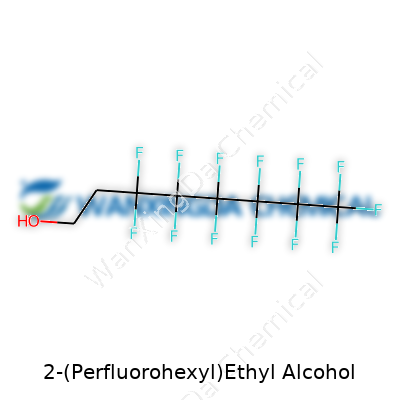
2-(Perfluorohexyl)Ethyl Alcohol: Chemistry, Safety, and What’s Next?
Historical Development
Perfluorinated alcohols tell the broader story of humanity’s ongoing journey with fluorinated organics, and 2-(Perfluorohexyl)ethyl alcohol stands out thanks to its unique blend of chemistry and industry influence. Back in the nascent days of fluorine chemistry in the twentieth century, most people only gave thought to Teflon pans or fluorine’s blistering reactivity. Chemists slowly built the expertise required to handle longer perfluoroalkyl chains, dodging the health and environmental problems that started appearing from indiscriminate use of PFAS compounds in everything from waterproof jackets to firefighting foam. As researchers faced more pressure to find sustainable and safe fluorinated materials, structural tweaks like installing a short (six-carbon) perfluoroalkyl tail and an ethyl alcohol head proved pivotal. Over the decades, the focus around these materials shifted from pure performance to the entire lifecycle—what happens to these compounds in soil, water, and living things didn’t go unnoticed. The modern research landscape no longer treats perfluorochemicals as miracle additives, but rather as promising chemistry requiring full transparency and responsibility from bench to field.
Product Overview
2-(Perfluorohexyl)ethyl alcohol hits a crossroads between tradition and innovation. While it shares a backbone with the notorious “forever chemicals,” some regulatory agencies and scientists examine it for both its benefits and its baggage. Its structure—a perfluorohexyl chain tethered to an ethanol group—creates a highly hydrophobic tail with a tiny, reactive hydrophilic handle. If you’ve ever dealt with stains that refuse to budge or tried to keep electronics dry, you’re already living in a world where products like this emerge as key players. Coatings, surfactants, and high-performance finishes rely on that perfect blend of oil- and water-repelling power. Unlike the older, longer perfluoroalkyl analogs (with eight or more fluorines), the hexyl versions draw less scrutiny when it comes to persistence and toxicity, but the conversation hasn’t ended—researchers dig deeper to balance function with safety.
Physical & Chemical Properties
Pick up a vial of 2-(Perfluorohexyl)ethyl alcohol and you’ll notice the clear, oily liquid with surprising slipperiness between the fingers—just keep in mind, gloves matter. The perfluoroalkyl tail makes it less prone to evaporation and surprisingly nonreactive under normal temperatures and pressures, but the alcohol group on its head gives chemists that little handle for more creative modifications. In water, the molecule stubbornly refuses to dissolve, favoring organic solvents like ethers or chlorinated hydrocarbons instead. This unusual combination—a fiercely hydrophobic chain paired with an ethyl alcohol—means its surface activity trumps that of classic alcohols yet doesn’t behave quite like a detergent. The stability from those carbon-fluorine bonds resists most acids and bases, protecting the compound even in tough processing conditions.
Technical Specifications & Labeling
The substance usually lands in the market at a purity topping 97% if not higher. Sensible chemical labeling emphasizes both its alcohol and perfluoroalkyl nature, reflecting concerns from regulators who want clear signals about volatility, persistence, and toxicity. GHS labels address skin and eye irritation, and up-to-date certificates of analysis from reliable suppliers come in handy for labs and factories wanting detailed impurity profiles. Containers require chemical-resistant liners. During transfer or dispensing, fume hoods and personal protective equipment aren’t just recommended—they’re a must in good laboratories and workshops. MSDS documentation covers more than the immediate hazards, pushing users to plan for environmental and chronic exposures as part of basic stewardship.
Preparation Method
Making 2-(Perfluorohexyl)ethyl alcohol involves several steps that blend the rigor of synthetic organic chemistry and the challenges posed by fluorinated intermediates. Typical routes start with a perfluorohexyl halide, which undergoes nucleophilic substitution with ethylene oxide or ethylene glycol derivatives. Some methods prefer a direct coupling using protective groups on the alcohol head to fend off side reactions before completing the deprotection. These processes take care to limit byproducts—especially perfluoroalkyl halide leftovers—due to strict environmental controls in most labs now. The product requires careful distillation under reduced pressure to avoid decomposition and to maximize yield while minimizing energy input. Waste streams, loaded with fluorinated residues, often undergo advanced capture or treatment before disposal, evidence that chemists pay attention to every drop that leaves the flask.
Chemical Reactions & Modifications
Once the molecule is ready, chemists see that alcohol group as a launching pad for new functions. Etherification and esterification slide in next, letting the compound join larger surfactant structures or block copolymers for specialty coatings. Sometimes researchers swap the alcohol for sulfonate or carboxylate moieties, tuning the physical and biological properties as needed. For certain ion-exchange resins or surface-modified nanoparticles, the perfluorohexyl tail offers superior repellency without pushing the toxicity envelope out of bounds—so long as the rest of the molecular structure cooperates. People working on greener chemistry explore partial fluorination or incorporate cleavable links, but no silver bullet has emerged to address the environmental downsides entirely.
Synonyms & Product Names
Depending on where you look—in academic literature, European product catalogues, or US regulatory documents—the chemical pops up as 2-(Perfluorohexyl)ethanol, 1H,1H,2H,2H-Perfluorohexyl ethanol, or sometimes abbreviated as PFHxEtOH. The name changes but the core structure stays, meaning that anyone handling the substance needs to decipher labels and synonyms to keep compliance and records accurate. Certain product lines in surface coatings and analytical standards call it by proprietary blends or truncated names, translating the academic lingo for industry-facing clients.
Safety & Operational Standards
Scientific consensus points toward good laboratory practice as the strongest defense against accidents and environmental release. Direct skin contact proves riskier than with common alcohols. Perfluoroalkyl chains show a habit of bioaccumulation and resistance to metabolic breakdown, so organizations take containment and exhaust filtration seriously. Storage stays restricted to cool, ventilated, and flame-resistant cabinets, ideally with secondary containment to catch leaks. Air monitoring in industrial settings prevents buildup of vapors. Disposal routes center on high-temperature incineration that cracks the tough carbon-fluorine framework, and some facilities favor chemical capture techniques to avoid atmospheric release. Any breach of containment brings problems that stick around—literally and figuratively—highlighting a responsibility that goes beyond the lab.
Application Area
Outside the controlled world of chemical plants, 2-(Perfluorohexyl)ethyl alcohol feeds into industries that value surface properties above all else. Paints and protective coatings, electronics assembly, oil recovery, and certain high-precision analytical methods all demand materials that bring together hydrophobic and oleophobic barriers. Specialty textiles and membranes count on the compact chain length to deliver water repellency without pushing regulatory red lines. Analytical chemists reach for this compound while making calibration curves for PFAS analysis, since it slips into the same environmental pathways under investigation—testing, ironically, the safety of itself and its cousins. Research teams in material science grab these alcohols to tailor nanoparticle surfaces for biological imaging, light harvesting, and separation science, recognizing the edge those highly fluorinated segments give to their experiments.
Research & Development
Behind closed doors and open-access journals, the push remains relentless to understand and improve the performance of perfluoroalkyl alcohols without repeating the public health mistakes of past decades. Analytical teams track down new metabolites and breakdown products in soil and water, mapping the toxicological fate of six-carbon compounds compared with longer chains. Chemists working on fluorinated surfactants probe for alternative ways to build the same molecular scaffolds with more tuneable or degradable pieces. Some focus on “short-chain” versions for slightly reduced persistence; others chase bio-based feedstocks or advanced polymer coatings that mimic fluorinated results with silicon- or phosphorus-based competitors. Regulatory pressure from Europe and North America raises the stakes, pushing R&D toward compounds that demonstrate defined degradation pathways, minimum toxicity, and proven separation from sensitive biospheres.
Toxicity Research
As toxicologists dig through the data on perfluoroalkyl alcohols, key findings emerge. Six-carbon perfluoroalkyl chains, while not as persistent as longer cousins, still show a reluctance to leave ecosystems or biological tissues. Researchers measure both acute and chronic effects in animal models, keeping a close eye on liver enzymes, hormone disruption, and subtle growth changes. Direct evidence for 2-(Perfluorohexyl)ethyl alcohol lands on a growing pile of case studies, some flagging low acute toxicity but noting a tendency to bioaccumulate under repeated exposure. Epidemiological surveys in communities exposed to similar compounds raise enough flags to encourage further restriction, especially where groundwater encounters run-off from production sites. Europe’s REACH legislation and US investigations pivot on these research threads, using new insights as the backbone for updated safety standards.
Future Prospects
Global debates over PFAS control don’t skip substances like 2-(Perfluorohexyl)ethyl alcohol, even if its specific risk profile seems lower than legacy chemicals. Scientific and regulatory momentum keeps shifting toward alternatives that match performance with a lighter environmental shadow. Manufacturers who once placed ultimate trust in perfluorinated chemistry now treat every new product as a candidate for full lifecycle analysis, evaluating both short- and long-term impacts. Behind the scenes, green chemistry rises as the central challenge for the next decade: finding ways to preserve water- and oil-repelling magic without saddling future generations with the cleanup bill. The conversation now draws from across academic, industrial, and grassroots voices, all recognizing that robust surface science and environmental responsibility belong together as two sides of progress.
Interest in 2-(Perfluorohexyl)Ethyl Alcohol usually comes from those working with chemicals that go far beyond everyday cleaning products or the odd bottle of bleach. This compound belongs to the family of fluorinated alcohols, and that alone ought to set off a mental bell for anyone paying attention to the way chemicals intersect with our daily routines and the planet’s well-being. Most people won’t see this chemical on the drugstore shelf, but it hides away as an ingredient in some powerhouse specialty products.
Protection and Performance: Why Fluorochemicals Like This Draw Attention
Imagine dropping your sandwich onto your raincoat and watching the condiments bead up instead of soaking through the fabric. That magic doesn’t just show up by accident. The secret lies in slick coatings and barriers, which science has refined using fluorochemicals. 2-(Perfluorohexyl)Ethyl Alcohol fills a niche here. Much of its value lies in its ability to help craft surfaces that repel water, oil, and stains. It’s no small feat to keep pizza grease from soaking into upholstery, and chemists have put this compound to work in coatings for textiles, leathers, and carpets.
This alcohol also plays a part in protecting high-end electronics. Think about devices that need to shrug off moisture from sweaty hands or the rare but catastrophic coffee spill. The protective layers that designers apply to screens and circuit boards often trace back to specialty chemicals that combine resistance to water and grime with flexibility and transparency. 2-(Perfluorohexyl)Ethyl Alcohol often gets blended in with polymers and resins to fine-tune this barrier effect. Add in uses for aerospace composites and specialized paints, and it’s clear that it makes a technical difference far removed from household duties.
The Environmental Trade-Off: Persistent Problems
By now, many people have heard stories about "forever chemicals" showing up everywhere from Arctic ice to human blood. This category covers certain perfluorinated compounds, and 2-(Perfluorohexyl)Ethyl Alcohol belongs in that conversation. These chemicals don’t easily break down, which means they can build up for years, moving through water, air, and soil. Reports have traced similar substances in water supplies, raising health questions that science still works to answer.
People have reason to worry when daily necessities and wild landscapes risk contamination from persistent compounds. Research has linked high levels of some related substances with health effects like hormone disruption and immune impacts. The point is not to panic, but to know which chemicals deserve extra scrutiny before letting them flood the supply chain unchecked.
What Can Change
The spotlight keeps growing on environmental impacts and workplace safety. Regulations around the world now push companies to justify each fluorinated molecule in their products, especially in places with high risk of direct human or environmental exposure. Brands making stain-proof and waterproof goods are rethinking their recipes and exploring safer options that don’t stick around forever in nature. Research groups look for alternative chemistries to deliver the performance demanded by industry without the environmental baggage.
In my own experience working on projects with technical fabric finishes, no one likes trading away useful features, but more people ask about what goes into durable coatings each year. Customers and designers want both the waterproof jacket and the peace of mind that comes from knowing the finish won’t cause trouble long after its time in use. The push and pull between performance and sustainability is reshaping whole markets, and compounds like 2-(Perfluorohexyl)Ethyl Alcohol find themselves right at the center of that debate. Progress comes from demanding transparency and searching out innovations that work for the long haul—on mountainsides, city streets, and wherever people live and work.
Why The Conversation About Chemical Safety Never Gets Old
2-(Perfluorohexyl)ethyl alcohol isn’t the most famous chemical in the lab cupboard, but it still raises some tough questions. Everyone has seen warnings on bottles, but experience with handling specialty chemicals has shown that safety routines start long before you pick up a flask. I remember early days in the university lab, eyes peeled for the smallest error, knowing that shortcuts with unknown substances could hurt someone. I believe conversations around chemicals like 2-(Perfluorohexyl)ethyl alcohol matter, especially as more labs work with advanced fluorochemicals.
What Do You Know About Perfluorinated Alcohols?
Perfluorinated compounds show up in more places than people realize. These are chemicals where hydrogen atoms in an alkyl chain get swapped with fluorine, and that swap makes the chain extremely stable—and extremely persistent. In 2-(Perfluorohexyl)ethyl alcohol, you get the properties that make it valuable: thermal resistance, repellency to water and oil, and chemical inertness most of the time. But it’s this same stability that turns heads on safety.
Hands-On Experience Points To Caution
In practice, handling perfluorinated alcohols calls for extra vigilance. Direct skin contact can lead to irritation. There’s often no immediate burning or sharp smell, so mistakes happen quietly. Inhaling vapors, even in small amounts, can irritate the lungs. I wouldn’t trust a fume hood less than perfect around these substances, because airborne droplets are hard to spot. Gloves have always been non-negotiable, but not all gloves keep fluorinated compounds at bay. A nitrile glove, changed out at the first sign of wear, beats risking exposure.
The Debate Over Long-Term Health And Environmental Worries
Research is pretty clear: persistent organic pollutants—the class that includes many perfluorinated compounds—linger in blood, soil, water, and air. The trouble is, these substances hardly break down. Studies on similar substances, like PFOA and PFOS, link long-term buildup to organ damage and some cancers. Regulators pay attention to these chemicals, sometimes calling for phaseouts or strict controls. While no massive public database has declared 2-(Perfluorohexyl)ethyl alcohol as acutely toxic, its close chemical cousins raise eyebrows for a reason.
How Labs And Workplaces Can Protect People
Getting careless around chemicals never helped anyone. For 2-(Perfluorohexyl)ethyl alcohol, a smart routine starts by reading every updated safety data sheet. Wear gloves, safety glasses, and never count on the naked eye to catch spills or splashes. Work in a well-maintained fume hood, and if there’s a spill, act like it’s a serious chemical until the cleanup is complete. Chemical waste management needs to keep up, too—never pour this stuff down a sink or into regular garbage. That reckless habit has poisoned enough groundwater already.
Where To Go From Here
People shouldn’t handle 2-(Perfluorohexyl)ethyl alcohol or any perfluorinated substance lightly. Even if acute risks seem low, the long-term effects are hard to walk back. Industry and research should keep searching for safer alternatives and tighter controls. Raising awareness and supporting strong safety routines make the biggest difference. My experience says it’s never safe to assume a stable-looking chemical is harmless. For modern chemistry, old-fashioned respect for the unknown protects people in ways the best glove or hood never could on its own.
Responsible Chemical Storage Protects Everyone
Anyone working with specialty chemicals like 2-(Perfluorohexyl)Ethyl Alcohol learns quickly that storage isn’t just about putting bottles on shelves. Poor handling can turn a safe lab into an expensive hazard zone. I remember early in my career, I helped move a set of what looked like mostly harmless reagents. Things went smoothly until someone neglected to check chemical compatibility during storage. One leaky cap plus proximity to an incompatible solvent led to vapor build-up, forcing an evacuation. That lesson stuck.
2-(Perfluorohexyl)Ethyl Alcohol is a fluorinated compound. These chemicals often resist heat and chemical attack, but they shouldn’t be treated carelessly. Storage isn’t just about the bottle. It’s about protecting people and extending the shelf-life of costly materials.
Practical Storage Steps to Reduce Risk
A few core rules apply for anyone handling this compound:
- Keep in sealed containers: Moisture and air degrade many specialty alcohols over time. Containers should remain tightly closed to keep out humidity and airborne contaminants. Even minor contamination can alter reactivity or ruin your next experiment batch.
- Store in a cool, dry location: I’ve seen more than one storeroom where sunlight or heat vents create a “hot spot” on certain shelves. Chemicals break down faster at high temperatures. Look for storage around 2–8°C if possible, but always away from direct sun or heat sources. Never leave bottles next to the window or above radiators—the compound’s quality wears out fast that way.
- Avoid acids, bases, and oxidizers: Mixing storage shelves for alcohols, acids, and strong oxidants spells disaster. Even sturdy fluorinated alcohols shouldn’t sit next to corrosive or highly reactive substances. This separation reduces the fallout from spills and leaks. It also keeps accidental mixing from turning a minor incident into a full-scale emergency.
- Proper labeling and dating: It seems obvious, but I’ve sorted through unmarked containers more times than I can count. Label every bottle with the compound name, date received, and date opened. That way, if product stability starts to slip, you don’t end up using degraded material and skewing experimental results.
- Ventilation matters: Some perfluorinated compounds release fumes if left uncapped or spilled. Near-airtight rooms trap these vapors, which is a problem even if the smell isn’t strong. Proper ventilation makes cleanup easier and protects everyone’s lungs.
The Benefits of Good Practice Are Real
Earning trust with chemicals boils down to a few simple routines. By sticking to consistent storage routines for 2-(Perfluorohexyl)Ethyl Alcohol, labs preserve the integrity of their stock and lower the risk for everyone nearby. Plenty of organizations fall short here—not out of neglect, but sometimes because storage recommendations aren’t enforced or training passed off as a box-ticking formality.
Precaution doesn’t need drama or red tape. Most costly mistakes happen because small details got brushed aside: an uncapped bottle, poor ventilation, incompatible neighbors on the shelf. Making good habits routine cushions against shortages, unexpected shutdowns, or worse—the kind of safety incident that gets everyone talking for months afterward.
Encouraging a Culture of Vigilance
Storage recommendations on a sheet don’t matter unless people act on them. In my years around chemistry labs, the groups with the fewest incidents rarely focused on fear. They built a culture where following a few key steps protected their work and the people around them. Keeping 2-(Perfluorohexyl)Ethyl Alcohol safe and stable keeps the work going and avoids unnecessary headaches. That’s worth more than any checklist or compliance tick-box.
What Makes 2-(Perfluorohexyl)Ethyl Alcohol Stand Out?
A lot of synthetic chemists deal with all sorts of alcohols, but 2-(Perfluorohexyl)ethyl alcohol draws special attention because it’s not just any run-of-the-mill compound. The unique thing here sits in the “perfluorohexyl” group attached to an ethyl alcohol backbone. In plain talk, part of this molecule carries a chain of six carbon atoms, every hydrogen swapped out for fluorine. The rest resembles something folks might recognize from vodka or rubbing alcohol—your classic -CH2-CH2-OH motif that defines an alcohol.
Breaking Down the Structure
If you picture it, there’s a stretch that looks like this: CF3–(CF2)5–CH2–CH2–OH. Those six carbons at the front end, all dressed up in fluorine, create a tail totally separate from most common alcohols. You get a part that wants nothing to do with water and another that actually loves it. That split between oil-loving (lipophilic) and water-loving (hydrophilic) parts opens serious doors in research and industry.
Why Do Fluorinated Chains Matter?
Anyone who's spilled a drink on their stain-resistant shirt or watched droplets skid across a raincoat has felt the power of fluorinated chemistry. Chains like the perfluorohexyl group shrug water off like it’s nothing, making them favorites for inventing new surface coatings and stain-proof fabrics. It doesn’t stop at water repulsion. These chains also show resistance against acids, bases, and even high temperatures. That’s a big deal for industries looking for performance in tough environments, like electronics or fuel processing.
Risks Hiding in Performance Chemistry
But there’s a flip side. The same carbon-fluorine bonds that fight off water and heat also hold onto life for decades, resisting normal forms of breakdown in soil and water. These compounds, which chemists call “PFAS” (perfluoroalkyl substances), have spread into drinking water and wildlife. It’s easy to overlook the impact in the excitement of new products, but stories keep surfacing about their persistence and links with health issues.
The Way Forward: Striking A Balance
Chemistry keeps raising tough questions, and the tale of 2-(Perfluorohexyl)ethyl alcohol is no different. On one hand, anyone working with tough-to-coat metals or reliable insulators turns to these molecules because they work beautifully. On the other hand, knowing what we do about environmental buildup and potential health effects, the urge to use these tools comes with responsibility. Researchers have started hunting for alternatives—ingredients that perform under fire without sticking around for ages or causing unseen harm.
Scientists and regulators should lean into testing and transparency, sharing findings with communities and industry groups. New solutions often come from conversation, not isolation. Substituting safer chain structures, designing less-persistent backbones, or capturing waste more effectively all play a part. Personal experience has shown me how quickly trust evaporates when information gets buried or brushed aside. Openness, paired with scientific rigor, carves a better path for everyone.
That’s the trick with molecules like 2-(Perfluorohexyl)ethyl alcohol—the challenge isn't just building smart materials. It’s about building trust alongside them and minding the footprint each innovation leaves behind.
Anyone digging into the world of specialty chemicals has probably run across more than a few unfamiliar names. Not many folks outside of research or advanced manufacturing would ever look for 2-(Perfluorohexyl)Ethyl Alcohol. People who need this compound usually hold a degree in chemistry or spend a lot of time with lab coats and safety glasses. The rest of us might need a compelling reason to learn who sells it, and why finding it can feel like a secret handshake.
Navigating a Tough Marketplace
This isn’t the sort of chemical you’ll catch on the shelf at Home Depot or a hobby shop. Most compounds like this—fluorinated alcohols with long, tongue-twisting names—move through channels reserved for companies or universities. Big names in chemical distribution such as Sigma-Aldrich, TCI, or Thermo Fisher have the resources and safety protocols to handle and ship specialty materials. Someone outside of a registered business, research institute, or similar organization might struggle to even place an order. These distributors comply with plenty of regulations—trade, environmental, and safety—and that matters, especially with fluorochemicals. My own forays into ordering unusual reagents for a research lab always required purchase order numbers, record-keeping, and a lot of hoop-jumping to meet compliance demands.
Safety and Regulations Lead the Way
2-(Perfluorohexyl)Ethyl Alcohol belongs to a family of chemicals drawing attention from environmental agencies. Perfluorinated compounds last a long time in water and soil. That’s not just a talking point for regulators—studies have found perfluoroalkyl substances spreading far from where anyone intended. The health impacts aren’t fully clear, but past examples with similar compounds left a trail of problems. This reality feeds into tight control over sales. Distributors need to know buyers have a legitimate reason to use and dispose of these chemicals correctly. Many places in North America, Europe, and parts of Asia enforce strong rules regarding buying and importing. From my own experience, ordering sensitive materials never ran on trust alone; paperwork, user credentials, and clear end-use declarations ruled the day.
Why It Matters
Ordinary folks probably won’t have much luck wandering online to buy this chemical. That guards against irresponsible use, dumping, or mishandling. Many communities and waterways already deal with persistent chemical pollution, and preventing potential damage takes more than goodwill. Responsible sourcing protects not only human health but ecosystems that struggle to recover from repeated chemical exposure.
Possible Solutions for a Safer System
If you’re part of a university, industrial research center, or production facility, the path to legal, safe purchase runs through approved suppliers and a string of verification steps. Make sure there’s proper training on storage, use, and disposal. Document every step. Software systems for inventory and regulatory reporting take some burden off individual memory and keep operations honest.
For researchers hitting dead ends with domestic suppliers, international distributors sometimes provide what’s needed, though import requirements can stretch patience and budgets. Stay honest about your project’s goals and always, always follow local and national laws. Society counts on transparent, careful handling of these substances so advances in science don’t end up poisoning the community.
Trust but Verify
Every time I dealt with tricky chemical orders, the lesson stuck—easy access shouldn’t mean taking shortcuts. Not everything should be bought online in two clicks. For buying chemicals like 2-(Perfluorohexyl)Ethyl Alcohol, the best choice remains working through recognized distributors, tightly managing inventory, and putting health and safety before speed or convenience.