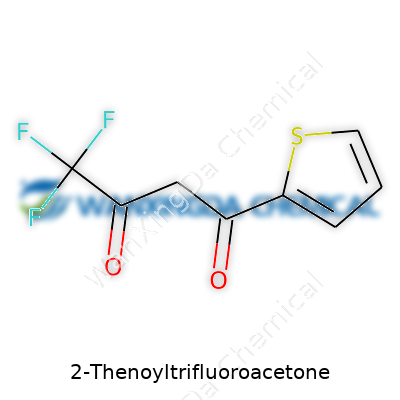
2-Thenoyltrifluoroacetone: A Chemical with Legacy and Modern Relevance
Historical Development
Back in the mid-twentieth century, chemists were hunting for ligands with distinct abilities to form complexes with metal ions. 2-Thenoyltrifluoroacetone (commonly called TTA) emerged during those quests, shaped by fundamental needs in analytical chemistry. Early studies revealed TTA’s knack for selectively binding with rare earth and transition metals in a way that improved both sensitivity and specificity. Its roots stretch into the era when metal chelates became essential in the laboratories mapping out atomic absorption and emission, and TTA found a permanent place in the toolkit for isolating elemental traces and studying coordination chemistry. This journey from scientific curiosity to essential reagent marks a story of both innovation and practical value.
Product Overview
2-Thenoyltrifluoroacetone isn’t the kind of compound that rests on corporate branding or aggressive marketing. Its quiet reputation stands on the backbone of practical achievement. Chemists reach for TTA when standard ligands falter—when they need something with a bit of backbone and subtlety. It exists as an off-white crystalline solid, with a whiff of chemical curiosity that hints at the mix of thienyl and trifluoromethyl arms on its backbone. People use TTA in a host of applications—mostly, it turns heads by making tough separations possible in analytical and radiochemistry.
Physical & Chemical Properties
Physical properties of TTA give it a solid foothold in the laboratory. With a melting point above standard room temperature, it doesn’t drift off the weighing paper as easily as some lower-melting chelators, making sample prep simpler for those with unsteady hands or drafty benches. The molecule combines a thenoyl group—adding aromatic character—with a trifluoroacetyl group, pulling electron density and lending chemical robustness. These features blend to form a compound that both resists easy hydrolysis and dissolves in most organic solvents, from chloroform to ethanol. The chelation ability of TTA pivots on these groups, creating stable metal complexes that don’t let go without effort.
Technical Specifications & Labeling
Most researchers working with TTA care about purity, particle size, and batch consistency. Chemical vendors often supply TTA in high-purity grades (often exceeding 99%) because trace contaminants can skew spectroscopic or separation results. Packaging typically comes in amber glass, since the compound shows some light sensitivity. Reliable labeling boils down to clear product names—avoiding confusion with similar chelators—and batch numbers to guarantee traceability in regulated or academic settings. Anyone who’s tracked down a spectral oddity to a mislabeled bottle knows the pain of unclear labeling.
Preparation Method
Synthesizing 2-Thenoyltrifluoroacetone starts from thenoyl chloride and trifluoroacetic anhydride, usually with catalytic support. Skilled chemists control the temperature and moisture rigorously, since water entering the reaction can throw off yields and purity. The steps begin with forming the intermediate, followed by careful distillation and crystallization. Techniques haven’t changed radically since early patent literature, but new methods aim to cut down on hazardous reagents and waste, reflecting modern green chemistry priorities. Each batch is only as good as the technician’s ability to keep the workspace dry and the conditions precise.
Chemical Reactions & Modifications
TTA serves as a starting point for a whole family of related compounds. Functionalizing the aromatic thienyl ring or tweaking the trifluoromethyl group can shift chelation selectivity, solubility, or thermal stability. Researchers routinely modify TTA to tailor ligands for actinides or special coordination geometries, especially in the quest for more efficient extractions or improved separation factors in complex matrices. Sometimes, chemists use TTA as a building block to synthesize larger, multi-dentate ligands capable of binding more than one metal ion—broadening its value well beyond its original scope. Some research labs even explore TTA derivatives as anticancer agents or fluorescent probes, opening new frontiers.
Synonyms & Product Names
Those on the ground in chemistry recognize that a compound’s name can create as many headaches as it solves. TTA goes by aliases including 1,1,1-trifluoro-4-(2-thienyl)-2,4-butanedione, or sometimes just Thenoyltrifluoroacetone. Some label it as Trifluoroacetylthienylacetone or TF-TAA. Different catalogs may sprinkle in alternative abbreviations, but seasoned researchers check CAS registry numbers to sidestep confusion. These naming quirks may feel minor, but even a single misclick can lead to the wrong reagent landing on a benchtop, derailing weeks of carefully planned work.
Safety & Operational Standards
People who work with TTA rarely treat it casually. The compound’s structure includes a trifluoromethyl group, which can kick up volatility and increase the potential for inhalation exposure. Handling calls for good ventilation, gloves, and eye protection—those who skip these steps risk headaches, irritation, or worse. Spills invite trouble if left unaddressed, especially since TTA can react slowly with water in the environment. Storage in well-sealed, labeled containers, away from direct sunlight and oxidants, remains standard best practice. Most researchers learn quickly to respect TTA’s quirks, following local or institutional guidance for disposal and emergency clean-up.
Application Area
The biggest footprint for TTA comes from analytical and radiochemistry. Technicians lean on TTA’s abilities for solvent extraction, where it grabs hold of rare earths like europium, lanthanum, or cerium, separating them out from complex mixtures. Its UV-Vis active complexes enable spectrophotometric determination of metal concentrations, making it invaluable in monitoring pollution, quality control in mining operations, or nuclear waste processing. Some labs look to TTA for more niche uses—light-emitting diodes, organic electronics, or as a probe in fluorescence tagging of biomolecules. A handful of material science researchers push its use into developing new films or sensors, inspired by its distinct chemical backbone.
Research & Development
The appetite for new applications of TTA grows with the trends pulling chemistry forward. Researchers experiment with tweaking the backbone to build smarter, more selective ligands. Ongoing projects include pairing TTA-derivatives with nanoparticles or developing cross-linked networks for selective ion exchange. There’s genuine excitement about using TTA-like compounds in luminescent devices or as medical imaging agents, tapping into metal complex luminescence. Chemists continue to publish work exploring new solvent systems, lower toxicity modifications, and routes toward greener synthesis, reflecting pressures from both industry regulations and climate-conscious grant agencies.
Toxicity Research
TTA doesn’t often appear in headlines for toxicity scares, but that’s no reason for complacency. Some studies point to moderate acute toxicity and some persistence in the environment, raising flags about long-term exposure in waste streams. Animal tests document irritation and potential central nervous system effects with high exposure, so prudence in handling and disposal matters. Environmental health and safety teams monitor emerging data, pushing for closed-system handling and reducing open-air experiments. Looking ahead, as more TTA-based ligands emerge, consistent toxicity profiling grows in importance, not just for bench safety but also for long-term ecological impact.
Future Prospects
Looking into the horizon, 2-Thenoyltrifluoroacetone stands poised both as a stalwart of current analytical methods and a springboard to new chemistry. The challenge remains in designing derivatives with improved safety profiles, lower environmental impact, or greater use in healthcare and high-tech devices. The rising interest in recycling rare earths and the burgeoning fields of clean energy and electronics give TTA a platform for innovation. With careful stewardship, continuous improvement in synthesis, and creative exploration into new frontiers, TTA’s future shines as brightly as its chelates under laboratory lights.
Most folks outside of research labs haven’t stumbled across 2-Thenoyltrifluoroacetone. It doesn’t roll off the tongue, and you certainly won’t see it on a grocery shelf. For scientists working with metals and probing the hidden corners of chemical reactions, though, this compound is a staple. In my own years fiddling around with lab glassware, this substance kept popping up right at the crossroads between chemistry’s theory and its daily grind.
The Chelator Chemists Count On
2-Thenoyltrifluoroacetone, usually shortened as TTA, grabs onto metal ions like a determined child clings to a toy. With its knack for “chelating,” or binding metal ions, TTA features in tests measuring trace metals in everything from polluted water to food samples. Researchers and technicians favor TTA for its reliability—once it latches onto a metal ion such as iron, copper, or lanthanide elements, it stays put, forming stable complexes. When I worked in labs focused on environmental testing, TTA helped us see tiny traces of contaminants nobody would trust in their drinking water.
Beyond just detection, these TTA-metal complexes play a starring role in separating rare earth elements. These metals form the backbone of electronics, magnets, and even medical imaging equipment. Chemical engineers rely on TTA in the extraction and purification grind. The process leans on simple chemistry, but without compounds like TTA, tailings and waste would bury efforts to reuse, recycle, or clean up after industry.
Lighting Up Science—Literally
One trick TTA pulls off involves rare earth ions and bright, greenish glow. Scientists take advantage of its properties to create complexes for advanced materials, including the technology inside fluorescent lamps, lasers, and medical tracers. I once watched a laser demonstration that started with someone mixing TTA and a rare earth salt on a lab bench, resulting in a compound that shimmered under UV light. This sort of real-life magic drives innovation in imaging, display screens, and optical devices. Researchers searching for energy-efficient solutions view TTA as a way to improve light emission and signal detection.
The Bigger Picture: Safety, Research, and Regulations
TTA’s ability to wrap itself around metal ions brings benefits but also risks. Improper handling, like with many chemical compounds, spells trouble for both workers and the environment. In well-regulated nations, labs follow strict guidelines for safe storage, use, and disposal. Across borders, regulations vary. Some regions lack safety awareness or equipment, meaning the same tool that helps one team measure pollution could cause headaches for another struggling with waste management.
Transparency, training, and better access to modern lab gear would cut down on many such problems. Universities and companies that invest in education make a difference. For those outside the lab, knowledge is power. Understanding that TTA is part of the toolkit that underpins water analysis and clean tech industries highlights its importance—and points toward solutions grounded in collaboration between scientists, regulators, and the public.
Why TTA Will Stick Around
Tools like TTA play an overlooked but powerful part in making sense of what’s in our soil, water, and high-tech gadgets. These compounds keep showing up because they work, plain and simple. From clean energy ambitions to more reliable food safety checks, 2-Thenoyltrifluoroacetone fits into daily life, even if most people never hear its name. Its story shows how behind every busy laboratory door, science connects to broader health and sustainability goals.
Chemistry in the Real World
There’s something almost magical about chemical compounds with the power to unlock complex puzzles in everyday science. One that often stays under the radar but quietly shapes big advances is 2-Thenoyltrifluoroacetone, or TTA. Anyone who has fiddled in a chemistry lab or read up on rare earth metal extraction eventually stumbles upon this complicated-sounding molecule. Once you look past the name, though, the real story comes out—not in the abstract, but in a mix of actual, studied properties and the practical jobs it takes on.
Structure Fuels the Story
TTA is easy to pick out by its structure, sporting both a trifluoromethyl group and that familiar five-membered sulfur-bearing ring—the thienyl group. This special combination pushes TTA into the spotlight for what chemists call “beta-diketones.” Beta-diketones are famous for their knack at chelating metal ions. Chelation means grabbing onto metal atoms, locking them in like a clam clamps down on a grain of sand. In the lab, I’ve watched this play out when TTA snatches up lanthanide or actinide metals, forming beautifully colored complexes with real uses in separation technology and luminescence research.
Reactivity: The Strong and the Subtle
With its architecture, TTA has both strength and subtlety. The strongly electronegative trifluoromethyl group pulls electrons away from its neighbors, giving the molecule a twist of acidity in the hydrogen attached to its central carbon. That’s more than just trivia—it means in the presence of a base, TTA forms an anion that grabs metal ions tightly, forming stable, often crystalline materials. You see this first-hand when a solution changes color rapidly as TTA meets a metal salt. This reactivity isn’t just a neat lab trick; it serves industrial-scale separation of rare elements, giving high yields because of the selectivity of those forces at play.
Solubility and Real Applications
What makes TTA even more helpful in science is its ability to dissolve in organic solvents such as chloroform or benzene, but not so much in water. Every chemist who’s tried separating metals from junk knows the headaches water-based extractions bring. TTA cuts down on noise, offering clean, almost surgical layers of separation when mixed with the right solvents. This pays off in nuclear chemistry, pharmaceutical work, and even luminescent material production for screens and sensors that show up in daily tech.
Potential Pitfalls and Straightforward Steps Forward
Working with TTA brings some baggage. Its chemical structure gives it enough volatility and skin absorption to warrant gloves and good ventilation. From experience, shortcuts here cause more trouble than they save, so established safety routines already go a long way. In a bigger context, handling organic solvents poses wider pollution and health questions. One answer already on the table comes from researchers looking at greener solvents paired with TTA, or reusing the molecule in closed systems to cut down waste.
Why This Matters
Even if someone never sees a jar of TTA, its chemistry shapes more than labs or academic journals. Better extraction of rare metals feeds into smarter batteries, safer medical scans, and lasting LEDs. Understanding and respecting the chemical properties of compounds like TTA helps point the way not just to new discoveries but safer and more sustainable technology for everyone. These connections, grounded in the science, show the value of keeping one eye on the molecular details and the other on the bigger picture.
Digging Into 2-Thenoyltrifluoroacetone: More Than Just Letters and Numbers
Molecules, whether they sit in a flask or in the memory of a struggling chemistry student, play a huge role in shaping the way laboratories function. Take 2-Thenoyltrifluoroacetone, an organic compound chemists love for its knack for forming complexes with metals. Its molecular formula, C8H5F3O2S, might look like a secret code, but these letters and numbers pull back the curtain on a world of practical chemistry and plenty of real-world applications.
Molecular Details Everyone Should Notice
Breaking down that formula, you spot eight carbons, five hydrogens, three fluorines, two oxygens, and a single sulfur atom. This makeup lays the groundwork for the compound’s behavior. You get a classic diketone backbone fused with a thiophene ring. That’s not trivia; it actually governs how the molecule interacts with metals in solution, especially in the context of separation processes and extraction techniques common in both the lab and industry. The trifluoromethyl group’s presence isn’t just for show. It modifies the molecule’s solubility and enhances its ability to bind with certain metal ions, giving chemists a reliable tool for isolating rare earth elements, refining nuclear materials, and even running analytical tests for environmental monitoring.
Going Beyond the Textbook: Why This Formula Stands Out
During my own trek through graduate-level chemistry, I remember late nights grappling with separation science, and 2-Thenoyltrifluoroacetone wasn’t just background noise. The compound’s special knack for forming chelates offers anyone handling toxic metals a safer, cleaner way to work. Unlike some extraction reagents loaded with environmental baggage, this molecule’s structure allows for strong selectivity. A chemist can grab onto precious heavy metals without loading the environment with useless waste. The downside comes out if disposal isn’t handled with care, as organofluorine compounds don’t just fade away when poured down the drain. That’s a reality the entire chemical industry faces day after day: finding the balance between performance and responsibility.
Supporting Responsible Chemistry With Fact and Experience
Reliable extraction of actinides or lanthanides for nuclear and electronic uses is tricky. The formula behind 2-Thenoyltrifluoroacetone has made it a staple in the toolbox for precisely this reason. Studies show robust performance when isolating uranium or separating traces of rare earths. In some cases, researchers have even used it to monitor water contamination by selectively binding to heavy metals, sparing communities from exposure to dangerous pollutants. On the other hand, regulatory agencies and environmental chemists have pushed for alternatives wherever fluoro-organic compounds create persistent pollution. Still, nothing overtakes the combination of selectivity and ease this molecule provides, especially in old-school chelation and solvent extraction.
Room for Progress and Safer Alternatives
No conversation about complexing agents gets far before sustainability pops up. Chemists need to keep pushing for alternatives with less environmental persistence. Bio-based ligands and greener solvents offer hope, though today they rarely match the muscle that the trifluoromethyl and thienyl components bring to the table. Until replacements catch up, knowledge of the molecular formula of 2-Thenoyltrifluoroacetone—C8H5F3O2S—remains essential for both careful application and responsible stewardship, helping chemists make better decisions for people and planet alike.
Toxic Risks Get Downplayed, But They Demand Attention
2-Thenoyltrifluoroacetone, known to chemists as TTA, pops up in many research labs. Folks use this compound for separating metals like lanthanides from solutions, and you’ll spot it in studies exploring metal detection or complex synthesis. TTA sounds like technical lab stuff, but the danger behind these chemicals rarely gets enough sunlight. It’s easy to shrug off as a concern for people in white coats, but the truth is, TTA reminds us that some chemicals come packed with real-world risk, even outside the science bubble.
Poking Holes in the Comfort Zone
I’ve seen lab workers get too comfortable handling exotically named substances, trusting gloves and goggles to shield them from every mishap. TTA shouldn’t inspire laid-back habits. Experts with the National Institute for Occupational Safety and Health (NIOSH) point to organofluorine compounds, like TTA, as possible threats to both skin and breathing. TTA fumes can irritate your nose, throat, and eyes. Take a deep breath near a spill, and you might wrestle with headaches, dizziness, or nausea. If the compound gets on your skin or splashes into your eyes, you risk burns. Ingest it? Now you face stomach pain, vomiting, or something worse.
Lab reports aren’t loaded with stories about TTA poisoning, which may give people false confidence. Limited testing doesn’t erase the troublesome side of this compound—it only reflects that more research is needed. TTA hasn’t cleared every regulatory review; instead, authorities flag it for careful handling and good reason.
Weighing Exposure in the Workplace
Many research settings skip proper ventilation, and old habits like open-beaker prep still pop up. I remember watching a grad student mix reagents by a window, thinking a breeze would replace a fume hood. Simple shortcuts like that add up. TTA turns volatile at room temperature, sneaking vapors into the air. Without real exhaust systems, workers inhale these vapors far too often. Contact with skin might go unnoticed if someone ignores a torn glove or dusty bench. Those small exposures have a way of adding up, and sometimes symptoms show up long after the fact.
Outside the lab, shipping and disposal make the compound’s risks everyone’s business. If TTA spills during transport or leaches into waste streams, communities nearby face exposure. The Environmental Protection Agency recommends treating this chemical as hazardous waste. There’s a call for care far beyond the researchers handling beakers.
Fixing the Problem Means Taking Personal Responsibility
No one can depend only on warning stickers or a dusty shelf of safety rules. I back up every claim with my own routine: double-checking gloves, refusing to pipette out from open flasks in shared spaces, and talking with colleagues about odd-smelling air. TTA becomes less threatening in a lab where people ask smart questions and train newcomers on why certain compounds get their reputation. A fresh safety audit, better ventilation, and regular talks make a difference. Also, waste management isn’t a task for the last person in the room. Community health ties back to every jar of hazardous chemical, and how it gets handled at every step.
Imagine research labs and chemical plants that stop taking risky shortcuts—not only for workers’ sake, but for neighbors too. Treating TTA’s dangers with respect depends on action, not just knowledge, and every person plays a part in keeping science safe for all.
Keeping Chemistry Safe at the Bench
Stepping into any lab, you see shelves packed with bottles—each needing its own kind of handling. Among the small-label names, 2-Thenoyltrifluoroacetone stands out for its clear demands. Working with chemicals for years, I’ve seen how basic common sense and care make all the difference between a safe lab and a risky one.
Risks Aren’t Just Academic
Chemists and lab techs won’t be surprised to hear this: trifluoroacetone derivatives react. What matters most is that this compound can evaporate and irritate, especially if the weather warms up or sunlight touches the bottle. Young researchers often overlook the slow creep of chemical vapors in a warm storeroom. Keeping 2-Thenoyltrifluoroacetone away from direct sunlight—ideally in a tightly closed glass bottle—isn’t just a best practice, it keeps everyone breathing easier.
Why Temperature and Ventilation Matter
From my experience, the temperature inside some storage rooms moves up and down through the seasons. Storing this compound cool and dry helps reduce risk, since it won’t break down as quickly or give off extra fumes that way. Chemical breakdown can cause dangerous byproducts—even if the bottle looks fine on the outside. It is always better to use a lockable cabinet, away from heat sources, with airflow good enough to sweep away accidental vapors if the cap ever fails.
The Problem with Leaky Lids
Any old chemist will tell you—more accidents come from neglected storage than any dramatic spill. A leaky cap or an old stopper turns a stable chemical into a headache (sometimes literally). Glass bottles with proper seals outlast thin plastic containers and won’t react the way many plastics might, especially with solvents like these. Marking the date on the bottle helps remind everyone how old it is. Clearing out expired bottles every year keeps clutter to a minimum and lowers the odds of a surprise problem.
Thinking Beyond the Storage Cabinet
It’s never just about one person; safety grows from habits at every level of the lab. I’ve seen great labs where student workers double-check labels and update records each month. They don’t rely on assumptions—if labels fade, they ask. If a bottle looks off, they flag it. This attitude beats any checklist handed down from above.
Working Smarter
Some changes in practice cost almost nothing but pay off every day. Putting similar chemicals together, away from strong acids or bases, stops cross-reactions before they begin. Storing 2-Thenoyltrifluoroacetone in a separate, labeled area gives extra peace of mind. Sometimes simple steps—like using secondary containers in case of drops or leaks—help catch small spills before they spread.
Staying Informed Means Staying Safe
Chemists and lab support staff who stay up to date on safety information set the bar for everyone else. Reading safety data, sharing what you find, and talking openly about storage concerns help teams avoid old mistakes. Publicly available guidelines from chemical safety organizations stress all these points for a reason: they work. The key is real-world commitment, not empty ritual.