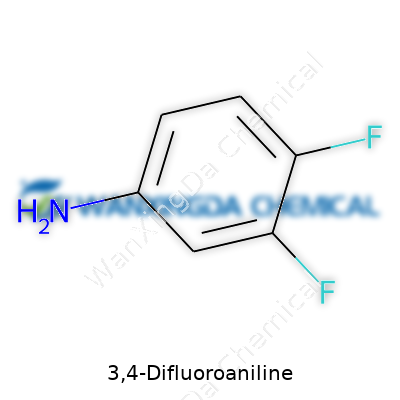
3,4-Difluoroaniline: Beyond the Basics
Looking Back: The Path of 3,4-Difluoroaniline
Chemistry has always pushed people to look for ways to tweak molecules, searching for new functions or stronger materials. In the world of aniline derivatives, 3,4-Difluoroaniline came along as researchers probed what happens when you slip in fluorine atoms right next to one another on the aromatic ring. The first reports noted its intriguing behavior, catching the eye of both academic and industrial labs eager to make new pharmaceuticals, agrochemicals, and specialty polymers. Growing demand for more potent fluorinated compounds in the late 20th century drove attention to this compound, sparking more research into its preparation and properties. From the start, 3,4-Difluoroaniline offered a balance between versatility and reactivity — two traits that continually fuel its relevance.
What 3,4-Difluoroaniline Brings to the Table
You can spot 3,4-Difluoroaniline by its distinct structure: an aniline skeleton with fluorine sitting on the third and fourth carbon. That pair of tightly bonded fluorine atoms influences how the molecule reacts compared to standard aniline. Its use as an intermediate in producing pharmaceuticals and crop protection products stands out. Chemists often look for ways to add fluorine to aromatic rings, chasing improved metabolic stability or bioactivity; 3,4-Difluoroaniline fits that need neatly, shaving time and steps from synthesis when you want those fluorines in the right spot from day one.
Physical and Chemical Profile
Unassuming at first glance, 3,4-Difluoroaniline usually appears as a colorless to pale yellow liquid. Fluorine atoms contribute to its greater stability compared to unsubstituted aniline, showing moderate water solubility but dissolving readily in most organic solvents. Fairly high purity standards typically apply — even small amounts of impurities can throw off later reactions, especially in pharmaceutical manufacturing. Unlike some anilines that oxidize or degrade quickly, this one holds up under many reaction conditions, with the double fluorine boost on the benzene ring giving it extra resistance to breakdown.
Technical Specifications & Labeling: Getting Precise
Working with 3,4-Difluoroaniline demands attention to detail. Chemical suppliers focus on metrics like purity percentage (typically over 98 percent for synthetic work), moisture content, and impurity profile. Accurate labeling isn't just regulatory box-ticking; it's a legal and safety shield, ensuring that the right molecule, in the right strength, reaches labs or factories. In my experience, a mismatch in labeling can derail an entire series of experiments, especially because trace differences matter in catalysis or formulation chemistry. Differences between technical and pharmaceutical grades carry big implications downstream, so clarity in communication is as vital as the chemical itself.
How Chemists Make 3,4-Difluoroaniline
There's more than one route to 3,4-Difluoroaniline, but most methods start with a difluorinated nitrobenzene. Reduction of 3,4-difluoronitrobenzene, often using catalytic hydrogenation or methods like iron-acid reduction, brings down the nitro group to an amine. Some labs push greener alternatives, like catalytic transfer hydrogenation, which trims hazardous waste compared to classic metal reductions. Sourcing pure starting materials and controlling the reaction environment become critical—any slip risks producing side products or lower yields. Cutting-edge research tries to trim steps and cut energy costs, but the key steps—fluorination, nitration, reduction—reflect the chemical’s fundamentally robust design.
Reactions & Transformations: A Platform for Synthesis
Aromatic amines have always been playgrounds for inventive chemistry, and 3,4-Difluoroaniline proves no exception. That amine group lends itself to acylation, sulfonation, and diazotization, opening up further modifications to reach dyes, pigments, and advanced pharmaceuticals. The ortho and para fluorine positions not only block certain substitutions but also steer reactivity, letting chemists shape complex molecules with precision. Modern cross-coupling techniques — think Suzuki or Buchwald-Hartwig reactions — harness the unique electronics of the difluoroaniline core to knit together new carbon-nitrogen or carbon-carbon bonds. These tools keep chemists coming back, turning what began as a curiosity into a workhorse of aromatic synthesis.
Synonyms & Names in the Literature
People refer to this compound with several names, sometimes as 1-amino-3,4-difluorobenzene, or 3,4-difluorophenylamine. In patent filings and older texts, you’ll see variations like NSC 105938. In my own reading, a search with these alternate terms catches a wider net of references, so casting a broad terminology net pays off during research or market analysis.
Health, Safety, and How to Handle It
Using 3,4-Difluoroaniline safely boils down to the fundamentals: glove use, eye protection, proper ventilation, and storage away from oxidizers. Like others in the aromatic amine family, it won’t do well if inhaled or splashed on skin, carrying risks from acute irritation to longer-term effects with repeated exposure. Regulations in chemical plants focus tightly on safe transport and waste procedures. My own regret in early days was skipping a fume hood once — the strong, pungent odor lingers and reminds you how just a momentary lapse can bring trouble. With proper care, though, risks are manageable, and new research aims to design safer processes and containers that limit spills or leaks.
Where 3,4-Difluoroaniline Plays a Role
Modern pharmaceutical labs prize 3,4-Difluoroaniline as a building block for active drug molecules, especially those chasing fluorinated scaffolds for improved performance. Crop protection products lean on it for the same reasons: added stability, sharper selectivity, better uptake by plants, or resistance to metabolic breakdown. Electronics and specialty polymers also tap into its chemistry — those fluorine groups tune the electrical properties and extend material lifespans. The world keeps demanding more finely tuned molecules to meet tighter specifications, so the value of pre-fluorinated anilines only grows.
On the Cutting Edge: Ongoing Research
Labs worldwide keep hunting for faster, cleaner ways to prepare and use 3,4-Difluoroaniline. Green chemistry sits front and center, with group after group testing milder reducing agents or recyclable catalysts. There’s a surge in exploring what fluorine addition brings to new medicinal compounds, as subtle tweaks can mean the difference between a promising lead and a transformative therapy. My own connections in academia talk about the challenge of introduction: how to graft additional useful groups onto the difluoro ring without blowing up yields or inviting safety hazards. Research on catalytic processes and reaction streamlining could lower costs and environmental impact.
Toxicity Studies: Looking Out for Risks
As with many aromatic amines, 3,4-Difluoroaniline isn't something to take lightly. Toxicologists pay careful attention to its effects on living cells, studying both short-term and chronic exposures. The double fluorine substitution can make breakdown products harder for the body to clear, which means regulators watch for any sign of bioaccumulation or mutagenicity. Some studies point to manageable risks with controlled laboratory handling, but the push for more data never ends—modern regulations demand clearer evidence of safety across all stages, from production to after-use environmental release.
The Road Ahead: What’s Next for 3,4-Difluoroaniline
Demand for fluorinated building blocks like 3,4-Difluoroaniline is unlikely to slow down. New therapeutic frontiers in drug design focus more on unique molecular features, often relying on pre-made fluorinated anilines to shorten development timelines. Advances in sustainable chemistry could cut the resource footprint further, addressing growing pressure to “green” the entire supply chain. What matters most is balancing innovation with responsibility—whether it’s safer manufacturing methods, more comprehensive risk profiles, or creating less hazardous derivatives. Real progress will center on smarter chemistry that doesn’t just serve industry but also respects health and the planet, building on the deep experience and careful work of researchers past and present.
Many people never hear about 3,4-Difluoroaniline outside a science lab, but this compound ends up in more corners of daily life than expected. Seeing complicated chemical names can feel a bit intimidating, but this one deserves attention because of what ends up getting built from it. Production floors and research labs quietly rely on specialty chemicals like this to move technology and medicine forward.
Chemistry Building Blocks Make More Than Just Chemicals
3,4-Difluoroaniline works like a tiny construction piece in the chemical world. Chemists use it mostly to craft other valuable molecules—a process known as synthesis—especially in fields like pharmaceuticals and agricultural chemicals. I’ve spent late nights in labs where everyone’s chasing a blueprint for a new medicine, and amines like this one serve as a starting point. These molecules get combined or swapped with others, giving rise to active ingredients in drugs designed to help people cope with cancer, infections, and chronic illnesses.
Companies making crop protection chemicals favor this compound because the fluorine atoms on the ring can help create molecules that stand strong against breakdown in the field. Farmers look for weed and pest control solutions that won’t wash away with rain or lose strength in the sun. The designers behind these solutions lean on sources like 3,4-Difluoroaniline to add a chemical backbone that sticks around and works longer—directly translating to less waste and lower application costs.
Medicine, But Only After Careful Checking
Drug discovery often looks like a puzzle, fitting together molecular pieces in different shapes until you find something that locks in and works. Fluorinated anilines—including the 3,4-difluoro version—get picked because they can make drug molecules both tougher and more likely to do their job in the body. Fluorine can block enzymes from chewing up medicine too quickly, which means patients get steadier benefits from the same dose. Reports show a big share of top-selling small-molecule drugs scatter fluorine atoms somewhere in their structure for exactly this reason.
Safety always stands front and center. Any intermediate molecule, especially one with a structure as active as 3,4-Difluoroaniline, demands strict handling. Toxicity isn’t just a regulatory box to check off; I’ve seen seasoned professionals in pharma stress over exposure limits, knowing the importance of a safe lab environment. Smart, up-to-date manufacturing processes and proper industrial hygiene stop this compound from causing harm outside its intended use.
The Road to Greener Chemistry
It’s easy to take for granted how invisible ingredients influence end products. Still, reliance on high-impact intermediates brings environmental questions. Fluorinated chemicals can linger in soil and water, and stricter rules have arrived for production and disposal. Chemists are actively investigating greener ways to make these key building blocks. I remember following a research project focused on catalytic reactions that use less hazardous reagents—real change isn’t easy, but the industry has every incentive to innovate to meet new sustainability targets. Sourcing, process improvements, and responsible use all play their part.
In the end, 3,4-Difluoroaniline plays its part quietly, but its influence stretches from hospital bedsides to the food on our tables. Understanding where and why such chemicals fit means more informed decisions and a better shot at safer, more sustainable progress.
Molecular Details That Shape Its Role
The molecule 3,4-Difluoroaniline isn’t just another name in an organic chemistry textbook. People who study chemicals for a living, especially those who work in pharmaceuticals or materials science, care about this compound’s details. Its molecular formula, C6H5F2N, shrinks down a world of complexity into a compact string of letters and numbers. For research teams exploring new antibiotics or electronics materials, these details matter more than one might think. It’s not enough to get close. The tiny changes in atoms and bonds right here often change how a molecule behaves, how it bonds, and what new products it can help us build.
The molecular weight lands at 129.11 grams per mole. Being able to calculate molecular weight helps scientists portion out exactly what a reaction needs. No one wants to waste reagents, and no one wants to misjudge the dose or safety data. That’s part of the discipline learned in every undergraduate chemistry course — and it becomes the cornerstone in research labs that count every gram.
Why Details Like This Stick With You
My own experience working with aromatic amines comes back every time I see a compound like 3,4-Difluoroaniline. Once, we ran a series of reactions aiming to make dyes for specialized fibers. The difference between a plain aniline molecule and a difluorinated one felt subtle on the page, but the colors, solubility, and even safety precautions changed. Every researcher in the room who overlooked a molecular detail ended up with unexpected results, or worse, a ruined experiment. That stays with you.
The two fluorine atoms in 3,4-Difluoroaniline don’t just shift the molecule’s properties a little. Fluorine changes electron distribution, which changes how the molecule reacts. That’s a goldmine for anyone designing molecules meant to resist breakdown in the body, or to cling to a substrate in industrial coatings. Protective equipment policies also get stricter because adding halogens can make these molecules more reactive in air, water, or with metals. Every bit of extra information helps people make safer decisions.
Supporting Responsible Progress
Chemists rely on trusted sources for these compound details. The molecular formula and weight aren’t guesses — agencies and peer-reviewed databases carefully check the numbers before they reach industry or academia. Mistakes in molecular weights don’t just lead to calculation errors; worse, they ripple out, causing issues in published research, wasted grants, or even faulty manufacturing. I’ve seen the confusion a single transcription error can bring to a group’s workflow. Places like PubChem and Reaxys pour time and expertise into keeping their databases accurate. People in the chemical industry know the cost of slipping standards.
To strengthen trust, research and industry groups could keep pushing for open, regularly reviewed databases. New chemists benefit from clear, accessible data, so mistakes become less likely. Science thrives on sharing facts, not keeping them behind paywalls or out of date PDFs.
Building Better Entire Fields, One Formula at a Time
Getting the formula and weight of 3,4-Difluoroaniline right isn’t about trivia. It’s about enabling innovation safely, keeping conversations consistent from classroom to boardroom, and protecting researchers. In chemistry, small details like a molecular formula can shape big outcomes. That’s a lesson drawn from lab benches, trade publications, and plenty of trial and error. Scientific work builds on details just like these, so accuracy should never feel optional.
Plenty of folks find themselves working with chemicals in labs, factories, or during research. 3,4-Difluoroaniline shows up more and more these days, especially in the pharmaceutical and agrochemical industries. Handling it safely isn’t just a checklist item; it’s about making sure everyone goes home healthy and whole at the end of the day.
Why 3,4-Difluoroaniline Catches Attention
3,4-Difluoroaniline gets used as a building block in chemical synthesis. It can help create medicines, pesticides, dyes, and other specialty products. I’ve watched folks rush into handling exotic chemicals thinking the danger only belongs to the stuff that hisses or glows. The truth is, sometimes a clear, simple-looking liquid packs a punch, and 3,4-Difluoroaniline fits that bill.
Direct Exposure, Direct Consequences
Let’s talk skin. Getting this on your hands or face might not burn right away, but that’s not a sign you’re in the clear. Some chemicals take their time. Prolonged or repeated skin contact can irritate or cause allergic reactions. Breathing fumes or mist, especially without proper ventilation, may also spell trouble for your respiratory system. I’ve seen noses and throats get raw after a short stint working without a mask or enough airflow.
This isn’t about abstract risk — records from companies and government agencies show workers develop health issues when they skip basic precautions. The American Conference of Governmental Industrial Hygienists flags many aromatic amines as suspect for health hazards, urging limits on exposure.
Smart Handling Starts with Paying Attention
I always wear proper gloves, long sleeves, and goggles handling these kinds of chemicals. Nitrile gloves work better than latex in this case. Closed-toe shoes — no exceptions. Even if you’re careful, spills happen. Clean up right away with absorbent materials, and ventilate the area. Never touch your face or phone until you remove your gloves and wash your hands well. Small habits make a big difference.
Ventilation matters too. People forget that fumes drift, and even a faint smell in the air means you’re breathing in something that doesn’t belong. Fume hoods or local exhaust fans pull vapors away from your breathing zone. If you’ve ever left a work area with a headache or sore throat, that’s a sign the air needs improvement.
Dealing with Spills and Waste
Clean-up plans should be clear to everyone who works there, not buried in a binder. Having spill kits handy, not locked away in a closet, speeds up response. I’ve found that regular drills help people act automatically instead of freezing when something spills. Waste needs proper storage in labeled, tightly sealed containers. Unmarked bottles left on a bench have no place in any safe workplace.
The Right Culture Reduces Mistakes
People make mistakes — that’s life. The safest labs and plants don’t ignore that truth. Instead, they foster environments where staff look out for each other. Reporting near-misses instead of hiding them helps everyone spot and fix weak points in procedures. Supervisors who lead by example encourage the same from their teams.
No single rule or piece of equipment gives perfect safety. Consistent habits, clear communication, and respect for the risks make the real difference. 3,4-Difluoroaniline won’t give you a second chance if you ignore what it can do. Listening to experience and sticking to proven precautions builds a workplace where accidents remain rare, and everyone can keep doing their job year after year.
Thinking Beyond Labels and Data Sheets
Working with 3,4-Difluoroaniline reminds me of long days in the lab, sorting out which bottles felt safe enough to handle with bare hands and which ones deserved the thicker gloves. The molecule, a useful building block in organic synthesis, isn't something you just prop on a shelf and forget. Its potential toxicity sits at the back of my mind, not to stir fear but to encourage respect.
Safety Over Simplicity
Personal experience in research has shown me that even mild aromatic amines like this one release vapors with surprising stubbornness. Once, after opening a bottle in an unventilated space, I found the smell clinging to my lab coat for days. Though no disaster followed, it taught me how these chemicals don’t give second chances if handled sloppily.
Ideally, one would tuck such chemicals in a cool, dry spot. But “cool and dry” means different things depending on context. In my own practice, I favored a flame-resistant safety cabinet specifically marked for organics. Not every workplace invests in these cabinets, which leaves some folks improvising—usually not for the better. Regular cabinets let fumes drift up and over into other labs or mingle with incompatible substances.
Understanding the Risks
3,4-Difluoroaniline is no kitchen cleaner. Skin absorption, vapor inhalation, and even slight leaks can set off headaches, nausea, or much worse. Reports show that aromatic amines have carcinogenic potential—something widely backed by occupational safety literature and public health agencies. Responsible handling means never assuming a closed bottle truly seals in all risk. I’ve seen tape around lids, bottles double-bagged, even a cling-wrap layer, all in the name of peace of mind, and sometimes out of necessity when storage resources ran thin.
Some labs invest in explosion-proof refrigerators to slow down the chemical degradation and lessen volatility. Unfortunately, I’ve also worked places where bottles sat behind a regular fridge, keeping local techs in the dark about the actual risks. Such shortcuts invite disaster—accidents rarely strike loud or sudden; they sneak in through routine.
Daily Discipline Matters More Than Rules
Labeling stands as simple advice, but it’s where most people trip up. In a rush, someone scribbles a fading note instead of a full name and hazard code, then forgets entirely about the original contents. In my own workflow, relabeling was a weekly ritual. Clear, legible warnings do a lot more good than complicated barrier technologies when the rush is on or the lights go out during a storm.
Some colleagues push for chemical tracking software in university settings, others trust handwritten logs. Both methods work if treated seriously. Good tracking warns when a chemical approaches its shelf life, guides safe disposal, and keeps an eye on inventory size. Unused or expired 3,4-Difluoroaniline piles up quickly if no one takes responsibility, and that’s when unnoticed leaks or forgotten bottles turn into emergencies.
Bridging Knowledge and Action
Teaching storage safety doesn’t end with handing out an SDS. It takes mentorship, habit-forming, and a willingness to swap horror stories honestly. Keeping 3,4-Difluoroaniline under lock and key only works if staff know why the lock matters. The most reliable method I’ve found is open conversation—sharing mistakes, insisting on rechecking labels, even role-playing emergencies during training sessions. No checklist or cabinet beats a workplace where people look out for one another’s safety every day.
3,4-Difluoroaniline pops up often in synthetic chemistry and pharmaceutical research. Folks working in research labs, API development, and even in agrochemical testing come across it when they dig into complicated organic schemes. But the question about purity isn’t just a technical detail—it shapes everything from yield quality to safety procedures.
Purity: More Than a Label
There’s a big difference between grabbing a bottle marked “lab grade” and one with a label boasting 99% or higher purity. In research, unexpected contaminants can mess up a synthetic route or cloud the results of a chemical reaction. For pharmaceuticals, impurities sometimes lead to regulatory headaches or safety concerns down the line.
I’ve sat in group meetings where a promising molecule failed scale-up work. After digging, we found impurities in our starting material tripped up our process. Looking back, we should have checked purity specs before committing to costly experiments. Many chemists nod knowingly at stories like this—one off-spec batch can waste days of effort.
Why Grades Matter: Impact on Work Downstream
Buyers looking at 3,4-difluoroaniline find more than just a price difference between grades; they discover practical trade-offs. Redistilled, analytical, or pharmaceutical grades address different needs. Research teams want confidence their reactions won’t carry hidden baggage. Process chemists depend on tight specs to keep batch reproducibility high.
Companies usually offer certificates of analysis. Still, not every supplier will put sample analysis up front. Sometimes, only vague purity ranges show up online, until you press for the paperwork. Anyone aiming for regulatory approval or high-end R&D has to push for clear data.
The Problem with Chasing Ultra-High Purity
Pursuing high-purity reagents brings its own headaches. Purification costs climb, lead times stretch out, and some small-batch suppliers don’t stock niche grades regularly. Prices can spike unexpectedly. If the project demands a common grade, switching to ultra-pure just hits the budget, with little benefit for routine synthesis.
There’s temptation to over-specify—especially when regulatory teams get involved. My own experience says that’s often a mistake. Matching grade to practical need saves both time and cash. Chasing “the best” for ordinary benchwork doesn’t guarantee better science. It just creates a paperwork trail and wipes out supply chain flexibility.
Solutions and Smarter Choices
Labs can save themselves grief by building long-term relationships with suppliers—this helps get batch-specific data without jumping through hoops. Sharing feedback with suppliers when impurities pop up has real value; they’re more likely to tighten their controls if customers demand it. Buying smarter starts with training junior scientists to scan certificates of analysis instead of trusting a standard catalog description.
Open communication helps too. If an organization runs into issues tied to raw material quality, passing notes across different research groups limits repeat mistakes. In-house testing, even spot checks, pays off for higher-stakes work. Relying only on supplier claims can lead to costly surprises.
Bringing It Together
Picking the right grade of 3,4-difluoroaniline rarely feels glamorous, but it plays a quiet role in scientific progress and safe manufacturing. Purity grades matter because the work downstream relies on them. Choosing thoughtfully, asking direct questions, and building supplier trust end up more important than just chasing a higher number on a spec sheet.