3,4-Dihydroxybenzoic Acid: A Ground-Level Look At A Cornerstone Compound
Historical Development
Curiosity about the world often drives science further than any deliberate search for profit. 3,4-Dihydroxybenzoic acid owes a lot to that spirit. Back in the mid-19th century, chemists explored the subtle changes made to benzoic acid by adding hydroxyl groups, hoping to find new flavors, medicines, or dyes. Work like that set the scene for later studies into catechols and similar molecules. The molecule popped up in plant extraction processes and slowly drew more attention for its antioxidant potential. In the early days, no one guessed that this compound would wind up circulating through modern research on heart health, skin protection, and even cancer prevention. Often, residues from plants or natural fermentation gave up this acid before laboratories took over with more refined methods, showing how the old and new worlds of chemistry often meet in one flask or another.
Product Overview
Today, chemists know 3,4-dihydroxybenzoic acid as protocatechuic acid. It lands squarely in the category of phenolic acids. Plants brew up a batch of this molecule during their daily grind of defending themselves from pests, drought, and stress, which explains why it shows up in everything from olives to green tea. Over the years, the food supplement market has noticed these natural antioxidants. As attention grows around plant-based diets, this molecule continues to make waves as both a supplement and a research tool. Whether it comes pure from chemical synthesis or gently coaxed out of plant materials, it keeps turning up for its protective properties, energy metabolism impact, and hints it may slow down aging in our cells.
Physical & Chemical Properties
No one forgets the sharp contrast between a white crystalline powder and a sticky aromatic syrup in a lab. 3,4-Dihydroxybenzoic acid comes in as a pale, crystalline solid with a slightly tart scent. It melts at a moderate temperature, not too stubborn, which makes handling straightforward during lab prep. Water gets along with it just fine—heat speeds up how much you can dissolve. The molecule draws its strength from two hydroxyl groups sitting on a benzene ring separated only by a single hydrogen. They make it reactive, both a giver and taker of electrons, while the carboxylic acid group lends its own signature sourness. These features end up at the core of why this molecule can neutralize oxygen radicals or latch onto metal ions floating around in our cells.
Technical Specifications & Labeling
In the supply chain, purity matters because unwanted byproducts can scramble test results and cause headaches all around. The purest forms of this acid often reach 99 percent purity, which deserves a closer look before use in food or biotech. Most labels will reference its chemical identity, like C7H6O4, and a unique set of numbers helping to track it in chemical registries. Color, melting range, and solubility set the standards for each lot. Reliable supply outfits include details like storage temperatures, warnings about light or humidity, and expiration dates. Labels also stress that, depending on the extraction method, traces of plant-derived residues can linger, so downstream applications like supplements have to keep an eagle eye on origin and purity.
Preparation Method
The journey starts out simple in some cases. Old school, people extracted it from plant bark or fruit skins with hot water and alcohol, then filtered the crude mix over and over. Modern industry goes for speed and cleaner outputs. Sometimes, manufacturers oxidize vanillic acid or demethylate certain lignins, using chemical catalysts that break up natural polymers. Less common but still valuable, some labs engineer bacteria to ferment sugars and build this acid from scratch, promising cheaper and greener production when the process gets efficient enough. Any method needs tight controls to keep leftover solvents and unrelated side products out of the final batch.
Chemical Reactions & Modifications
In practice, a molecule like this earns its keep in how easily it accepts new chemical attachments. Esterification turns that acid group into something more lipophilic, widening where the molecule can travel in the body. The catechol core often undergoes oxidation reactions; those transformations spill into natural pigment formation and antibody research. Sometimes, chemists tweak one of the hydroxyls to map where antioxidants go in the body after consumption. Metal chelation is another handy trick, helping tie up metals like iron and copper that often stir up unwanted oxidative chemistry in plants and people alike. These modification routes anchor research in everything from new pharmaceuticals to the hunt for improved food preservatives.
Synonyms & Product Names
You likely hear this acid called protocatechuic acid in most life sciences circles. Other names pop up, like PCA or 3,4-DHBA. These synonyms mark the molecule’s chemical structure but also reflect its broad reach across food science, cosmetics, and supplements. Certain product lines will push one name or another depending on their marketing angle, but the science stays the same.
Safety & Operational Standards
With any lab chemical, respect for safety stays non-negotiable. 3,4-Dihydroxybenzoic acid doesn’t act as a wild card like some strong acids or volatile solvents. It still needs gloves and goggles to prevent irritation if it lands on skin or eyes. Dust inhalation stands out as a risk, especially in manufacturing or lab bulk handling, so good air circulation and dust control figure prominently in standard protocols. Regulatory agencies, like the FDA or EFSA, routinely ask for full toxicology packages before approving any compound for consumer markets. Keeping everything clean and dry, away from light and open air, helps prevent degradation and contamination.
Application Area
The reach of this molecule extends far past the lab bench. Most people encounter it unknowingly in plant-derived foods high in antioxidants. Nutrition researchers probe its behavior in the gut, where it appears to prevent some kinds of cell damage joined to heart disease and diabetes. Pharmaceutical teams explore it as an anti-inflammatory or adjunct to more advanced cancer drugs, betting that it can limit side effects or boost the performance of chemotherapy. In skin care, its antioxidant activity puts it in creams and serums that promise to help reduce oxidative stress and slow down photoaging. Food technologists turn to it as a preservative or flavor enhancer, drawing on its sharp taste and natural origins. Each new study opens the door to using it in another health or wellness application; only regulations and economics set the pace.
Research & Development
In academic science, I watch researchers go after big questions about how protocatechuic acid influences human health. New tools like mass spectrometers and omics profiling unravel how efficiently the molecule survives digestion and where it lands in body tissues. Teams drill into the mechanisms it uses to mop up free radicals or change gene expression tied to inflammation. Recently, collaborations between food scientists and pharmaceutical labs put the spotlight on how molecules like this may explain the "French paradox"—why some populations with fat-rich diets avoid sky-high rates of heart disease. Efforts ramp up to stretch the molecule's shelf life and tailor new derivatives with sharper therapeutic effects or better absorption rates.
Toxicity Research
Every promising molecule carries risk, which holds true for all phenolic acids. Toxicity studies in lab animals and cultured cells show a generally safe profile at normal dietary levels, but things change at very high doses: oxidative stress, organ stress, and changes to gut microbiota appear in some test groups. People rarely hit these dosages through normal eating, but supplement markets keep the pressure high for clear answers about long-term safety. Regulatory watchdogs evaluate real-world intake data against animal studies, with new research filling in gaps about bioaccumulation or interactions with other medications. Careful design of supplements—dose limits, purity checks, and transparent ingredient lists—helps keep consumers on the safe side.
Future Prospects
Judging from the speed of recent advances, the story around 3,4-dihydroxybenzoic acid feels like it has chapters yet to be written. Research momentum only builds as health science leans further into polyphenols and antioxidants for disease prevention and healthspan extension. The drive for cleaner manufacturing means the methods for making this acid will change, too. Expect to see more attention on green chemistry and bio-fermentation routes that cut waste and save energy. On the consumer side, more interest in natural products drives investment into safety, traceability, and transparency, raising the bar for how these compounds reach shelves and pharmacy counters. At the same time, emerging data will sharpen the division between science-backed health benefits and unsupported hype, pushing both the supplement world and pharmaceutical industry to raise their standards even higher.
It’s easy to let a name like 3,4-Dihydroxybenzoic acid get lost in a blur of scientific jargon. I remember running into it once in a college biochemistry course and thinking it sounded more like a riddle than something useful. These chemical compounds rarely get a headline, but many of the comforts of everyday life have deep roots in these unassuming molecules. This acid, also called protocatechuic acid, holds surprisingly practical value—spanning from health supplements to food science and even environmental cleanup.
Nutrition and Health—Closer Than Most Realize
You’ll spot 3,4-Dihydroxybenzoic acid in the wild, so to speak, inside lots of edible plants, including berries, olives, and some whole grains. Research teams keep discovering links between diets high in natural plant polyphenols and reduced risk of chronic disease. A lot of that work hinges on how these acids help mop up free radicals—the culprits behind cell damage and aging. Picture someone eating blueberries for breakfast, and they’re getting a healthy dose of this compound without a second thought.
Some supplement manufacturers use extracts standardized for protocatechuic acid, banking on its antioxidant properties. Studies point toward real promise in slowing inflammation and even helping certain cells fend off tumor growth. Nobody calls it a miracle drug, but adding it to the food chain definitely fits into efforts to keep inflammation in check and metabolic processes running smoothly.
Food Preservation—A Natural Ally
I used to work in a bakery that struggled with keeping whole grain breads fresh. Turns out, acids like 3,4-Dihydroxybenzoic acid have shown antimicrobial activity, keeping some types of bacteria and fungi from running wild. Researchers chase this property when they search for safer, less synthetic food additives. With people starting to turn their backs on chemically-sounding preservatives, natural antioxidants look friendlier on a label. Here’s a compound that can sneak a bit more shelf-life into foods—without dumping artificial stuff into the dough.
Environmental Clean-Up—A Silent Partner
Take a stroll around any wastewater treatment site and you’ll see tanks full of efforts to scrub out nasty byproducts of industry. Plant-based acids help break down pollutants and heavy metals. Sometimes, 3,4-Dihydroxybenzoic acid acts as a chelating agent, teaming up with other molecules to lock up harmful metals, which then makes cleanup safer and easier. It doesn’t work alone, but it definitely plays a supporting role. Environmental groups often look at these reactions to help tackle hard-to-manage spots like old industrial dumps—someone’s got to leverage every tool possible.
Beyond the Obvious—New Roles Emerging
Pharmaceuticals depend on molecules like this as building blocks for new therapies. Chemists tweak the base compound and get fresh leads for drug development. Sometimes the structure inspires new synthetic drugs, hinting at untapped medical applications. Bioplastics researchers dig into these plant acids to come up with new, more biodegradable materials. Fresh angles pop up every year as scientists keep looking for eco-friendly raw materials.
Each time I see this acid show up in a research paper, it’s a reminder that powerful solutions sometimes hide in places where only the curious go digging. Whether it’s about fighting spoilage, cleaning up environmental messes, or nudging health a little further, 3,4-Dihydroxybenzoic acid delivers more than a mouthful—both in name and in real-world value.
The Science and Real-World Context
Walk down the supplement aisle or scan labels on personal care items and it’s easy to run into complicated names like 3,4-Dihydroxybenzoic acid. This compound belongs to the family of phenolic acids, molecules found all over nature, especially in fruits and vegetables. Scientists know it as a breakdown product of plant polyphenols, and research keeps uncovering new roles for phenolic acids in foods and the human body. The presence of 3,4-Dihydroxybenzoic acid in certain berries and even coffee puts the compound into everyday diets already.
Concerns about any new compound boil down to one point: will eating or using it harm anyone? This largely depends on dose, quality control, and long-term studies. Most laboratory results paint a positive picture, showing low toxicity even at doses higher than anyone would get from drinking coffee or eating berries. Studies in animals show no strong signals for organ toxicity or big changes in blood markers, especially at dietary-level consumption. When looking at what’s in our foods, 3,4-Dihydroxybenzoic acid doesn’t stick out as a problem ingredient.
What Sets Real-World Safety Apart
Real safety relies just as much on preparation as it does on chemical properties. In my experience, natural does not mean harmless. Plenty of so-called safe compounds can cause issues if purity slips, or someone consumes industrial quantities. The risks change based on whether the acid comes as part of a whole food, in a supplement blend, or is extracted for use as a preservative or flavoring. If companies source impure or contaminated material, problems start to creep in that wouldn’t show up in the original research. Trustworthy sourcing and quality checks matter here.
The general use of 3,4-Dihydroxybenzoic acid in trace amounts in foodstuffs and cosmetics suggests a long baseline of human exposure. Most reports of negative effects are rare and usually tied to allergic reactions instead of inherent toxicity. These cases show how individual responses shape the picture more than statistics—the same goes for peanuts, gluten, or dairy allergies. People with known intolerance or allergies should watch out for reactions in new products, but for the average healthy person, the scientific consensus holds strong for safety in customary amounts.
Using Evidence, Not Hype, to Make Decisions
Too often, online chatter about chemical names turns into a storm of half-truths and worry. It helps to remember that research does not support panic over trace amounts of 3,4-Dihydroxybenzoic acid in food. Regulatory groups like the U.S. Food and Drug Administration focus more on high-dose, chronic exposure when sounding alarms over food ingredients. Global food safety authorities have not placed heavy restrictions or issued public warnings for products with this compound, especially at the levels found in everyday foods.
Advice for anyone who wants to go beyond the label: lean into variety, stick to reputable brands, and keep an eye on any new supplement with high concentrations or unclear sourcing. Too much of anything, from vitamins to water, carries potential hazards. Current evidence gives little cause for fear as long as 3,4-Dihydroxybenzoic acid stays in the realm of normal dietary exposure and tested consumer goods. Continued research and vigilance from both regulators and shoppers keep safety at the forefront.
A Closer Look at the Storage Puzzle
Storing chemicals properly rarely gets much attention outside of the lab, but 3,4-Dihydroxybenzoic Acid deserves a closer look. People often overlook the impact of small details—light exposure, bottle caps left open, or fluctuations in temperature. Those details can mean the difference between a stable reagent and a ruined experiment. Years spent working around organic acids taught me one thing: shelf life isn't just a matter of ticking months off the calendar. It's a story of how well you respect the stuff and how you treat it from the start.
Why Storage Really Matters for Stability
This compound, also known as protocatechuic acid, falls into a family of antioxidants that easily pick up changes in their environment. If moisture sneaks into the container, that powder clumps and degrades. Leave the bottle open on a bench under harsh lighting, and color changes might show you the beginnings of oxidation. Those subtle shifts steal away the purity researchers count on, leading to unreliable data or extra costs replacing product.
Published studies and chemical suppliers recognize 3,4-Dihydroxybenzoic Acid as sensitive to air and light. Most recommend storing it in airtight, amber bottles. Cool, dry places like dedicated chemical refrigerators reduce the risk of unwanted reactions. I once lost half a bottle to clumping after storing it on a warm shelf near a window. After that, I made it a rule to transfer new solids to smaller vials and squeeze out as much headspace air as possible.
Making Shelf Life Last Longer
Three years seems to be a common time frame for many solid organic compounds if unopened and kept cool. Opened containers risk contamination every time the lid comes off. Truth is, it’s better to use up what you need quickly. As soon as you spot color darkening or sticky crystals, it's wise to question whether that batch is still good enough for careful experiments.
One lab habit that helps: recording the date you opened the bottle. This seems basic, but plenty of people forget or think it won't matter. Months later, you’ll be glad for a clear reminder, especially if you start seeing unexpected results. Regular checks for moisture and physical changes go a long way, too.
Dealing With Practical Challenges
Some labs don’t have endless cold storage or specialty containers. What works: Stash small amounts in tightly sealed glass vials with a desiccant packet inside. In most situations, the fridge does more good than harm—just keep the bottle out of the freezer, as repeated thawing can wreck delicate powders. Many suppliers send product with extra air removed or in inert gas-flushed packaging, but those defense layers vanish once the seal breaks.
Disposal of expired or suspect samples requires care. Local chemical waste programs expect accurate labeling and safe handling. Ignoring expiration on the label may lead to ineffective results downstream in synthesis or biological assays. Fresh, well-kept acid acts more predictably and keeps costs down by preventing avoidable mistakes.
Better Practices Mean Better Science
The story of 3,4-Dihydroxybenzoic Acid storage is a lesson in respect for details. Maintaining quality starts with capped bottles, dry hands, cool shelves, and away from light. Many forget that old habits like label dates and periodic checks can rescue entire projects from unwanted surprises. Small changes build up over time, leading to more consistent research and less wasted material. Good stewardship of your chemicals gives you tools you can trust.
Seeing the Formula Beyond the Textbook
As someone who has navigated the world of biochemistry textbooks and research benches, I have often wondered how a simple arrangement of atoms can spark such deep curiosity—especially with molecules such as 3,4-Dihydroxybenzoic acid. Known amongst chemists as protocatechuic acid, this compound draws attention for more than just its complicated name or its role in leafy foods.
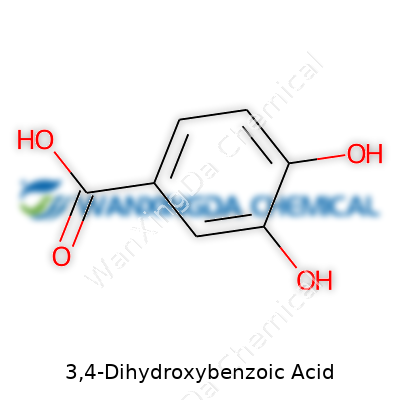
Let’s get real about its structure: a benzene ring carries two hydroxyl groups at carbon positions 3 and 4, flanking a carboxylic acid at position 1. That arrangement gives it the chemical formula C7H6O4. Its molecular weight falls at 154.12 g/mol. It’s clean and tidy—an aromatic layout that lets this acid do more than just look good on paper.
Why Should Anyone Care About This Molecule?
Most people don’t wake up thinking about chemical structures. I get that. But this acid shows up in plants and even in us after we munch on veggies and fruits high in polyphenols. Studies link it to antioxidant activity—something we hear a lot about when talking about fighting free radicals or supporting heart health. My graduate advisor always said, "Structure begets function," and in the case of 3,4-Dihydroxybenzoic acid, those two hydroxyl groups seem to boost its ability to neutralize reactive oxygen species. Put simply, the way the molecule looks on paper translates to how it acts in the body.
This isn’t just high-level science for the sake of curiosity. Research papers point to its potential—not just for personal health but also for food preservation and possibly even medicine. For people interested in sustainable agriculture or food science, this molecule isn’t just another oddity; it’s a clue to how plant-based diets may protect against chronic diseases.
Connecting Lab Science to Everyday Life
Real progress in science means someone finds a link between the shape of a molecule and the way a plant fights off disease or how our bodies handle inflammation after a tough workout. That’s where the exact atomic arrangement comes in. This isn’t about building chemistry exam questions; it’s about figuring out why eating blueberries or olives—a couple of sources for this acid—actually seems to matter.
Clearly, one small molecule won’t solve every health problem. But studies keep providing evidence that individual compounds like this support a bigger story about nutrition, plant resilience, and possibly human disease prevention. Without knowing the structure, or caring about it, those connections get lost in the shuffle of endless health claims and food trends.
Where Do We Go From Here?
Deeper research helps us draw clearer lines between the structure of natural compounds and their real-world impacts. From a chemist’s perspective, understanding 3,4-Dihydroxybenzoic acid paints a picture of how even subtle tweaks in molecular arrangement lead to different outcomes in both living systems and industries using plant-based materials.
Having grown up around gardens and later spent time trudging through labs, I’ve seen how curiosity about a single molecule can ripple into new insights for farmers, food producers, and health researchers. For all the complexity that science brings, a solid grasp of molecular structure remains one of the simplest—and most essential—steps toward smarter, evidence-backed choices in health and the environment.
Real-life Experiences Meet Chemical Handling
Most people scroll right past topics like chemical packaging. It sounds so specialized, not something that crops up outside professional circles. But anyone who has worked in a lab, tinkered with research, or kept an eye on supply chain news knows that how chemicals travel from manufacturer to bench impacts more than just the science. I remember the first time I ordered 3,4-Dihydroxybenzoic Acid for a university project. The supplier boxed it up in workable amber glass bottles, each sealed tight, and the package had enough labeling to leave no guesswork. Seems ordinary, but that small detail protects the product and everyone in the room.
3,4-Dihydroxybenzoic Acid isn’t just another powdered reagent. It’s got a knack for picking up moisture, and it doesn’t take much humidity in the air to turn a free-flowing solid into a stubborn lump. That’s why experienced folks prefer containers that keep the air out, like screw-cap bottles with proper liners. In the years since my first order, I’ve seen wrong calls—cheap plastic jars that crack under stress, shipments packed loose without desiccants. One slip, and the batch downgrades from ‘analytical grade’ to mystery goo.
Details Matter in Every Step
Suppliers often use airtight glass bottles or strong HDPE plastic jars, typically ranging from small research-sized 10-gram vials up to larger 500-gram or kilogram formats. These containers aren’t random—they stand up to rough handling, stay inert, and can be resealed after each use. The bottles usually arrive tucked into insulated packaging or cushioned boxes. I’ve received shipments in the peak of summer that stayed bone-dry only because the supplier added silica gel pouches inside the cartons. That bit of foresight makes a difference.
Safety labeling isn’t just a nod to regulations. Good packaging includes dot-matrix-printed labels that stand up to solvents and feature batch numbers for traceability. Some suppliers even list recommended storage conditions, warning you not to treat this acid the way you’d treat shelf-stable sugars or salts. That established trust. If there’s a problem—say the bottle lost its vacuum or you spot a clump—the batch and lot information gets you a quick answer rather than a shrug from customer service.
Risks and Ongoing Challenges
Recent years have seen an uptick in regulations on chemical shipping, especially across borders. Customs officers want clear hazard stickers, compliant secondary containers, and shipping manifests matching what’s inside the box. Laboratories are caught in these checks, anxiously refreshing tracking links and hoping nothing gets flagged. Small slip-ups—illegible print, leaking lids—hold up projects for weeks. Clear communication from suppliers helps, but sometimes everything hinges on basics like packing tape and the right container.
Reduce hassle and risk by building relationships with reputable suppliers. Check for tamper-proof seals, up-to-date documentation, and responsive sales teams who explain what’s new in packaging instead of reciting old policies. I’ve learned over time that the best suppliers invite feedback. If enough people speak up, you see changes: glass gets thicker, seals improve, and labeling leaves less room for error. As science leans harder on reliability and traceability, the humdrum business of how you get your 3,4-Dihydroxybenzoic Acid matters more than ever.