3,5-Difluorobenzoic Acid: An Editorial Perspective
Historical Development
Organic chemistry shapes the evolution of countless tools, from medicine to material science. 3,5-Difluorobenzoic acid, a fine-tuned benzene ring dressed in fluorine’s reactive armor, only entered the mainstream chemical landscape once the petrochemical boom pumped out affordable, pure precursors. Early research on substituted benzoic acids trailed the development of synthetic dyes and pharmaceuticals. As labs in the twentieth century refined methods for aryl halogenation and selective fluorination, compounds like this acquired a reputation for versatility in both academic and industrial research. The focus shifted from mere synthesis toward understanding substitution patterns, especially when chemistry demanded more from molecules— whether it involved safeguarding a drug from metabolic decay or prepping a precursor for high-end electronics.
Product Overview
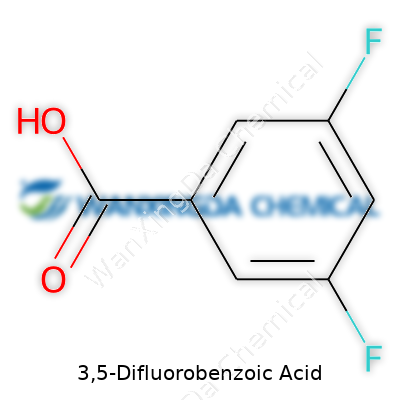
What draws attention to 3,5-difluorobenzoic acid is not some flash-in-the-pan novelty, but its reliable character in real world labs. This molecule wears its two fluorine atoms at the 3 and 5 positions on a benzoic acid backbone, giving it the toughness that only fluorinated aromatics can claim. Working with it gives researchers a solid foundation: predictable reactivity, a manageable handling profile, and compatibility with an array of downstream transformations. Unlike many obscure intermediates, it holds a track record in both published syntheses and patent filings. Navigating literature, chemists find reports noting it as a stepping stone toward agrochemicals, specialized polymers, and even diagnostic agents for imaging. To see it in action is to appreciate how small changes at the atomic level open doors for discovery.
Physical & Chemical Properties
This compound crystalizes as a white solid, often forming fine needles or powder depending on solvent and temperature during isolation. The presence of fluorine delivers a two-pronged benefit: altered electronic properties and increased metabolic resistance for those applications that venture into biology. The melting point sits comfortably above ambient temperatures, making storage and transfer routines relatively straightforward. Solubility favors organic solvents over water, aligning with the affinity expected of halogenated aromatic acids. The acid group offers a well-understood point for further chemistry—deprotonation, amidation, or activation—while the aromatic core, especially when partially fluorinated, resists unwanted side reactions under many standard laboratory conditions.
Technical Specifications & Labeling
Common chemical supply catalogs list it with the CAS Registry Number 446-18-8. Purity typically exceeds 97 percent in research and industrial lots, reflecting both the reliability of production and the need for consistent downstream results. Labels include a molecular weight of about 174 g/mol and note the distinct structural formula with the fluorines on the 3 and 5 positions. Spectroscopic profiles—proton and carbon NMR, FTIR, and mass spectrometry—form the backbone for authenticity checks, eased by the characteristic shifts associated with the aromatic fluorines. Such transparency enables researchers to spend more time on meaningful experiments, less on troubleshooting questionable batches.
Preparation Method
From the viewpoint of the working chemist, the main approach involves selective fluorination of a parent benzoic acid or fluorination at an earlier precursor stage, followed by oxidation or carboxylation. Electrochemical fluorination once fascinated old-school process chemists for its brute-force simplicity, but milder, catalytically-controlled reactions have gradually taken center stage. Advances in palladium-catalyzed coupling, direct fluorination with reagents like Selectfluor, and elegant regiochemical control let labs access these derivatives with less waste, fewer hazardous byproducts, and greater scalability. The real win comes from methods that reduce steps and cut reliance on rare metals or overly aggressive reagents. Not every route lines up for full industrial adoption, but the toolbox grows every year—a testament both to curiosity and to the push for sustainable chemistry.
Chemical Reactions & Modifications
The two fluorine atoms alter reactivity both electronically and sterically. This effect becomes obvious in electing sites for further functionalization. For example, electrophilic aromatic substitution takes a back seat, nudged by the electron-withdrawing effect of fluorine. Nucleophilic attacks at the aromatic ring almost disappear, but transformations at the carboxylic acid are ripe for exploitation. Simple steps turn the acid into an ester or amide, or activate the carboxyl for coupling with drug fragments or specialty polymers. Where other benzoic acids act as commoditized templates, the difluorinated version invites scrutiny in medicinal chemistry, especially where biostability, membrane penetration, or binding affinity require a unique “bite.”
Synonyms & Product Names
This molecule answers to many names in technical catalogs and archives. 3,5-Difluorobenzoic acid itself remains most direct, but you’ll spot references to m,a-difluorobenzoic acid, 3,5-DFBA, or its IUPAC equivalent 3,5-difluorobenzoic acid. Each name tells a story about the user—a pharmacologist concerned with regulatory rigor, a process chemist jotting shorthand, a patent attorney ensuring every synonym is in play for coverage purposes. Such multiplicity keeps research inclusive and accessible across borders and backgrounds, even if it at times crowds online searches with redundant entries.
Safety & Operational Standards
Lab experience shows that fluorinated aromatic acids trend toward low acute toxicity, but prudence stays at the forefront. Dust and powder inhalation can irritate respiratory tracts and skin, prompting glove use and operation in well-ventilated hoods. Handling procedures mirror those for similar small-molecule carboxylic acids—closed transfers, labeling, and careful disposal. Fluorine’s reputation in combustion and organofluorine toxicology tends to raise alarms out of proportion with risk on the bench scale, yet training against complacency remains crucial. Standard regulatory frameworks, including classification in many countries’ chemical inventories, guide both workplace safety and downstream product stewardship. My own practice puts a premium on risk assessment and communication—more vital with unfamiliar students than with veteran researchers who learned hard lessons firsthand.
Application Area
3,5-Difluorobenzoic acid has quietly shaped breakthroughs by delivering stability and functional options that non-fluorinated acids often lack. In medicinal chemistry, it’s a stepping stone in the design of drugs that resist metabolic breakdown—fluorine’s signature effect. Its structure attracts interest in agrochemicals, giving herbicides or fungicides not just efficacy but crucial selectivity and persistence. Specialty polymers, dyes, and imaging agents have all benefited from its unique electronic properties. Diagnostic applications include precursors for radiolabeled compounds in medical imaging, with fluorine-18 posing minimal synthetic challenge once the parent scaffold is on hand. Day-to-day, its impact lies not in high glamour but steadfast utility—turning up at the early stages of research programs that later make news in popular science.
Research & Development
New chemistry rarely develops in a vacuum; it thrives in the overlap between curiosity and necessity. 3,5-Difluorobenzoic acid smiles on both camps. Academic groups use it to probe fluorination effects on aromatic reactivity, drug metabolism, or material durability, while industry teams pick it for functional performance in coatings, electronics, and crop protection. I’ve seen colleagues publish on its use to tweak enzyme inhibitors, chasing that razor’s edge between blocking a disease pathway and sparing healthy cells. Strong patent activity signals both the molecule’s perceived value and the breadth of its application. Emerging research examines its fate in the environment and its downstream breakdown products. Each discovery in the lab, though incremental, pushes the baseline for pharmaceuticals, diagnostics, and specialty chemicals forward.
Toxicity Research
Few synthetic chemicals arrive fully mapped for risk, and 3,5-difluorobenzoic acid is no exception. Most available studies report mild acute effects, with little bioaccumulation in standard model organisms. Fluorine substitution makes metabolic degradation slower, which sits well in drug design but invites questions about long-term effects in the environment. Still, available toxicity screens tilt in its favor, especially compared with legacy organofluorines that have stirred regulatory scrutiny in recent decades. Vigilance remains wise. The field asks more nuanced questions today than in the past: Will transformation products linger in soil or water longer than expected? Are there pathways to safe degradation? Sharing answers, even when inconvenient, serves both the public and the scientific community better than false reassurance. Ongoing research, both published and in-house, will keep filling gaps in the safety story.
Future Prospects
The road forward for 3,5-difluorobenzoic acid traces the broader arc of fluorinated chemistry. Demand in advanced pharmaceuticals and sustainable agrochemicals is not slowing, and neither is interest in high-performance materials built for extremes of temperature and corrosion. Improvements in selective fluorination—faster, safer, more cost-effective—could pull this compound down the price curve, opening access for smaller companies or academic groups. Greener synthesis routes, perhaps biocatalytic or harnessing renewable feedstocks, could address regulatory and environmental pressure. As with every molecule in this class, success will depend on meeting performance needs without skating past the obligation for transparency and responsibility. My years in the field have taught the unpredictable power of small building blocks like this: their quiet presence often drives stories of unlikely progress and next-generation technology.
Why Chemists Keep Turning Back to 3,5-Difluorobenzoic Acid
3,5-Difluorobenzoic acid doesn’t get much headline space outside scientific circles, but its uses touch industries that affect daily life for millions. I’ve spent enough time working alongside pharmaceutical researchers to know that small molecular tweaks can open up major breakthroughs, and this compound plays a role in some of that progress. Having watched research division staff order pounds of the stuff, I can tell you that the drive to improve medicine and materials always leads scientists back to building blocks like this one.
Setting the Stage for Medical Research
Modern drug discovery depends on assembling molecules from key components that tweak how a finished medicine behaves inside the body. This acid offers a simple structure with a pair of fluorine atoms placed just right for synthetic chemists to tinker with. Adding fluorine tends to change a drug’s stability or how it interacts with target cells, and these changes aren’t just theory—the difference shows up in clinical results. Over half of top blockbuster drugs pack at least one fluorine atom for this reason, so raw ingredients like 3,5-difluorobenzoic acid become regulars in pharmaceutical labs. Biotech startups often lean on similar intermediates as they work to speed up the development of cancer treatments or antivirals, and every time, the compound helps them get a little closer to a safe new product.
Beyond Pills: New Materials and Agriculture
Applications don’t stop with medications. In my experience working with folks in chemical manufacturing, specialty materials often owe their unique features to small molecules that sometimes go unnoticed. For example, fluorinated building blocks can deliver polymers with better resistance against heat, moisture, or harsh chemicals. These qualities make a difference anywhere from automotive parts to smartphone coatings. I’ve seen chemical companies choose difluorinated acids to create tailored polyesters and resins, winding up with improved durability that lasts for years instead of months.
On another front, agriculture keeps looking for safer, more effective crop treatments. Pesticide chemists prize molecules that break down less rapidly in sunlight and don’t wash away with every rainstorm. By starting with something as specific as 3,5-difluorobenzoic acid, they can craft compounds that hold up in the field without running the risk of sticking around forever in soil and water supplies. Regulatory agencies ask for data showing better safety profiles, so this kind of chemistry turns into a selling point for agrochemical firms aiming to do the right thing.
Challenges and Smarter Choices
Even with its versatility, scaling up the production and handling of difluorobenzoic acids brings up safety and cost concerns. Engineering teams look for cleaner processes and safer handling protocols that minimize environmental impact. I’ve worked alongside people who design advanced filtration and waste treatment systems because they see firsthand the need for responsible chemical management. Green chemistry advocates continue to develop new ways to make and use these molecules more safely, pushing for methods that limit exposure and reduce waste.
Looking Ahead
We keep coming back to 3,5-difluorobenzoic acid because industry keeps demanding more: better drugs, smarter materials, and cleaner agriculture. It won’t get the same name recognition as final products, but without foundational compounds like this one, progress in these fields would stall. Real change means not just using what works but thinking through the full life cycle—making the most while minding our impact. As science marches forward, paying close attention to both the power and the responsibility that comes with chemical innovation will steer us in the right direction.
Getting Real About Chemical Structures
I still remember my first days in an undergraduate lab, staring at hand-drawn molecular diagrams, trying to make sense of lines and letters. Chemistry loses people quickly if it gets too abstract, but the basics matter more than most folks think. Take 3,5-Difluorobenzoic acid, for instance. Its chemical formula, C7H4F2O2, looks technical, but really, it’s just a representation of straightforward hands-on chemistry: seven carbon atoms, four hydrogens, two fluorines, and two oxygens. Each element and every bond has a role in how this compound behaves, whether in the lab or in a larger manufacturing process.
The Numbers Don’t Lie: Molecular Weight and Why It Matters
The molecular weight of 3,5-Difluorobenzoic acid comes out to about 158.10 grams per mole. That number gives researchers and industry folks a standard to work by. You can’t measure out chemicals for a reaction without knowing how much a mole weighs. It gets referenced in shipping labels, lab notebooks, quality checks—practically everywhere this compound travels. I’ve seen mistakes happen when molecular weights get copied wrong. The downstream mess can set back projects, burn up budgets, and frustrate even the most patient chemist.
Fluorine Makes a Difference
Tossing fluorine atoms onto a benzoic acid ring isn’t just a textbook example of substitution. It actually shifts chemical behavior in ways that matter quite a bit for industry and research. Fluorine’s strong electronegative pull tweaks the way the acid dissolves, reacts with other molecules, or even breaks down in biological systems. I’ve seen pharmaceutical teams choose difluorinated benzoic acid derivatives because the extra fluorine atoms make a drug candidate more stable or, sometimes, better at targeting the right enzymes. These design choices get debated for weeks in development meetings, because a single atom swap changes how a compound acts in the real world.
Experience Shows: Why Details Like This Matter
Details like molecular formula or weight might seem dry, but time and again, I’ve seen how they build the foundation for productive lab work and smart business decisions. High-precision measurements keep experiments reproducible. Accurate formulas stop mix-ups in synthesis, storage, or transfer—especially crucial for pharma, where even tiny deviations might cause big problems for patient safety or regulatory review. There’s a reason why the top journals push for accurate data reporting, and why senior researchers always double-check these numbers before signing off on a report.
Potential Solutions for Common Hurdles
People trip up over chemical data all the time. Manual transcription errors still pop up in reports and shipping manifests. One way forward is using verified digital platforms that double-check entries for formula and weight. Cloud-based lab notebooks, automated synthesis software, and regular training help reduce human error. Setting up clear standard operating procedures to verify these fundamentals before moving on to riskier stages pays off. Teams that spend more upfront on digital accuracy tools save more in the long run, both in time and in the cost of fixing preventable mistakes.
Keen Eyes and Sharp Pencils
At the end of the day, 3,5-Difluorobenzoic acid’s value comes from a mix of careful measurement and hard-won experience. Chemistry may be built on numbers, bonds, and formulas, but success depends on people willing to sweat the small stuff, double-check their work, and embrace the idea that every atom counts. The details really do make the difference in chemistry, from lab bench discoveries to real-world applications.
Everyday Storage Makes a Difference
Every lab comes with its own set of unspoken rules, learned through spilled vials, pungent smells, and mornings spent searching for lost reagents. 3,5-Difluorobenzoic acid may not shout for attention like some volatile chemicals, but ignoring its need for proper storage can trip up even seasoned chemists. Many see white crystalline powders and assume they're harmless, stacking bottles side by side without a second thought. That laid-back approach can open the door to contamination, accidents, and wasted money.
Fluorinated Acids and Risk
Fluorinated compounds deserve respect. Their chemistry brings extra stability, but it can also introduce risks you might not expect. 3,5-Difluorobenzoic acid might not leap from the shelf in a gout of flame, but dust from a leaky bottle can irritate the skin and lungs. The acid sits in the category of “low to moderate hazard,” according to GHS criteria, which means open jars and poor habits catch up with you over time. I've seen bottles caked with powder, caps cracked from over-tightening, and containers wedged into sunny spots next to the window – none of these bode well for quality control or safety.
Temperature and Environment Matter
Some chemicals forgive lapses in storage standards. This one won't. Sunlight speeds up degradation for aromatic compounds, and temperature swings can leave you with unusable material or an unreliable experiment. It pays to keep bottles in a cool, dry cabinet, far from heat sources. Many labs rely on flammable or corrosive cabinets, though this acid doesn't demand a fireproof safe. Instead, it wants stability: somewhere away from acids like hydrochloric or hydrofluoric, away from bases, and definitely separated from oxidizers. A dry box with silica gel does wonders in humid climates, halting the clumping that makes powders infuriating to measure.
Labeling and Mindfulness: Small Steps, Big Impact
A faded label tells you everything you need to know about a lab’s attitude. Without clear marking, date of opening, and the chemical’s full name, you might as well be tossing coins. Safety doesn’t rely on memory. Clear labels and an up-to-date logbook help avoid confusion, prevent accidental mix-ups, and make inventory less of a headache. Nobody wants to waste time tracking down who left a container open or which bottle is past its prime. Quality suffers, and so does safety.
Reducing Risks, Building Better Habits
Some labs swear by once-a-month checks for degradation and leaks. Routine inspections help catch issues early, long before they spiral. Using dedicated spoons or spatulas for solids like 3,5-difluorobenzoic acid keeps everything cleaner. Regular hands-on practice with proper PPE – gloves, goggles, dust mask for weighing – becomes second nature. Rules posted on the chemical shelf may look old-fashioned but set a tone that protects everyone. Chemical management is about mindful habits, not just storage containers.
Clear, Careful Storage: An Act of Respect
Digging through bins of poorly labeled powders doesn’t inspire confidence in a lab’s results or its culture. Simple precautions and steady habits shape a safer, more reliable research environment. It’s easy to cut corners, especially under pressure, but storing compounds like 3,5-difluorobenzoic acid with care saves time, money, and headaches down the line. Every bottle on the shelf tells a story – making it a good one is up to us.
Seeing Chemistry Beyond the Formula
Working in a chemistry lab for years teaches you to give every substance its due respect. Even something with an innocent-sounding name like 3,5-difluorobenzoic acid can introduce trouble if ignored or underestimated. This isn’t sensationalism; this comes from seeing folks get caught off guard by what they thought was an everyday compound.
The Real World: Skin, Eyes, and Breathing
I’ve watched students new to organic synthesis brush off the need for gloves only to regret it. Aromatic acids and their derivatives—especially those with halogen atoms like fluorine—aren’t just theoretical hazards. 3,5-difluorobenzoic acid, with its two fluorine atoms, behaves like its benzoic acid cousins, but the added halogens step up the toxicity game. Touch this compound and you might end up with skin irritation. Splash to the eye? Think burning, redness, even possible long-lasting damage. Breathe it in? The nose and throat can sting. This lines up with what you learn from trusted sources like the Material Safety Data Sheet or regulatory guides. The issue here isn’t just risk to workers, but the ripple effect to shared spaces, unknowing colleagues, and the environment.
Regulations and Why They Exist
Some folks shrug off the warning labels, thinking regulators exaggerate. Over the years, I’ve seen regulations shift in response to real incidents. 3,5-Difluorobenzoic acid hasn’t become a household threat, but the presence of fluorine rings alarms bells in countries with strict chemical control, like Germany and the United States. National Institute for Occupational Safety and Health and European Chemicals Agency carry listings for chemicals of concern, even for those not flagged as highly toxic or carcinogenic. The point isn’t to create red tape, but to help keep labs and workers out of the emergency room.
Bringing Awareness Into the Everyday Lab
No matter the size of the operation, consistent, careful practice matters. Splash goggles, gloves, and a fume hood aren’t overkill for 3,5-difluorobenzoic acid. Store it with other organic acids, away from strong bases or oxidizers, inside a well-labeled container. If in doubt, double-check storage guidelines to avoid dangerous reactions. I’ve seen accidents where improper storage led to unexpected chemical vapors, or worse, unexpected chemical fires.
Waste Isn’t an Afterthought
Disposal gets overlooked far too often. Pouring this acid down the drain counts as both a health risk and an environmental hazard. Municipal treatment plants aren’t set up to handle specialty organic chemicals, especially those with fluorine, which resist breakdown. Instead, set aside a properly labeled waste container and follow local hazardous waste procedures. In my own lab, this routine kept both pipes and consciences clean.
No Substitute for Education
Education can close gaps that lead to accidents. New workers benefit from orientation sessions covering handling procedures for all chemicals, including those that seem routine. Stories—warts and all—from those with more years in the lab go a long way toward making lessons stick. I always found that concrete examples made the rules real, not abstract. No scare tactics, just respect for what’s possible if safety steps get skipped.
Small Steps, Big Difference
At the end of the day, treating 3,5-difluorobenzoic acid with the right mix of caution and habit protects people, places, and careers. It may never make headlines, but it’s part of the unglamorous, vital backbone of every safe science workplace. And for that reason alone, special care isn’t just wise—it’s fundamental.
A Closer Look at Purity
In the world of chemistry, there isn’t much room for guesswork—especially when handling specialty compounds like 3,5-Difluorobenzoic Acid. Most researchers and industry folks know that purity is more than just a technical number. It’s the backbone behind reliable results. From what I’ve seen, major suppliers strive for purities above 98 percent, with some batches pushing into “trace impurity” territory. This high standard doesn’t just support tight experimental controls in the lab. It also drives consistent performance in pharmaceutical synthesis, agrochemical development, and electronics work where tiny fluctuations can throw an entire project off course.
When you’re working with reagents like this, you start spotting the difference between a compound that’s 95 percent pure and one that’s 99 percent. The odd percent or two could mean you lose reproducibility, trace contaminants muddy analytical readings, or yield dips unexpectedly. I’ve learned that high purity saves time and reduces headaches for anyone working at the bench or managing a supply chain. If the bottle tells you above 98 percent, you can usually trust that today’s top-tier vendors back that up with HPLC or NMR data. You pay a premium, sure, but you know what you’re getting.
Why Packaging Sizes Shape Usage
Packaging isn’t just about what fits on a shelf. In practice, size influences workflow, waste, and safety. Standard packaging forms for 3,5-Difluorobenzoic Acid cluster around small bottles—think 5 grams, 10 grams, and sometimes 25 grams—catering mainly to academic labs or those running pilot reactions. For larger industrial players, suppliers roll out 100-gram and half-kilo jars. Here’s the thing: the way these bottles are sized and sealed matters. Good packaging stops contamination and moisture, maintains product quality for longer, and cuts down on disposal problems.
I’ve been in labs where someone tried to buy bulk, thinking they’d save money, only to watch most of a big container go stale over time. If you’re working in research, it makes sense to buy only what you’ll use up in a month or two. Smaller packs mean fresher chemical for every project. Bulk containers find their place in production environments, with strict procedures to reseal after every use. Manufacturers have started to respond to these needs with tamper-evident seals, amber glass for light-sensitive chemicals, and improved labeling that keeps track of batch information.
Finding a Better Path Forward
It’s not enough to throw high-purity chemicals into the market without some thought behind the packaging. Some companies have begun partnering with researchers to rethink container design and sizing. I’ve seen initiatives introducing single-use sachets or environmentally safer materials for jars—creative moves that cut down on both waste and contamination risk. The next step? Encouraging an open line between makers and end users. If chemists and engineers keep sharing what works (and what doesn’t), the miracle of high-purity 3,5-Difluorobenzoic Acid won’t just be a win for science. It’ll be a win for safety, cost, and sustainability too.