vials into NMR and running IR spectra, I learned that PFBA acrylate impresses with its clear, nearly colorless appearance and mild odor, which belies its robust chemical backbone. The molecule boasts strong carbon-fluorine bonds—some of the toughest in organic chemistry. This tight architecture brings a high level of resistance to polar and non-polar solvents, and it resists hydrolysis and photodegradation where lesser monomers wilt. Its relatively low surface energy, attributed to the fluorinated tail, allows it to repel liquids with ease. In actual coatings, this turns into visible beading of oil and water—a small visible miracle that comes straight from chemical symphony. Handling in the lab reveals a moderate viscosity, which can ease formulation, and its acrylate end holds all the reactive charm polymer scientists lean on for cross-linking.
Technical Specifications & Labeling
Before any material reaches the bench or pilot plant, compliance officers and chemists will look for clear labeling—chemical names, CAS numbers, storage instructions (often in dark, cool places to slow down unwanted polymerization), and hazard information. As I saw firsthand, issues like acrylate skin sensitization aren’t theoretical; a single careless splash creates irritation. Many producers ship PFBA acrylate with a stabilizer, often a trace of inhibitor like MEHQ, to keep the product fresh through shipping and storage. Technically, the compound echoes other liquid monomers—keep away from fire, avoid direct inhalation, and wear proper gloves and goggles. In my own experience, it’s the gloves and fume hood that keep minor incidents from turning into official case studies.
Preparation Method
Syntheses of PFBA acrylate takes careful balance and some specialty equipment. The chemistry starts by building the 2-(perfluorobutyl)ethyl alcohol via telomerization or alkylation of perfluorobutyl intermediates, and then the alcohol meets acryloyl chloride in the presence of base to produce the ester. In the lab, this means controlling reaction temperatures and drying glassware obsessively, as water sneaks in and spoils yields. One ex-colleague described the acrid scent that clouds the fume hood when acryloyl chloride gets out of control—nobody forgets it. This method may sound routine to anyone steeped in ester chemistry, but the cost and scarcity of fluorinated precursors always heighten the stakes. Every lost milliliter stings the research budget. Waste management also weighs on the mind since both organic and fluorinated leftovers demand special care.
Chemical Reactions & Modifications
PFBA acrylate’s main claim to fame lands in free-radical polymerization, joining up with methacrylates, styrenics, or other acrylates to form tough, slick copolymers. For professionals steering reaction pathways, chain transfer agents guide molecular weights while photoinitiators allow UV-curing at industrial scales. The fluorinated arm introduces a unique set of properties that’s tough to replicate by any simple hydrocarbon route. Post-polymerization, chemical modification isn’t common—once the fluorinated piece locks in, its stubborn stability makes functional tweaks difficult. Some research groups keep exploring clever ways to bond PFBA acrylate segments into block copolymers, or to “graft-on” further functionality for medical or sensing applications, but most industry applications stick to conventional copolymerization.
Synonyms & Product Names
Older chemical catalogs and patent documents sometimes use the tag “2-(1,1,2,2-Tetrafluorobutyl)ethyl acrylate” or even “perfluorobutylethyl acrylate.” I have seen a range of trademarks and brand names assigned by large fluorochemical producers, mostly alphabet soup codes that vary by region and catalog. Reviewing any historical work or regulatory filing, an experienced hand checks at least one synonym to avoid missing relevant safety or restriction data—there’s no global agreement on product tagging, and language can obscure science in frustrating ways.
Safety & Operational Standards
Safety culture grows from past accidents and lessons. I remember early days scrubbing beakers and noticing old glove stains that lingered from acrylate hands. 2-(Perfluorobutyl)ethyl acrylate, like its cousins, demands respect: skin contact leads to irritation or sometimes serious allergic reactions. Inhalation of vapor risks more than a headache—it sets up symptoms from mild coughing to severe distress, depending on exposure. Regulatory guidelines call for proper ventilation, full PPE, and regular safety audits. Any spill, even on the floor of an advanced lab, gets prompt cleanup with specialized fluorinated absorbents. As with many fluorinated chemicals, open flames and high heat increase risk, and standard practice keeps sources of ignition far from working areas. Disposal rules lean on licensed chemical waste handlers capable of tracking both organic and fluorinated refuse—lurking beneath the advanced performance lies a permanent environmental cost if handled badly.
Application Area
Walk through any modern manufacturing space that produces advanced coatings, and chances are you’ll see products built on monomer blends including PFBA acrylate. Its real strength lies in boosting the weather- and stain-resistance of coatings for aerospace, automotive, and high-performance textiles. The textile sector, in particular, relies on this chemistry to deliver “easy-clean” surfaces without constant recleaning or aggressive detergents. Medical device makers look to the biocompatibility and anti-fouling promise in limited diagnostic and sensor innovations. Not every application welcomes fluorinated materials—cost and sustainability concerns block its reach into mass low-end goods. Where properties matter more than price, though, you find a community ready to troubleshoot the quirks that fluorine always brings, including low adhesion, reactivity to metals, and complex end-of-life disposal.
Research & Development
Research on PFBA acrylate pushes forward in both incremental and bold steps. My friends in surface science see regular projects that blend PFBA acrylate into new nano-coatings or test how it stands up under salt spray and ultraviolet exposure. Universities dig into blends for membranes, sensors, and even next-gen filtration—not just slick surfaces but active interfaces. R&D teams chase ways to recycle or degrade fluorinated polymers without releasing persistent compounds. Researchers in regulatory science, on the other hand, keep drilling down with better analytical tools like high-resolution LC-MS to monitor breakdown products and trace migration into air, water, or biologic samples. Open literature grows richer with every study that analyzes environmental impact or seeks alternatives, but no one calls this problem solved.
Toxicity Research
Toxicologists and environmental chemists grew vocal after the lessons of longer-chained PFAS compounds, and scrutiny remains high. While PFBA (the acid form) draws less attention for persistence than its lengthier relatives, debates over bioaccumulation and long-term health effects continue. Limited in vivo and in vitro studies suggest the shorter chain brings lower risk, but the data isn’t unanimous. In lab handling, acrylates as a class have shown evidence of skin sensitization, allergic contact dermatitis, and in rare cases, respiratory symptoms. Chronic exposure studies still lack the full, decades-long picture. I’ve attended enough workshops to hear researchers stress regular monitoring for lab workers and waste discharge tracking to prevent accidental long-term poisonings—nobody wants to repeat past mistakes.
Future Prospects
Acrylic monomers with fluorinated side chains, including PFBA acrylate, face a strange crossroads. Material scientists still demand the non-stick, chemical-resistant traits for satellites, wind turbines, and premium textiles. Regulatory pressure on all fluorinated organics rises year by year. Sustainable chemistry circles search for ways to recover or depolymerize fluorinated plastics, and some work focuses on finding hydrocarbon alternatives that approach PFBA’s unique performance. Most likely, restricted and responsible use will define the field for the next decade, with recycling methods and process control standing at the center of new investment and science. I expect to see shrinking use patterns in consumer spaces and sharper focus on critical needs, where performance pays its environmental toll in longer-lasting, repairable goods. Ultimately, the industry owes transparency on life cycle and health impacts, and this story, from bench chemist to end-user, will keep growing in complexity as both science and values evolve.
Unpacking the Power of a Specialized Acrylate
I’ve always been interested in the quiet chemistry shaping our surroundings. Take 2-(Perfluorobutyl)ethyl acrylate as one small example: most don’t know its name, but its impact stretches from high-rise windows to your smartphone’s sleek exterior. This chemical belongs to a family of acrylates armed with an added perfluorinated tail, and that tweak changes everything. Its backbone lets it polymerize into tough films, while the fluorinated chain shrugs off oil, water, dirt, and even harsh chemicals.
What Makes It Stand Out?
From experience in materials science, hydrophobic materials never get old. Yet, most coatings break down or become sticky over time. 2-(Perfluorobutyl)ethyl acrylate delivers nonstick without losing durability. Whether walking into a modern office or using outdoor gear during a rainstorm, odds are you’re in contact with a thin film of this kind of polymer. This stuff gives glass, metal, and plastic the ability to resist fogging, corrosion, and fingerprints, helping products look new even after years of hard use.
Main Fields That Rely On It
Building coatings soak up huge quantities of this acrylate. Architects and developers want windows that block stains and repel dirt, cuts down on cleaning and keeps city skylines bright. Windshields and solar panels benefit as well. In electronics, companies prize surfaces that repel oil and sweat, protecting displays and touch panels. I’ve seen labs blend the compound into polymers that survive repeated contact with solvents or corrosives—common in both laboratory equipment and medical devices.
Industrial manufacturing is hungry for surfaces that don’t foul, clog, or break down. Factory machinery with coatings using this acrylate doesn’t gum up or corrode as quickly. In the textile world, raincoats and technical apparel stay dry and light because manufacturers treat fibers with these high-performance fluorinated monomers. There’s demand wherever water, oil, or chemicals threaten to compromise performance—from nonstick cookware to automotive finishes.
Health, Safety, and Environmental Questions
Recent years brought tough questions around fluorinated chemicals. Regulations are mounting, especially in Europe and North America, as evidence piles up on how persistent some of these compounds can be. While 2-(Perfluorobutyl)ethyl acrylate brings real value, there’s pressure to manage waste carefully, limit emissions, and invest in safer production methods. Wastewater treatment plants and recycling programs face new technical hurdles dealing with stable fluorinated residues. I’ve seen some innovators in the field try to develop less persistent versions, but substitutes often lack the unique mix of durability and repellency.
Looking Toward Better Solutions
It’s not possible to walk away from high-performance surface coatings altogether. Demand isn’t shrinking as society wants longer-lasting, cleaner, easier-care materials. Finding a balance means digging deeper into green chemistry—sourcing better raw materials, building more efficient processes, and fine-tuning product life cycles to reclaim and recycle fluorinated polymers. Research labs keep experimenting, searching for safer alternatives with similar benefits but without the environmental baggage. Until then, every player in the supply chain—chemists, engineers, regulators—owns a piece of the push toward safer surfaces and smarter manufacturing.
Looking at the Molecule: Why Structure Matters
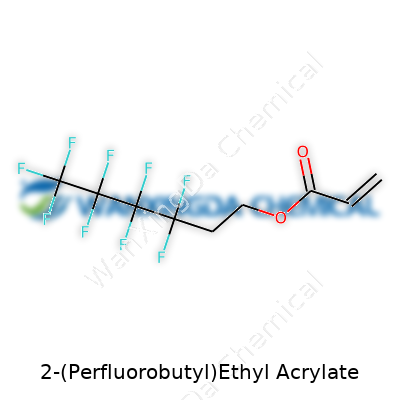
Chemistry isn’t just gritty textbooks and funny-smelling labs. Each molecule tells a story. 2-(Perfluorobutyl)Ethyl Acrylate sits squarely in that camp. This compound layers up its carbon and fluorine atoms in a way that shows how science keeps reinventing building blocks. Its chemical structure falls into the family of acrylates, but it’s dressed up with a perfluorobutyl tail—essentially a four-carbon chain where every hydrogen gets swapped for a fluorine. Imagine drawing this out: you have the acrylate backbone, an ethyl linker, and then a perfluorobutyl arm—CF3CF2CF2CF2CH2CH2O2CCH=CH2. No magic, just carbon, hydrogen, oxygen, and a heap of fluorine atoms locked together.
The molecular weight stands at about 338 grams per mole. That number jumps out when you think how each fluorine is about nineteen units—fluorine piles on serious heft. You won’t get this from a plain ethyl acrylate. Swap in those fluorine atoms, watch the weight go up, and see new possibilities open in materials science.
Why Add All That Fluorine?
There’s a reason chemistry keeps returning to fluorinated chains. Fluorine fends off water, resists solvents, and shrugs off heat and chemical attack. Wrap that into an acrylate and new coatings, adhesives, even specialty plastics show traits that fit stubborn environments. As a writer who’s had more than my share of sticky label failures in humid summers, it’s clear the quest for better materials isn’t just high tech—it touches the everyday.
Years ago, waterproof jackets and nonstick pans felt like science fiction. Today, these items rely on chemistry like 2-(Perfluorobutyl)Ethyl Acrylate to keep moisture and muck from sticking around. The magic comes straight out of the molecular structure—everything engineered to dodge trouble.
Challenges with Fluorinated Compounds
Not all stories go one way. Fluorinated materials build incredible products, but some of these same chains linger in the environment. While 2-(Perfluorobutyl)Ethyl Acrylate itself isn't a headline-grabber for pollution, the industry is feeling the heat to balance innovation with health and environmental care. Studies have found long-chain perfluorinated compounds taking a long walk through soil and water, showing up in places far from their original use. People want waterproof coatings but dislike chemicals that stay long after the rain dries up.
The answer probably won’t come fast. Shifting to shorter-chain alternatives, improving waste handling, and creating more efficient manufacturing processes all offer hope. Tackling the problem means listening to researchers who track what these molecules do in the wild and what it takes to build breakable, less-persistent versions without throwing out performance.
Finding the Balance: Purpose and Responsibility
Chemistry shapes how we live, from protecting electronics in tropical storms to sealing sinks at home. Getting familiar with the structure and weight of something like 2-(Perfluorobutyl)Ethyl Acrylate reminds us that progress blends together small details. Materials built for endurance and specialty demands are only one side of the coin—the other side carries a responsibility. Science doesn’t stand still. Neither do the ethics that guide what gets made, sold, and even replaced as we learn more.
Practical Chemical Safety Starts in the Storage Room
In any lab or production setting, specialty chemicals like 2-(Perfluorobutyl)Ethyl Acrylate call for a careful touch and a respect for the risks. This substance finds its way into modern coatings, electronics, and specialty polymers. It stands out for its slick, hydrophobic nature. But with that fluorinated edge comes a set of storage and handling needs that can’t be brushed aside. My years in research have shown me that little lapses—a cracked flask, a forgotten lid, sloppy labeling—snowball quickly, especially with acrylates that aren’t your everyday solvents.
Why Clean Storage Conditions Matter
Fluorinated acrylates stick around in the environment and inside hardware, so the wrong move brings headaches for months. Storing this compound in sealed, chemically resistant containers isn’t just about shelf life. Vapors from acrylates seep into cardboard boxes and plastic bins, leaving residue and odors that travel to shared workspaces. I once watched a shared fridge turn into a mini toxic waste zone because someone let a small leak go unchecked.
Glass and high-grade fluoropolymer containers keep chemicals pure and air-tight. Even a few drops of organic solvent or stray water in the container can set off unexpected polymerization or destroy the chemical’s value. No one forgets the first time a drum liner dries solid with the last bit of expensive acrylate glued to it.
Don’t Overlook Temperature and Light
Acrylates react to light and heat, both in storage and during handling. Cool, dark storage rooms slow down unwanted reactions. Leaving bottles in sunlight or near a steam line isn’t just a minor mistake—it’s asking for degradation, loss of material, and sometimes a nasty pressure build-up. During hot months, temperature spikes sneak up on you, accelerating breakdown and slamming the shelf life.
Get Personal Protective Equipment Right
Too many folks grab thin gloves or skip eye protection because “this isn’t sulfuric acid.” Fluorinated acrylates cut straight through most kitchen-grade gloves. Only thick nitrile or specialized laminate gloves stand up. Inhalation of vapors brings lung irritation, especially in small or poorly ventilated labs. Decent goggles and a good lab coat add a simple but necessary layer, and proper hoods make a difference when pouring, sampling, or transferring these liquids.
Clear Labels and Training for Real Life
No fancy system or regulatory binder replaces clear, honest labeling. Date every new bottle, mark decanted containers, and write legibly. In big teams, I’ve seen how a misplaced decimal point or an old sticky label turns confusion into real risk. Weekly safety walkarounds spot leaks, check expiration dates, and nudge people into better habits. Having one chemist in charge of enforcement doesn’t work; habits only stick when everyone—new hires and veterans—buys in and expects the same discipline from each other.
Room for Improvement: Building a Good Culture
Safer storage and handling of 2-(Perfluorobutyl)Ethyl Acrylate build off a foundation of good habits and honest communication. Facilities should invest in better ventilation, training, and storage hardware—not just check boxes for audits. Regular real-life drills for spills and exposures, rather than paper-only exercises, prep people to act when it counts. And easy access to reliable chemical information, not buried behind digital paywalls, makes all the difference on a busy day.
Getting Real About Chemical Safety
Many of us don’t pay attention to long chemical names like 2-(Perfluorobutyl)Ethyl Acrylate until they pop up in the news or in research tied to water quality, food packaging, or air around workplaces. Fluorinated chemicals, such as this one, have built a reputation. The wider family—also called PFAS—has shown persistence in the environment and the bodies of animals, from otters in river deltas to people living near factories.
What the Science Tells Us
Peer-reviewed studies link similar PFAS to health problems. Some longer-chain versions have shown up in drinking water, soil, and even blood samples. While data on 2-(Perfluorobutyl)Ethyl Acrylate itself stays narrower, its structure suggests it may act just as stubborn as its chemical cousins. That means it won’t break down easily, hanging around in water streams and then climbing up the food chain.
Impact on Humans and How the Risks Appear
People working directly with this chemical face the highest exposure. Workers in manufacturing or those handling liquid coatings feel the effects first if safety rules slip. Research on exposure to similar chemicals points to liver and kidney toxicity, reproductive damage, and likely impacts on the immune system. These effects usually build up after ongoing contact, and the body can store PFAS much longer than many other industrial chemicals.
Neighbors near plants using these compounds have added reason to care. PFAS, including 2-(Perfluorobutyl)Ethyl Acrylate, have a knack for leaching through soil, showing up in wells and even municipal drinking water supplies. Some communities near PFAS production sites now run expensive filtration systems, and families worry about the links between PFAS and cancer or thyroid disease.
Where the Environment Gets the Short End
I’ve walked streams near old chemical plants and seen the warning signs telling anglers not to eat the fish. This is not just a scare tactic. Fish collect PFAS in their fat, and local wildlife struggle with hormones, growth problems, and lower chances to reproduce. In Europe, government agencies measured PFAS, including chemicals close to 2-(Perfluorobutyl)Ethyl Acrylate, in river water far from city centers. Once released, these chemicals stick around, often outlasting current pollution laws.
You find the same pattern in soil and air. In farming regions, biosolids taken from wastewater sometimes land on fields as fertilizer, bringing persistent chemicals with them. Rain doesn’t carry these away; instead, they find their way into crops and grazing cattle.
Paths Forward: Reducing Harm and Making Smarter Choices
Some countries already restrict whole classes of PFAS. The United States Environmental Protection Agency and the European Chemicals Agency run research and encourage industries to find safer alternatives. Public awareness, grassroots monitoring, and better standards make a difference. It takes industry transparency to help workers, and strong support for scientists and local governments to say no when safer choices exist.
Personal experience shows local groups can spearhead water testing and push for action. At the policy level, the clearest step involves phasing out persistent PFAS where possible, requiring disclosure about chemical use, and investing in cleanup tech. Until 2-(Perfluorobutyl)Ethyl Acrylate and its relatives come under tighter watch, their story serves as a reminder—what goes down the drain may come back years later, rarely kinder than before.
Looking Past the Label — Why Packaging and Purity Matter
Chemistry labs never overlook the details, and with a specialty monomer like 2-(Perfluorobutyl)Ethyl Acrylate, small decisions shape big outcomes. Packaging size and purity are two critical factors that people sometimes ignore until halfway through a synthesis, suddenly realizing that a minor impurity or too much leftover solvent can throw everything off. Years working alongside researchers and polymer chemists made it clear: the real frustration comes when a project hits a wall over what seemed like a simple packaging decision.
What Packaging Sizes Actually Hit the Shelves?
Most chemical suppliers offer 2-(Perfluorobutyl)Ethyl Acrylate in a range of sizes, guided by demand from both academic and industrial labs. The small end usually features 5-gram or 10-gram glass vials. These are handy for labs doing material screening or pilot projects with limited runs. Those looking at production or scale-up typically step up to 100-gram bottles or one-liter containers, shipped in amber glass to protect from light. Bulk needs? Drum packaging above five kilograms appears in the catalogues, but ordering this much often means negotiating special handling, given the chemicals’ sensitivity and niche market role.
There’s no one-size-fits-all here. Once, a small team devised a new fluoropolymer for electronics coatings but ran out halfway through their trial. Scrambling for a second shipment, they noticed the cost of small vials far exceeded the price per gram of larger bottles. Yet, large containers are not always practical, especially if reaction conditions depend on maintaining the chemical’s freshness. Unopened packaging saves the monomer from contamination and unwanted polymerization—an issue that every researcher running acrylic reactions knows all too well.
The Real Stakes Behind Purity Grades
Purity isn’t a marketing word for chemists, especially with fluorinated acrylates. Even a half-percent impurity can introduce noise into surface coatings, disrupt microelectronic fabrication, or tank a polymer’s UV resistance. High-purity grades—97% and up—are standard for synthetic chemistry and advanced material development. Researchers use purification methods in-house at times, but this takes extra resources, forcing trade-offs in time and budget.
Suppliers might offer “technical grade” for less demanding work, but material for electronics, healthcare devices, or automotive coatings comes in higher-purity versions—sometimes flagged as “assay ≥98%,” or even “HPLC grade.” In my experience, the more ambitious the application, the more people want to see independent analytical data, such as NMR, GC, or MS traces, before placing any real order. This isn’t fussiness; it’s chasing consistency and performance, especially for high-value parts or safety-critical coatings.
Supply Chain and Sourcing Strategies
Sourcing specialty chemicals takes more than picking the right bottle. Labs hunting for 2-(Perfluorobutyl)Ethyl Acrylate routinely ask about trace impurities, stabilizers, and inhibitors that might be present to prevent premature polymerization. Some find that smaller suppliers can custom-purify in response to particular requests, though this can increase lead times and costs.
Mismatches between project timelines and supplier stock can trigger months-long delays. Collaborative purchasing, shared storage solutions, and pre-negotiated purity requirements help buffer these risks. Chemical distributors with established cold chain or light protection practices give added peace of mind, especially for sensitive fluorinated monomers that degrade out of sight on a warm shelf.
Moving Toward Clearer Labels and Better Access
Transparent labeling of purity and packaging sizes helps researchers avoid wasted material and unpredictable results. The specialty chemical sector has seen slow improvements in making analytical data and supply options visible online. As demand for advanced coatings and electronics applications grows, the call for standardized, well-documented packaging and purity grades won’t let up anytime soon.