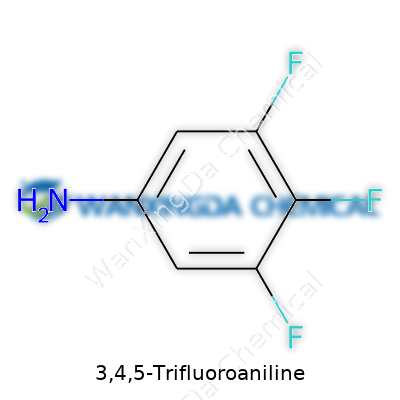
3,4,5-Trifluoroaniline: A Closer Look at Its Journey and Impact
Historical Development
Looking at 3,4,5-Trifluoroaniline, it’s easy to forget just how much quiet innovation can shape chemical history. Chemists first explored fluorinated anilines in search of new possibilities in pharmaceuticals and crop protection, always eager to tweak aromatic rings for unique effects. The push for new building blocks in organic synthesis lit the path toward trifluorinated derivatives. Once fluorination methods improved during the late 20th century, laboratories managed to produce these tightly tailored molecules more reliably, opening up a toolkit that’s still in heavy use today. Each small change—an extra fluorine atom here, a fresh catalyst there—expanded the potential for researchers to play with electronic effects, basicity, and metabolic stability. Now, even smaller specialty labs have the experience and equipment to make 3,4,5-Trifluoroaniline on a scale fit for both research and niche production runs.
Product Overview
3,4,5-Trifluoroaniline sits among a cluster of aromatic amines that draw attention from synthetic chemists. It stands out because three fluorine atoms line the benzene ring, shifting how it reacts and interacts compared to plain aniline. Incorporating this molecule into chemical routes has become common in efforts to design targeted pharmaceuticals, specialty agrochemicals, and some advanced materials. The demand for ever-more-precise chemical tools means trifluorinated aromatics like this become a hot topic as soon as new uses pop up in journals or patent filings.
Physical & Chemical Properties
In the lab, 3,4,5-Trifluoroaniline shows itself as a pale, often yellowish liquid. The trio of fluorines delivers a noticeable high density and moderate volatility, which brings both convenience and headache—it evaporates if left open, but a strong enough bottle keeps it contained. Odor can’t be ignored, though it’s less sharp than its parent aniline. The amine group gives this compound opportunities for further chemistry, but the fluorines sap some of the classic reactivity, making it more stubborn to transform. It dissolves easily in common organic solvents and stands up better to oxidation than non-fluorinated cousins. The boiling and melting points land in a familiar range for substituted anilines, but with the caveat that small differences have a big impact on isolation and purification steps, especially under vacuum or at scale.
Technical Specifications & Labeling
Walking through compliance in chemical supply, labeling matters. With 3,4,5-Trifluoroaniline, suppliers make sure to highlight its purity in percentages, batch numbers, and hazard information as required by global standards. Proper storage and handling instructions prevent headaches, given its fragrant nature and the risk of skin or eye irritation—chemicals without clear safety information can bite. The best suppliers use up-to-date certificates of analysis, offering transparency and peace of mind to research teams, since nobody wants an inconsistency in the starting batch to ruin days of work or introduce hidden contaminants into complex syntheses.
Preparation Method
Making 3,4,5-Trifluoroaniline usually starts from a trifluorinated nitrobenzene—or sometimes a substituted aniline with heavy protection steps. Chemists cut their teeth on reductions: using iron filings and hydrochloric acid, catalytic hydrogenation, or clever modern alternatives. As fluorine atoms resist many classic transformations, careful planning matters. The real trick lies in getting good regioselectivity during the initial trifluorination, since throwing fluorine around a benzene ring tends to make multiple isomers unless controlled by directing groups and temperature. Once the nitro group is in the right place, reliable reduction yields the target amine. It’s a process that feels simple on paper, but lab realities remind everyone just how tricky purification can get with similar fluorinated analogs peppering the product mixture.
Chemical Reactions & Modifications
Working with this molecule in the flask, the impact of fluorines stands out. The amine function survives mild to moderate acidic and basic conditions, letting chemists build on its core or swap protective groups as needed. Electrophilic aromatic substitution slows down markedly, though, as the electron-withdrawing power of three fluorines tames the benzene ring. Nucleophilic aromatic substitution remains on the table, especially at positions activated by the electron-poor character. When connecting to bigger molecules in drug or dye development, routine reactions like acylation or sulfonation work, but demand a patient hand to maximize yields. I’ve found that lighter fluorination gives you more flexibility, while the dense trifluoro setup makes predictability a prized trait—if you want precision, you know what you’ll get.
Synonyms & Product Names
If you ask around—or thumb through a catalogue—3,4,5-Trifluoroaniline shows up under a range of tags: “benzeneamine, 3,4,5-trifluoro-”, “1-amino-3,4,5-trifluorobenzene”, and perhaps a few abbreviated internal codes in larger organizations. The IUPAC name sets the tone in academic settings, but shorthand and trade names move quickly between industrial users, especially in high-throughput screening or development. All point back to the same distinctive fluorinated ring with a lone amine sidearm.
Safety & Operational Standards
Laboratories and plants don’t take aromatic amines lightly, and fluorinated versions aren’t an exception—though some safety margins improve, thanks to lower reactivity. Handling 3,4,5-Trifluoroaniline involves gloves, goggles, and a working fume hood. Inhalation and skin contact can lead to unpleasant symptoms, from irritation to more serious effects after prolonged exposure. Consistent practice means tightly sealing containers, keeping spill kits handy, and running regular checks on ventilation. Full documentation and staff training turn regulations from red tape into real risk reduction. In my own lab days, time spent on careful safety routines spared us trouble, even on long, hectic days running parallel reactions or packing columns.
Application Area
Route this chemical into early-stage pharmaceutical research, and it quickly earns its keep. Those three fluorines can block metabolic breakdown, making new leads stick around longer in animal tests. Agrochemical companies lean on it to design herbicides and pesticides that resist sunlight, water, and the relentless appetite of soil microbes. Outside life sciences, trifluoroanilines crop up in specialty dye chemistry and in certain high-performance materials where chemical stability or unique spectral properties count. Knowledge of trifluorinated aromatics helps synthetic chemists fine-tune their search for new and better bioactive compounds, while at the same time answering new regulatory and safety concerns about persistence in ecosystems.
Research & Development
Research into 3,4,5-Trifluoroaniline digs deep into both synthesis and application. Academic projects look for ways to install the trifluoro motif more efficiently—every time the yield ticks up or a catalyst gets cheaper, whole branches of research open up. Medicinal chemists want to know how swapping trifluoro for other groups shifts biological activity or reduces toxicity. Teams focused on sustainable chemistry keep hunting for less resource-intensive routes, maybe using electrochemistry or biocatalysis. Recent years have shown this molecule appearing in combinatorial drug candidate libraries or tagged for imaging studies, showing that curiosity and technical refinement go hand in hand.
Toxicity Research
Fluorinated aromatics ask for a careful eye on toxicity. Metabolism studies in rodents and cell cultures aim to reveal any quirks in how the human body or environment breaks down trifluorinated anilines. Raw data suggest lower acute toxicity compared to some unprotected anilines, yet breakdown products and chronic exposure can still pose concerns. That’s the flip side of chemical stability—substances that hang around can build up, so precision in application and ongoing surveillance of residue levels matter in fields like crop protection and hygiene products. Researchers rely on reliable animal models, in vitro testing, and environmental monitoring to spot trouble early before it reaches critical mass.
Future Prospects
Looking ahead, the story of 3,4,5-Trifluoroaniline seems set to keep evolving with broader adoption of 'design-by-property' in drug and material development. Cheaper or greener synthesis will expand its reach outside well-funded pharma or agrochemical giants. The combination of stability, fine-tuned reactivity, and impact on metabolic fate makes it a recurring figure in chemical research roadmaps for the coming decade. Regulation may shape the market too, especially with growing concern about environmental persistence. More comprehensive toxicity, environmental fate, and alternatives assessment could reshape priorities. But in the lab or on the product line, this compact and unassuming molecule continues to deliver both opportunities and challenges—fueling the creativity and caution that push chemical science forward.
Every time we talk about chemicals and their impact, the big names always come up—paracetamol for a headache, aspirin for your heart, glyphosate for your weeds. Rarely do we hear about unsung molecules like 3,4,5-trifluoroaniline. This simple-looking compound pops up as a small player, but it touches several corners of innovation, especially in pharmaceuticals, agrochemicals, and advanced materials.
Why This Molecule Draws Attention
In my years following chemistry trends, smaller aromatic amines with tricky substitutions—like multiple fluorines—catch the eye of scientists for a key reason: the fluorine atoms. Fluorine changes the way a molecule behaves in the body or in the environment. It helps pharmaceuticals last longer in our system, helps pesticides break down slower on crops, and even boosts materials in electronics.
3,4,5-trifluoroaniline shows up as a building block for molecules that demand resilience and precision. Because fluorine changes polarity and metabolic stability, researchers use substances like this one to tweak a drug’s performance, making it more useful to patients or more selective as a crop treatment.
From Lab Bench to Product Shelf
Pharmaceutical chemists use this compound to stitch together larger, more complex drugs. The three fluorines positioned on the benzene ring mean you can attach, modify, or elongate chains in a very selective way. This sort of controlled chemistry lets companies create drugs that resist breakdown by enzymes, promising longer-lasting effects in the human body.
Beyond pills, 3,4,5-trifluoroaniline’s backbone slips into the world of crop protection and dyes. My experience watching crop science team meetings reminds me how vital precision is for new insecticides or fungicides. Minor changes in a chemical’s shape can double or halve its power, and the trifluoroaniline group is one tool for fine-tuning activity. On the colorants side, those same fluorines add brightness and stability to dyes, especially in textiles or specialty coatings.
Opportunities and Challenges
Many chemical innovations come with baggage. Once you’ve added multiple fluorines to a compound, you often make it harder to break apart. Environmental chemists keep a close eye on these substances, since some fluorinated chemicals last in soils or waterways for years. So, the opportunities for better medicines and smarter pesticides always come with more responsibility. In my view, anyone working with this class of compound has to think two steps ahead—wondering not just about performance, but about what happens after the job is done.
To manage that responsibility, there’s a growing movement looking at green chemistry and end-of-life breakdown. Research now leans toward designing molecules with possible biodegradation in mind. Maybe, using 3,4,5-trifluoroaniline as part of a bigger, purposely degradable scaffold makes sense. If companies align new discoveries with responsible breakdown, we’ll see fewer headlines about chemical persistence.
The Path Forward
This molecule won’t headline news anytime soon. Still, it carries weight behind the scenes in research labs and new product pipelines. Using it wisely, with careful thought around both innovation and environmental impact, means balancing progress with caution. I’ve seen how easily details get lost in the excitement for new molecules, but steady attention to both benefit and risk ought to guide choices in labs and boardrooms alike.
Understanding the Nuts and Bolts
Anyone who has spent time in a chemistry lab knows there are plenty of compounds with fancy names and a knack for raising eyebrows. 3,4,5-Trifluoroaniline is one of those molecules that grabs a chemist’s attention, not because it’s fancy, but because of what its structure and formula can tell us. Looking at its chemical formula, it reads as C6H4F3N. That boils down to a benzene ring holding on to three fluorine atoms at the 3, 4, and 5 positions, with an amino group tagging along for the ride. Add up the weights from each atom—carbon, hydrogen, fluorine, nitrogen—and you hit a molecular weight of around 163.1 grams per mole. It’s straightforward enough, though I’ve seen more than a few students stumble trying to balance fluorines and hydrogens without double-checking the ring positions.
Why Knowing the Details Isn’t Just for Chemists
Diving into chemical formulas or molecular weights can sound like a slog meant only for professionals. But knowing what goes into a molecule like 3,4,5-Trifluoroaniline pays off in real-world situations. Fluorinated aromatics, for example, show up in the hunt for new drugs and in the design of materials way tougher than anything you’d find at your local store. Fluorine swaps often give new flavors to medicine by making the molecule more stable or messing with how the body processes it. Staying sharp about chemical details has clear payoffs—new treatments, faster diagnostics, smarter materials.
What Makes Fluorinated Rings Special?
There’s a reason researchers keep circling back to compounds loaded with fluorine. It isn’t just about making the name longer. Adding fluorine atoms changes how a molecule behaves—sometimes making it more resistant to breaking down, other times letting it slip past enzymes that would chew up ordinary anilines. This holds a lot of weight in pharmaceuticals, where resilience against metabolism means a pill works better or lasts longer. It’s no accident that many blockbuster drugs on pharmacy shelves hide a fluorinated ring down in their blueprints.
Keeping It Safe and Smart
Getting too cozy with compounds loaded with halogens, including those with fluorine, calls for a bit of caution. Handling 3,4,5-Trifluoroaniline, or anything with a similar punch, means understanding both its strengths and risks. Industrial researchers, for instance, always track how such compounds interact with the environment or living tissue. A molecule’s mass or formula can hint at volatility, toxicity, and how it might travel once it leaves the flask. Mistakes in these calculations cost both money and safety—two things no lab wants to gamble on. In my early career, I learned this lesson the hard way, watching a senior chemist catch a subtle error in a mass calculation before it snowballed into a hazardous waste problem.
Staying Ahead with Careful Work
Solid information and careful calculations don’t just protect labs—they help everyone from researchers to regulators make better choices. Reliable sources, good training, and attention to detail make a difference. Each piece of data, whether it’s a molecular weight or a formula, serves as a small piece of a bigger puzzle, especially as chemists and manufacturers look for molecules that meet tomorrow’s challenges. Taking shortcuts seldom leads to smart results. Building a tradition of accuracy, fact-checking, and healthy skepticism isn’t just about following rules—it pays off for innovation, safety, and trust down the line.
Understanding Real Dangers in the Lab
Anyone working with specialty chemicals like 3,4,5-Trifluoroaniline should start with a healthy respect for the hazards on the label. My years in research and production labs taught me the hard lesson that the invisible risks often create the biggest problems for workers who get casual about basics. 3,4,5-Trifluoroaniline isn’t on people’s radar like strong acids or cyanide, but it deserves careful attention. The chemical acts as a skin irritant, and can be harmful through both inhalation and contact. The same way you wouldn’t mess around with bleach or solvents without gloves, you shouldn’t ditch goggles, gloves, and chemical-resistant coats here either.
Protected by Habit and Preparation
Spray bottles, beakers, and pipettes never go far in a lab without proper safety gear. Nobody wants to develop a cough or rash because a few drops landed on exposed skin or vapor slipped past a surgical mask. Proper use of a fume hood took on new importance in my early days, after colleagues got headaches because someone decided to “just do a small transfer at the bench.” Aniline derivatives, including this one, can produce dangerous fumes. Good ventilation serves more than dust control; it’s the difference between a routine day and a trip to occupational health.
Unseen Risks: Spills and Waste Handling
Most accidents happen not while pouring from the bottle, but when cleaning up forgotten spills or disposing of leftovers. Trifluoroaniline shares the risks of its close relatives, showing both acute and chronic toxicity. Repeated low-level exposure adds up. I learned to double-bag waste and label it clearly for the disposal team. Dedicated chemical waste bins make sure that nothing mixed down a drain ends up in groundwater, and that disposal crews can spot hazardous waste at a glance. It’s not just about following the rules; it’s about protecting everyone along the way.
Data-Driven Practices
Facts ground our actions. According to research on similar aromatic amines, chronic exposure may link to organ toxicity and other systemic effects. Risk increases with repeated contact—something easy to avoid with well-fitting nitrile gloves and long sleeves. Industry standards point to fume hoods, and for a reason: levels in the air must stay below safety limits, which real-world monitoring can confirm. OSHA, NIOSH, and the European Chemicals Agency track incidents with these chemicals, providing real-world stories of what goes wrong when shortcuts become the norm.
The Path Forward: Culture of Safety
Handling 3,4,5-Trifluoroaniline safely never rests on a single person. Training, clear signage, regular risk reviews, and easy access to material safety data sheets encourage habits that stick. My workplace improved dramatically with weekly tool-box talks that turned near-misses into shared lessons. No one likes the hassle of double-checking labels, swapping out torn gloves, or logging waste, but routine focus saved us from medical incidents more than once.
Instead of thinking of chemical safety as a box-checking formality, I see it as peer protection. By treating every handling of 3,4,5-Trifluoroaniline as a team effort, we protect health and careers, and keep lab work productive and safe. Directly facing the risks and building smart habits benefits everyone, long after the vials are packed away.
3,4,5-Trifluoroaniline in the Real World
Some chemicals play hard to get, especially when you try dropping them into water. 3,4,5-Trifluoroaniline is one of those that doesn't like to mingle with water molecules. Chemists know this routine: add a pinch of an aromatic, fluorinated aniline to water and watch as it refuses to mix. Water works best with things that can form hydrogen bonds or that carry a charge. Here, the trifluorinated ring and that shy amine group combine to make a stubborn, mostly nonpolar character. Its trifluoro groups repel water even more than an unmodified aniline would.
Why Solubility Matters
Solubility is not just academic trivia. It decides how a compound gets purified, processed, and even cleaned up in lab spills. I've spent hours scratching my head, trying to pick the right solvent for stubborn organic compounds at work. With 3,4,5-Trifluoroaniline, you quickly see that water doesn’t stand a chance. That means, if you want to handle or use this stuff—for synthesis or analysis—organics like ethanol, ether, or chloroform become the main tools in your kit. Anyone who has dried out a reaction mixture will tell you that fighting poor solubility wastes time and chemicals fast.
No Surprises from Chemical Structure
Organic chemists often glance at the structure and predict solubility in seconds. For 3,4,5-Trifluoroaniline, three fluorines line up on the aromatic ring, making the whole thing less eager to interact with water. Sure, there’s an amino group, but it can’t overrule the influence of those electron-grabbing fluorines. Aromatics like this gravitate toward organic solvents. That's chemistry’s street smarts—like oil mixing with oil.
Workarounds from the Lab Bench
Folk wisdom in the lab says: pick the right tool for the job. Here, organic solvents play matchmaker. Chloroform, dichloromethane, and even ethyl acetate often bring these stubborn compounds into solution. Sometimes it takes a mixture—add a splash of methanol to a mostly organic bath, and the compound gives in. These are tricks picked up from years tracking down elusive yields in glass flasks.
Solubility and Safety Go Hand in Hand
Poor water solubility has real consequences outside the test tube. Waste disposal becomes tricky, as you can’t flush these chemicals down the drain. Environmental chemistry teaches us: fluorinated aromatics linger in nature. The wrong waste method, and you risk contamination. The solution takes more than just a swap of solvent—it means chemists owe attention to waste routes, using activated carbon, or advanced oxidation when needed.
Looking Forward with 3,4,5-Trifluoroaniline
The world of specialty chemicals brings ever more fluorinated organics, each one with quirks. Chemists could push for greener solvents—less toxic, less persistent in the environment—when dissolving compounds like this. It often comes down to habit: reach for a halogenated solvent, or look for alternatives before starting a synthesis. Investing in solvent recycling and safer practices saves time and trouble in the long run.
In my time running reactions and troubleshooting solubility, I’ve learned that paying real attention to these patterns makes chemistry less frustrating. It keeps the process efficient, safe, and responsible. With compounds like 3,4,5-Trifluoroaniline, experience matters as much as textbooks.
Treating Specialty Chemicals with Respect
Having worked with specialty aromatic amines, I learned fast: some compounds react to the slightest change. 3,4,5-Trifluoroaniline ranks among those that want careful handling, especially once you’ve invested money and effort to source or synthesize it. Chemical stability here doesn’t just save dollars—it safeguards people’s health and delivers reliable results in the next synthesis step.
Don’t Let Light or Air Ruin Your Stock
From my graduate school days, I remember the tinge of disappointment spotting colored streaks in what should have been a clear sample of aniline derivatives. Many aromatic amines, especially substituted ones, darken over time if sunlight finds its way into the bottle. 3,4,5-Trifluoroaniline can behave the same way—ultraviolet exposure accelerates degradation, changing chemical makeup and trashing purity.
Storing bottles away from windows, always reaching for amber glass rather than clear, blocks most of that damaging light. Sealing the container tightly after each use, purging the headspace with a bit of nitrogen when possible, keeps both air and moisture out, both of which threaten stability. Oxygen sneaks in and slowly oxidizes sensitive groups, and you end up with a layer of unpleasant byproducts or sludge.
Temperature: The Forgotten Culprit
Most labs install a chemical fridge for a reason—heat speeds up just about every reaction you don’t want. If a sample sits all summer in a warm stockroom, don’t count on finding the same molecule next fall. Lowering the storage temperature slows down decay and oxidation, so bench-top storage rarely earns the chemist’s trust.
I’ve seen companies invest in temperature monitors for their solvent stockrooms, not just for safety, but to protect expensive organics like 3,4,5-Trifluoroaniline. Regular fridge temperatures work fine if you rebottle and warm the chemical to room temperature before opening, avoiding condensation inside the bottle.
Chemical Compatibility Cautions
No chemical deserves a handwave on compatibility, and fluoroanilines push the point. I once watched someone store their amine stocks near strong acids—only for cross-contamination and stray vapors to start a chain reaction. 3,4,5-Trifluoroaniline demands segregation, away from anything oxidizing or strongly acidic. These friends-in-theory convert it into byproducts or, in worst cases, leak dangerous fumes. Good practice spaces incompatible chemicals apart, labels clearly, and reserves specialty cabinets for tricky cases.
Stability Doesn’t Rely on Chance
All chemists run up against the cost of improper storage: lost money, wasted time, questionable data. Keeping 3,4,5-Trifluoroaniline stable builds from simple, consistent habits—store in an amber glass container, keep it sealed from air, chill below room temperature, mark dates and keep incompatible materials out of reach. Lab personnel benefit from regular checks and refreshers; knowing the signs of decomposition keeps surprises at bay.
Much of this draws from direct experience: I’ve seen even small lapses create big headaches. Treating 3,4,5-Trifluoroaniline like a fleeting investment pays off every time a clean, reliable product shows up when needed. Good storage is not just a procedure; it’s respect for the compound’s quirks and the people relying on it.