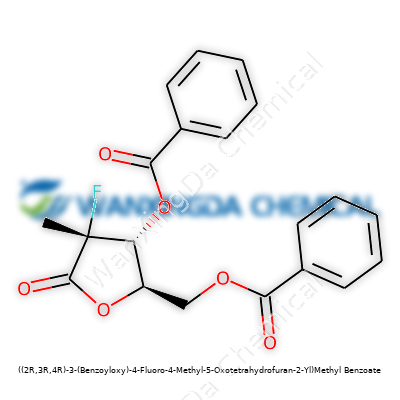
Shining a Light on ((2R,3R,4R)-3-(Benzoyloxy)-4-Fluoro-4-Methyl-5-Oxotetrahydrofuran-2-Yl)Methyl Benzoate: Its History, Properties, and Where It’s Headed
The Roots and Rationale Behind This Compound
The never-ending hunt for more precise, tailored molecules in organic and medicinal chemistry led to ((2R,3R,4R)-3-(Benzoyloxy)-4-Fluoro-4-Methyl-5-Oxotetrahydrofuran-2-Yl)Methyl Benzoate. Synthetic efforts in the late 20th and early 21st century focused heavily on combining functional groups that could nudge molecular behavior in new directions. The structure here, with its benzoyl esters and the distinctive presence of both fluorine and methyl groups, reflects real work in chem labs attempting to create scaffolds that could be used as pharmaceutical building blocks, enzyme inhibitors, or specialized intermediates in fine chemical synthesis. Drawing from a tradition that began with relatively simple benzoate esters and evolved as organic fluorine chemistry found its way into the mainstream, this molecule embodies the way scientists keep pushing complexity and performance, bit by bit.
Peeling Back the Layers: Product Overview
Anyone familiar with organic synthesis can spot why chemists would get excited about this molecule. The core, a substituted tetrahydrofuran ring, means you get rigidity and a certain set of spatial relationships between groups. Add in that fluorine atom—well known for changing how molecules interact with biological targets—and the interest runs deeper. Modifying core structures with benzoyloxy and benzoate moieties brings the potential for controlled hydrolysis, and these changes shape how the compound dissolves, reacts, or interacts in various settings. In short, this isn’t just an abstract oddity; it’s the kind of molecule that represents new value in specific pharma research or as a versatile intermediate for new chemical entities.
What Sets the Substance Apart: Physical and Chemical Features
It's easy to understand the fascination with the physical and chemical quirks of this compound if you've ever watched a batch change color, solidify, or dissolve in minutes under the right conditions. The fluorine gives it a bit more resistance to metabolic breakdown as well as higher electronegativity, shifting both reactivity and binding profile. As a solid, its melting point will be noticeably higher than smaller esters, reflecting increased intermolecular interactions. In solution, it tends to behave predictably in organic solvents such as dichloromethane or ethyl acetate, as both the benzoate and benzoyloxy groups lend a certain lipophilicity. The combination of aromatic and aliphatic functionality makes the compound compelling for further transformations—chemists look for exactly these features if they want both stability and reactivity when they prepare related targets.
Key Specifications and Proper Labeling
Quality control, down to the last impurity, drives modern chemical industry practice. Exacting standards for isomeric purity, clarity, and content of trace metals keep this molecule ready for use in high-stakes R&D. Proper labeling, supported by accurate IUPAC naming and registry numbers, helps avoid confusion—a must, especially when one wrong isomer can throw off a crucial biological screen. In practice, solid storage under dry, inert conditions remains the norm. Synthetic chemistry, for all its futuristic flavor, still relies on careful attention to these details because a single deviation can affect downstream research or commercial viability.
Making and Tweaking: A Closer Look at Preparation and Reactivity
Assembling ((2R,3R,4R)-3-(Benzoyloxy)-4-Fluoro-4-Methyl-5-Oxotetrahydrofuran-2-Yl)Methyl Benzoate draws from both classical esterification and more recent advances in selective fluorination. The route typically starts with functionalizing a protected tetrahydrofuran core, with stepwise protection and deprotection steps to avoid unwanted reactions at sensitive positions. Benzoylation, often via benzoyl chloride or related agents under basic conditions, attaches the aromatic groups, and the incorporation of fluorine comes with its own challenges—often handled through nucleophilic fluorination of a pre-activated intermediate. Each modification, right down to the last step, can be tuned by adjusting temperature, solvents, or stoichiometry—skills that come only after long days in the lab bench, working through more than one failed batch. Once prepared, there’s room for further modification: hydrolysis of the ester, oxidation of the methyl group, or even additional substitution on the aromatic rings. Every research group with a penchant for method development dreams up new ways to tweak such molecules, searching for just the slightest edge or altered property.
Understanding the Role of Synonyms and Alternate Product Names
Any seasoned chemist learns fast that molecules of this complexity often go by more than a single label. Between the full IUPAC clunker and shorter, more practical descriptors, tracking literature and replication demands attention to all possible names. Synonyms gather from main functional components, such as “benzoyloxymethyl tetrahydrofuran fluoro methyl ester,” and sometimes from internal project codings—though only formally published variants carry weight beyond a company’s walls. This loose nomenclature landscape doesn’t just complicate literature surveys; it can trip up procurement, data mining, or regulatory filings if not managed with rigor. Keeping records consistent stands as the only real fix; that level of diligence defines credible work.
Operational and Safety Considerations in Practice
No one who’s mixed organic fluorinating agents or benzoyl chloride forgets the sharp reek or occasional splash hazard. The safety profile for this molecule mainly ties to its reactive precursors and the behavior of the ester and fluorine moieties, both of which call for gloves, eye protection, and solid fume hood airflow. Like most aromatic esters, the risk profile doesn’t spark panic—handling with typical care for organic chemicals suffices at the bench scale. At industrial scale, though, the focus shifts: dust from finely-divided solids, runoff from spills, and waste disposal set the stage for more detailed protocols. Documentation of storage stability, incompatibilities, and breakdown products helps maintain safe, repeatable handling far beyond the research environment. The best labs put as much faith in procedural discipline as they do in new chemistry.
Where Chemists and Industry Put This Compound to Work
This isn’t a household name in staple industries, but for project teams in pharmaceutical research, chemical biology, or material science, it provides a welcome option for developing new candidate molecules. The dual benzoyl and benzoate moieties mean it sees use in processes needing staged deprotection or selective hydrolysis. The tetrahydrofuran core, functionalized with fluorine and methyl, hints at why researchers in metabolic pathway probes or enzyme inhibition might build analogs to this template. Fine chemical synthesis also draws on such scaffolds to route pathways to more complex end targets. Access to reliable intermediates shortens timelines and sidesteps bottlenecks, and in specialized devices or analytical chemistry tools, new modifications based on this molecular backbone still find fresh ground. Turns out, real progress often rides on access to just such versatile compounds.
What the Lab Really Learns: Ongoing R&D and Toxicity Testing
Crucial insights into this molecule’s behavior haven’t stopped at the benchtop. Researchers, in both university and commercial settings, keep pushing for more data on metabolic fate, possible off-target effects, and environmental persistence. Most fluorinated organic molecules raise eyebrows for their stubbornness in natural systems—once you make them, breaking them down rarely comes easy. Early toxicity screens suggest that, like many esters, the core toxicity profile looks manageable, provided exposure is controlled and occupational limits followed. Ongoing in vitro and in vivo studies continue clarifying residue buildup, excretion pathways, and acute effects on model organisms. More than just compliance, this represents the responsible course for any chemistry driving modern commercial or medical applications. Thorough research keeps the field credible, and only repeated, peer-reviewed work will clarify both risks and potential.
Looking Toward Tomorrow: Future Prospects and Next Steps
The future for this compound, and others like it, sits closely tied to broader trends driving chemical and pharmaceutical progress. Engineers and process chemists keep searching for greener or more scalable fluorination techniques; synthetic organic chemists explore expanded utility for novel esters and protected alcohols, especially as building blocks in the design of new drugs. With tightening regulations on toxic and persistent chemicals, efforts to make safer, more biodegradable analogs spring from the foundational work done on molecules with similar cores. The value of this compound depends on its adaptability—every added functional group or reactive position opens new doors for tailored roles in intricate synthesis or precise biological targeting. Only continued, collaborative research and real engagement with both safety and sustainability will write the next chapter for this class of molecules.
The Human Side of Scientific Curiosity
Every molecule tells a story, and some stories require breaking things down to the basics to understand why they matter. The chemical name ((2R,3R,4R)-3-(Benzoyloxy)-4-Fluoro-4-Methyl-5-Oxotetrahydrofuran-2-Yl)Methyl Benzoate sounds like the tongue-twister some people rushed through in university labs. That weighty name isn’t just a pile-up of jargon—it encodes an entire architectural blueprint for a little chunk of matter. In chemistry, each detail points to a specific part of a molecular machine, so learning to read a name like this opens up a richer view of what the molecule does out in the wild.
How Structure Gives Personality
The tetrahydrofuran ring forms the backbone, a five-membered structure with one oxygen atom tucked in alongside carbon neighbors. Most folks in a research setting recognize this ring by its stability, often featuring in drug scaffolding and building blocks for more complex compounds. Bring in the “4-Fluoro-4-Methyl” badge, and the molecule shifts—fluorine often changes how the whole thing interacts with the rest of the world. Chemists know fluorine slides into organic rings easily and likes to stick around, adding metabolic resilience and sometimes even a dash of unpredictability in biological settings.
The “3-(Benzoyloxy)” and “Methyl Benzoate” markers call out two aromatic additions—a benzoyl group attached by an ester linkage, which tends to show up for both protection of reactive sites and influence on molecule shape. Aromatic rings don’t just lend a certain elegance, they make molecules more likely to dissolve in some places, less likely in others. At this point, real-world impact becomes clearer. Protection groups like benzoyl often surface in the process of making pharmaceuticals and advanced materials. They keep sensitive oxygen atoms or alcohol groups from reacting before the right moment, much like a jacket thrown over delicate glassware.
Why Stereochemistry Matters
Peel back another layer and the “(2R,3R,4R)” notation gets personal—the molecule’s handedness. Anyone who has set up a chiral reaction in the lab knows the struggle. The body’s enzymes, for instance, treat each enantiomer as totally unique, sometimes firing it through a keyhole, sometimes stopping it dead. Pharmaceuticals featuring these chiral tags need selective synthesis, because a misstep doesn’t just waste material—it could flip a medicine’s character, which brings both regulatory nightmares and practical headaches.
From Naming to Practice
On paper, the chemical structure makes sense, but the journey from notation to real impact depends on how these pieces behave together. For research teams, mapping these connections means running time-intensive characterization: hitting the compound with NMR, mass spectrometry, and IR, just to confirm the architecture matches expectations. Students who grimaced at three-hour spectra readings know this pain well. On the upside, recognizing the whole framework early lets chemists predict solubility, reactivity, and potential therapeutic value.
Real-world Relevance and Solutions
Instead of wading knee-deep in endless speculation, it helps to look at how chemists practically approach such structures. Stereochemical labeling acts both as a guidepost and a warning sign. Miss the mark in a synthesis and the product can lose function or even safety. The best labs build in steps for rigorous chiral analysis, using both cutting-edge chromatographic separation and seasoned hands with a pipette. To make progress, many teams share data openly and develop clearer tools for visualization, trying to ease the steep learning curve.
Digging into real chemistry—down to each atom—matters, not only because the molecule might someday matter for new materials, drugs, or diagnostics, but also because tackling these challenges gives researchers hands-on experience with teamwork, precision, and creative troubleshooting. That’s where the biggest lessons often lie.
The Many Roles a Compound Can Play in Modern Life
Most folks don’t spend much time thinking about what’s behind the things they use every day. Take a humble chemical compound—for years, it might just sit tucked away in lab drawers or busy factories. Look beneath the surface and you’ll find these compounds making life safer, more comfortable, and sometimes tastier.
Let’s look at one that pops up everywhere you turn: sodium bicarbonate. It’s not only in the box in your fridge but helps firefighters tackle kitchen grease fires and doctors use it to treat certain metabolic issues. At home, a sprinkle in the laundry keeps clothes fresh. Cooking wouldn’t be the same without it. That simple white powder lifts cakes and pancakes, making brunch a treat instead of a brick. These jobs go far beyond the stereotype of chemistry as dry or distant.
Industry Relies on Consistent Performance
Factories often need compounds nature doesn’t provide directly. In everyday construction, take Portland cement. Every sidewalk, bridge, and building depends on its strength and its ability to mix well with water and sand. This simple mixture makes cities possible. Getting it right prevents disasters—there’s no room for shortcuts when structures have to last for decades.
Pharmaceuticals lean on chemical compounds in equally practical ways. Think of aspirin, for instance. Generations have relied on it to knock out pain, reduce fever, and even lower the risk of stroke. The process that churns out billions of pills each year uses protocols first hammered out decades ago. Every pill needs to deliver exactly what’s promised, or someone’s health suffers.
Protecting Health and the Environment
Some compounds help keep communities healthy in less obvious ways. Consider chlorine-based disinfectants. Local water treatment plants use these to kill bacteria and viruses so families don’t worry about what’s coming out of the tap. Experience shows that budget cuts or mismanagement put lives at risk, as seen in waterborne disease outbreaks across the globe. You only notice the absence of such protections when they falter.
Further up the supply chain, agricultural fields depend on compounds like ammonium nitrate, used as fertilizer. Crops grow faster, and yields go up, turning barren soil into productive farmland. On the other side, misuse has fueled pollution or even tragedy when handled unsafely. The need for tighter rules and better education grows as risks increase—modern farming depends on striking this balance.
Supporting Innovation and Sustainability
Innovation can’t happen without the right materials. The boom in electric vehicles owes much to advances in lithium-ion battery technology. Lithium compounds let batteries hold more energy and last longer through thousands of charge cycles. As more drivers switch away from fossil fuels, demand for such compounds will only increase.
A responsible path forward means investing in research for alternatives, recycling technologies, and cleaner methods of extraction. Scientists in my own circle talk about sustainable chemistry as the next big frontier. If we hope to hand a livable planet to future generations, these compounds must stay both effective and safe.
The Big Picture: How Compounds Shape Daily Life
From the bread on your table to the power in your car, everyday compounds maintain the rhythm of society. They save time, prevent disease, and sometimes transform whole industries. Jobs, health, and the planet itself depend on how wisely people use and regulate these building blocks. Fact-driven oversight and a commitment to ethical use can keep these silent helpers working for everyone’s benefit.
Why Storage Quality Makes or Breaks Compound Value
Anybody with hands-on lab experience knows how fast things can go sideways thanks to simple storage mistakes. Chemicals, whether delicate or robust, do not behave magically well just because the label outlines their chemical structure. Take ((2R,3R,4R)-3-(Benzoyloxy)-4-Fluoro-4-Methyl-5-Oxotetrahydrofuran-2-Yl)Methyl Benzoate. This kind of molecule, with its ester bonds and a fluorinated ring, won’t tolerate slapdash storage.
Keys to Protecting the Compound’s Integrity
Too much heat, sunlight, moisture, or oxygen changes outcomes for all sorts of compounds, especially those carrying sensitive groups. Exposure means the risk of hydrolysis or oxidation jumps. It’s about more than simple product loss—contaminants, unpredictable byproducts, upset experiments, and wasted money all lurk when you skip proper handling.
Based on the structure here, you get a sense of what the molecule doesn’t like. Ester groups break down in the presence of water or strong bases. That’s not speculation; it’s straight out of every organic chemistry bench failure. Leaving these types of compounds on the bench top might seem fine for a few hours, but breakdown isn’t always dramatic. Sometimes a subtle change in melting point or a few strange peaks in the NMR spectrum signal trouble—usually after it’s too late to fix.
Recommended Storage Practices from Direct Experience
People sometimes stash chemicals in random cabinets because of habit or lack of guidance. That approach leaves everyone guessing about potency or safety. Here’s what has worked best for compounds like this one, both from standard practices and from hard-won lessons in working labs:
- Store under inert gas if possible. For air- and moisture-sensitive samples, argon or nitrogen prevents oxidation and hydrolysis. No, it’s not just for “super sensitive” materials. Oxygen sneaks in cap threads and does real damage over weeks or months.
- Refrigerate or freeze below room temperature. Fluctuating room temperatures speed up degradation reactions. Keeping this class of compound in a fridge at 2-8°C or a dedicated freezer brings the rate of decomposition way down. It also helps avoid accidental heat that might arise from nearby equipment or sunlight.
- Keep dry and tightly sealed. Even a touch of atmospheric moisture inside the vial can trigger problems. Desiccators or sealed vials with fresh desiccant packs work as a good backup. I’ve seen half-used vials sprout visible crystals of hydrolysis byproducts because the lid didn’t fit snugly.
- Avoid strong light. Many aromatic esters are light sensitive. Non-transparent secondary containers or foil-wrapped vials give extra security. Exposure to ambient lab light sometimes causes slow discoloration or degradation in compounds sitting on upper shelves.
The Ongoing Importance of Reliable Storage
Cutting corners on storage undermines research and trust. Failures seldom announce themselves up front. Instead, odd results pile up or shelf lives shrink, frustrating researchers and costing money. With the cost of rare and complex compounds rising, it makes sense to put effort into getting storage right.
Documenting the storage approach pays off as well. Down the road, tracing issues back to storage helps avoid repeating old mistakes. Having a record keeps teams on the same page, even when people rotate or move on.
Moving Forward: Culture Over Instructions
Nobody enjoys tossing out degraded chemicals or repeating failed syntheses. Storing compounds like ((2R,3R,4R)-3-(Benzoyloxy)-4-Fluoro-4-Methyl-5-Oxotetrahydrofuran-2-Yl)Methyl Benzoate under chilled, dry, airtight, and low-light conditions isn’t just textbook advice—it’s habit born from experience and reinforced by every experiment that worked out because the material stayed pure. Clear guidelines, easy-to-access fridges or freezers, and shared responsibility make the difference.
Taking Safety Seriously — Everyday Choices, Everyday Impact
Most people look at a product label, glance at a few warnings, and move on. Maybe it’s cleaning supplies, DIY adhesives, or gardening chemicals. Sometimes, companies print out instructions like they’re covering themselves for a lawsuit, but forget that real people have to make sense of those rules while juggling a dozen tasks. No one expects to spill bleach on their jeans or discover that mixing two harmless-seeming liquids builds up toxic gas. Safety habits often start with a single simple step: seeing risk where most folks see routine.
No One Thinks It’ll Happen to Them—Until It Does
Years ago, a buddy of mine ended up in the ER—red skin, stinging eyes. All because he skipped gloves while opening a new paint remover. These stories crop up in every household, workshop, and yard. People trust the familiar. They downplay what can go wrong with a bottle that sits on the same shelf with peanut butter and dish soap.
Accidents rarely announce themselves with a trumpet. Usually, it’s a small splash from a bottle. Or leftover residue on your hands before you rub your eyes. The trouble isn’t so much in the product alone but in the shortcuts we take.
Labels Are Written for a Reason
There’s real knowledge behind the bold warning signs. The U.S. Consumer Product Safety Commission reports thousands of injuries each year tied to misuse or mishandling of core household and industrial products. Whether it's harsh chemicals, volatile adhesives, or even strong cleaning agents, injuries are real. Eyesight fails. Skin burns. Even the air we breathe behind a closed window can turn dangerous if a product releases gases.
Gloves and goggles may look clunky, but they’re cheaper and far less embarrassing than a trip to urgent care or a frantic internet search for antidotes. Ventilation seldom gets enough attention either. Airing out a room can save lungs, even if the smell isn’t too strong. Most instructions are grounded in lab tests and federal standards, not just tradition.
Kids and Curiosity—A Dangerous Combo
Everyday storage habits matter, too. I’ve seen parents stash bottles under the sink or leave colorful containers on low shelves. Children see bright labels, reach for what seems new, and taste-test things just because. Poison control centers stay busy for a reason. It isn’t always neglect—sometimes it’s just forgetting how fast little hands can move.
Simple tools like high shelves or childproof caps change the odds. Secure storage can mean the difference between peace of mind and an emergency call.
Building Better Habits—Step by Step
Changing behavior doesn’t need heroics. It starts with routines. Read the label. Wear gloves for harsh sprays or unknown substances. Never assume two products can mix together harmlessly, even if each seems fine on its own. As much as possible, keep workspaces airy. Wash hands, close bottles, and keep them out of sight and out of reach.
No rule covers every situation, but respect for risk makes everyday life safer. No shortcut outweighs health, comfort, or the security that comes from knowing you’re not leaving safety to chance.
The Realities of International Shipping for Specialty Chemicals
Researchers and product developers keep discovering new molecules like ((2R,3R,4R)-3-(Benzoyloxy)-4-Fluoro-4-Methyl-5-Oxotetrahydrofuran-2-Yl)Methyl Benzoate. The possibilities for medicine, agriculture, and materials science keep stretching. At the same time, navigating the web of international chemical shipping laws rarely sparks the same excitement among scientists. Instead, it tends to spawn stress, delays, and sometimes outright scrapped projects. I’ve seen plans get held up for months at customs just because some paperwork was missing, or a customs agent somewhere flagged something as suspicious.
Rules, Risk, and the Pursuit of Knowledge
Moving a novel compound between countries is nothing like mailing a book or a box of cookies. Many nations have regulations built on decades of safety incidents, environmental caution, and political risks. Interesting chemicals, especially the obscure ones, often hit extra scrutiny thanks to controlled substance lists, dual-use rules, and shifting interpretations of what’s allowed. There’s a legitimate reason for vigilance — everyone wants to prevent misuse, accidental exposure, or environmental release. Some chemicals morph from laboratory breakthrough to regulatory minefield simply because their structure resembles something already flagged.
Experience teaches that regulations aren’t only about keeping people safe; they’re also shaped by economic competition and security. Sometimes, a brand new compound gets swept into broad restrictions before commercial value kicks in. This swing can interrupt research. Due diligence means checking the chemical’s status in both the shipping and destination country. It’s work that falls on scientists, legal teams, procurement folks, and sometimes customs brokers who have never seen such a name in their career.
Impact on Science and Innovation
Real progress slows if every novel molecule becomes a paperwork headache. It’s not just about regulatory burden — it also shapes who can keep pushing boundaries. Big companies and universities have compliance officers and lawyers. Small startups, universities in less wealthy countries, and individual researchers get hit hardest. Many simply give up, especially when faced with opaque customs processes or ambiguous requirements. Lost opportunities pile up.
Some countries treat even small samples of specialty chemicals as potential threats, pending weeks or more of extra paperwork. There’s no global standard. Sometimes what counts as a research reagent in one nation falls under “psychotropic substance” or “toxic precursor” in another. The end result? Progress, collaboration, and global discovery become hostage to paperwork.
Working Solutions: Transparency, Education, and Advocacy
Real solutions won’t come from yelling at regulators. A strong first step involves pooling knowledge: open databases of chemical regulations, case studies on shipping success and failure, and ongoing international scientific dialogue. I’ve heard of graduate students who had to learn customs law on their own just to get a single shipment approved. Better training and readily available guidance could lift a real weight off young scientists. Scientific societies and journals can play a role here. If the research community shares more about these bottlenecks, pressure mounts for policymakers to tune rules for both safety and progress.
Online tracking systems and digital submissions have started to cut delays. Some customs offices respond well to professionally written supporting documents explaining the compound’s purpose. A culture of clear communication between shippers, buyers, and authorities helps. It may not end the problem, but it keeps more options on the table.
In the end, advancing science means both discovery and the responsibility to move things lawfully. The risks remain real, but so do the costs of falling behind because ideas get stuck at the border. It’s not glamourous, but understanding this struggle can make the difference between scientific stagnation and breakthrough.