The Layered Story of 3,5-Difluorophenol: Discovery to Destiny
Tracing the Historical Footsteps
Looking back, the story of fluorinated phenols reveals progress right alongside challenge. The tale of 3,5-Difluorophenol runs parallel to the broader movement in organic synthesis where adding fluorine to aromatic compounds became a lever for changing properties. Industrial chemists first tackled phenolic fluorination as part of demands for advanced drugs and agrochemicals in the last half-century. Once separated out from other isomers, 3,5-Difluorophenol opened doors for new types of molecular building blocks. This single compound mirrors the era’s push to balance innovation with new safety awareness.
Understanding the Substance
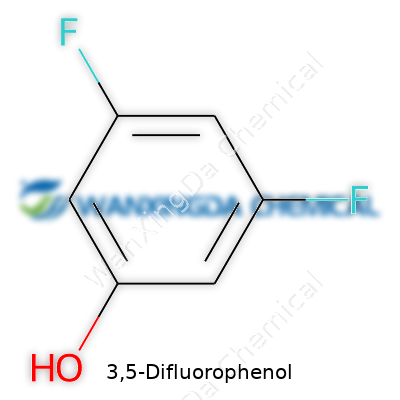
3,5-Difluorophenol stands as a simple molecule with two fluorine atoms fixed to a phenol ring. On paper, it falls into the group of halogenated phenols but, in reality, those two fluorines change everything about the way it behaves. The liquid looks clear, has a strong odor, and dissolves just enough in water to make handling it a careful business. Its melting and boiling points drop compared to unmodified phenol, proving that even small substitutions turn old rules on their head. Chemists have learned the hard way that the electron-pulling force of fluorine reshapes solubility, volatility, and acidity. Testing shows 3,5-Difluorophenol’s acidity fits between the parent phenol and trifluorinated versions—a detail that guides how it reacts in labs and factories.
Technical Details and Product Identification
Every bottle of 3,5-Difluorophenol arrives marked by its structure: a benzene ring carrying fluorine at the 3 and 5 spots and an -OH group. Chemists sometimes call it 3,5-DFP or 3,5-difluorohydroxybenzene, though the IUPAC name remains the golden standard in journals and regulatory filings. The CAS number never changes and stays linked to purity checks and analytical results. Labels on lab vials spell out hazards—corrosive, capacity for serious eye damage, and potential for acute inhalation risks. Above all, technical datasheets place purity at the center, since trace impurities can skew reaction results or downstream safety calculations.
How Production Happens
Making 3,5-Difluorophenol isn’t like brewing coffee. It starts with fluorinating a phenol derivative through direct substitution, leveraging modern fluorination agents under careful conditions. Experienced hands know to avoid over-fluorination, controlling time, temperature, and reagent ratios carefully. Purification calls for distillation under vacuum or complex chromatography steps. Labs chasing higher yields spend years tweaking catalysts and reactant order, using insights from both chemistry and engineering. Yields rise with new reagents and better protocols, each tweak driven by the relentless need for reliable and pure output.
Reactivity and Further Modification
3,5-Difluorophenol isn't just a bench curiosity; its electron-balance and steric profile let it play the middleman in countless reactions. Nucleophilic substitution stands out—engineers use it as a launchpad for attaching sulfonates, alkylating the aromatic system, or prepping for cross-couplings. Some downstream users run it through etherification or transform the phenolic function as part of custom synthesis for agrochemicals and pharmaceuticals. Cross-coupling chemistry, especially Suzuki reactions, rely on such difluorinated rings to tune biological activity and stability. Every tweak to its ring has downstream impact, a fact that feeds ongoing structure-activity investigations across industries.
Why Labels and Synonyms Matter
The chemistry world thrives on precision, but common names still pop up—3,5-difluoro-1-hydroxybenzene, or its shorter cousin, 3,5-DFP. But walk into any regulatory check or customs clearance and the only thing that counts is the IUPAC handle or CAS identifier. Companies filing for patents, environmental impact reports, or shipment logs face a world in which synonyms ease conversation but only strict identifiers ensure compliance and recognition. These barcodes of chemical identity keep the research, production, and legal sides all communicating.
Making Safety Real, Not Just a Label
Safety demands respect in the hands-on world of 3,5-Difluorophenol. Think skin burns, eye damage, and hazardous fumes if containment ever fails. Ventilation and personal protective equipment stand as the baseline, not as afterthoughts. Storage needs a cool, low-moisture place away from reactive agents. While accident rates stay low with careful planning, the potential for both acute and delayed health effects echoes through every handling guideline. Any laboratory or production plant must train teams not only on the basic hazards but also on emergency actions and the quirks of fluorinated materials. Waste management involves neutralization and legally mandated hazardous waste disposal. Inhalation risks grow with scale, demanding robust fume extraction and early warning systems for leaks or spills.
Practical Application in Science and Industry
3,5-Difluorophenol earns its keep in more places than chemical catalogs suggest. Pharmaceutical developers look at its molecular backbone as a shortcut to synthesize enzyme inhibitors, receptor ligands, or as a stepping stone in the hunt for better drug candidates. Agricultural science taps its reactivity to drive up selectivity in herbicides or insecticides, since the difluorinated phenol core gives molecules a mix of stability and targeted activity. Material chemists, especially those working on specialty polymers or electronics, use it to tweak thermal and chemical resistance. The power of the compound lies not just in its structure but in its flexibility across different research and product themes.
Research and Toxicity Realities
Academic groups, as well as research arms of major chemical makers, put 3,5-Difluorophenol under the microscope for both performance and toxicity. Studies on its breakdown in the environment point to moderate persistence, thanks to the carbon-fluorine bonds holding firm against typical biodegradation routes. Toxicology groups run battery after battery of tests—acute oral and dermal, reproductive, and long-term inhalation. Evidence so far points to risks from concentrated exposures rather than from trace background levels, but regulatory watchdogs keep their guard up, given the broader concerns about persistent organofluorine pollutants. There’s still a gap between what lab rats experience and the thresholds regulators set for field and workplace limits.
Prospects: Where Does the Story Go From Here?
As markets shift and green chemistry keeps gaining traction, 3,5-Difluorophenol stands at a crossroads. On one side, ongoing demand for precision building blocks in pharma and agrochemistry drives up research budgets and supplier investment. On the other hand, rising scrutiny of all things fluorinated gives policy-makers and chemists new reasons to rethink legacy routes and assess lifecycle footprint. Innovation will likely focus on reducing waste, cutting down harsh reagents, and chasing routes from renewable starting points. As someone whose own work revolves around specialty chemicals, it’s easy to see that safer, more efficient synthesis counts for more than legacy formulas. The next chapter will depend on finding that balance—between performance, safety, and sustainable production—without losing the versatility that has kept 3,5-Difluorophenol in labs and factories for decades.
You rarely hear about chemicals like 3,5-difluorophenol in everyday conversation. Still, this tongue-twisting compound deserves a spotlight, not just because it sounds technical, but because it sits quietly at the intersection of modern chemistry and the realities of daily life. If you’ve ever counted on prescription medicines, crop protection, or advanced materials, there’s a slim but real chance this compound played its role somewhere along the line.
The Quiet Workhorse in Chemical Synthesis
Some folks hear “phenol” and think back to high school chemistry—the sharp, distinctive smell, maybe even the odd headache after too much exposure. Swap in a couple of fluorine atoms, and chemists gain a building block that opens doors to all sorts of new molecules. 3,5-Difluorophenol is a base material for crafting active ingredients in pharmaceuticals and agrochemicals. Its structure makes it appealing; fluorine’s presence can help molecules behave differently inside the human body. Drugs often become more stable or more effective because of these tiny tweaks.
It’s not a household item, but medicinal chemists have leaned on difluorinated building blocks to improve drug candidates. They look for better ways for drugs to survive in the stomach, reach the bloodstream, or avoid being broken down before they work. I’ve seen research that points to fluorinated rings as a way to increase a medicine’s potency or fine-tune its side effects. These shifts aren’t always dramatic, but stacking enough small changes can flip a mediocre compound into a life-changing treatment.
A Niche Player in Crop Protection
Beyond the doctor’s office, farmers unintentionally encounter 3,5-difluorophenol’s chemical relatives. Scientists develop pesticides and herbicides using similar raw materials. The aim isn’t to keep backyard weeds at bay; it’s to provide crops shelter from pests and blight, sometimes with a fraction of the chemical load needed by older solutions. Researchers design these molecules for selectivity—they want to target organisms threatening to ruin food without causing excessive collateral damage.
These applications usually come with heated discussion. People have every reason to ask tough questions about chemical use in food. Regulatory bodies set limits, inspect residues, and ask for reams of safety data. Still, the presence of phenolic compounds, especially those custom-built with fluorine, helps make modern farming viable, especially under growing pressure from climate change and rising demand.
Stepping Up Safety and Sustainability
Handling any phenol, especially one designed for lab use, requires caution. Toxicity isn’t a rumor; it’s a fact. Respiratory, skin, and eye exposure create genuine risks. Factories and research labs gear up with personal protective equipment and strict handling procedures. There’s a big push to curb waste and consider greener chemistry when possible. Wider adoption of environmentally friendlier solvents, lower energy processes, closed-loop systems—these steps matter just as much as the discovery of fancy new molecules.
Solutions come from innovation more often than regulation. Safer manufacturing, reduced exposure, and new synthesis strategies have already cut down on environmental hazards. For 3,5-difluorophenol, the best outcome blends benefits in medicine and agriculture with smarter systems behind the scenes. Chemists who know these building blocks from the inside can shape the future: not by banning, but by improving, substituting, and finding pathways that work for both people and the planet.
Why Does 3,5-Difluorophenol Matter?
Chemistry shapes our world in small ways that easily slip past most people. 3,5-Difluorophenol stands out from the crowd, not because it’s a famous molecule, but due to how it shapes pharmaceuticals, agrochemicals, and even some material sciences. This isn’t a showstopper like paracetamol, but it often plays a supporting role in the labs and factories where compounds take shape before heading into medicines or crop treatments.
Chemical Formula: More Than Numbers and Letters
Ask a chemist about phenols, and you’ll find that tiny changes—like sticking fluorine atoms onto a benzene ring—can create big differences in how a molecule behaves. With 3,5-Difluorophenol, you start from phenol (that’s C6H5OH), and swap out hydrogens in the third and fifth positions for fluorine atoms. You end up with a formula: C6H4F2O. It looks pretty simple, but those two fluorine atoms switch up the molecule’s reactivity, boiling point, and how it fits into larger chemical designs.
Why Are Fluorinated Molecules Important?
Adding fluorine to aromatics may sound like a small tweak, but it messes with everything from metabolism in humans to resistance against degradation. Many drugs that need to last longer in the body, or survive harsh soils and pests in the field, borrow tricks from molecules like 3,5-Difluorophenol. These modifications help products stick around long enough to do their job—whether that’s killing a weed or targeting a tumor.
This approach has feet on the ground in everything from cancer therapy to advanced polymer development. Fluorine’s electronegativity controls how electrons move, making the molecule less susceptible to enzymes trying to break it down in living systems. That’s why you’ll spot tricks with fluorine throughout the patent literature on new drugs and next-gen materials.
Challenges in Making and Handling 3,5-Difluorophenol
Tinkering with fluorinated aromatics can get tricky. While fluorine makes a molecule more stable to many processes, actually getting those fluorine atoms attached takes some know-how and careful lab work. Some methods use hazardous reagents or call for special handling because of toxic byproducts. This isn’t a molecule you’ll whip up in a home chemistry set.
Sustainability creeps in too. Fluorine-containing chemicals sometimes resist breaking down after their useful life. Researchers keep hammering away at new routes that cut down the environmental impact, focusing on greener chemistry and safer waste processing. Some of the best ideas have come from swapping out older reagents in favor of milder, less toxic alternatives.
Solutions Through Better Chemistry and Policy
Adopting clean production routes makes sense, not only for worker safety but for reducing environmental footprint too. Incentives for green chemistry could shift how companies approach synthesis, especially as regulations tighten worldwide. Sharing best practices between chemical producers, universities, and watchdog agencies leads to more transparency and faster adoption of safer manufacturing.
Public awareness ties in here, too. When people outside labs understand why molecules like 3,5-Difluorophenol land in their prescription drugs or farm fields, conversations about safety and sustainable development move forward. If future research untangles safer, cheaper ways to make and dispose of these molecules, everyone gains.
Getting Real About Chemical Handling
Anyone working around chemicals like 3,5-difluorophenol knows lab safety isn’t just a checklist — it’s about respecting the risks. This compound shows up in a lot of research labs, especially in pharmaceutical and agricultural settings. Its structure, with two fluorine atoms punched into a benzene ring, means it's not just another bottle on the shelf. Handling it demands more than a shrug and a pair of gloves.
Understanding The Hazards
My years in the lab taught me that even experienced researchers can let their guard down with substances they use often. That’s where accidents sneak in. 3,5-Difluorophenol stands out for its reactive edge, and its fluorinated structure can up the stakes. Exposure through skin contact, inhalation, or even a splash in the eye could lead to irritation, burns, or worse. Some phenols absorb quickly and wreak havoc before you know it. While data on this specific compound’s toxicity isn’t always clear-cut, it’s smart to assume there’s serious risk until proven otherwise.
Why Protective Gear Isn’t Optional
One of the biggest mistakes beginners make is thinking a simple lab coat covers all bases. Nitrile gloves, chemical splash goggles, and a decent fume hood matter every single time. I’ve seen folks end up with rashes or breathing problems after skipping these basics. Even if a procedure feels routine, every open bottle or pipette transfer could be the one where fumes or drops go astray.
Ventilation and Safe Workspace Practices
Good airflow makes a difference. Working with solvents and fluorinated phenols often throws invisible vapors into the air. In my experience, a fume hood isn’t just a box with a fan; it’s the main line of defense against accidental inhalation. Cluttered benches make spills more likely, so keeping things tidy isn’t about being neat — it’s about cutting down risk.
Storage: An Overlooked Weak Point
Too many dangerous chemicals get left on countertops or shoved in crowded cabinets. With 3,5-difluorophenol, heat and sunlight can speed up degradation or trigger unwanted reactions. It’s best kept in tightly sealed containers, in a cool, dry spot, clearly labeled. Mixing it up with acids, bases, or oxidizers in shared storage can invite disaster. Clear labels and organization save lives, as much as they save time.
Spill Response Matters More Than People Think
Every lab ends up with a spill sooner or later. Preparing ahead saves people from scrambling in panic. Absorbent pads, neutralizing agents, and clear walkways all matter. Training is key — knowing who grabs what, who evacuates, who calls for help. Practicing for emergencies has made the difference between a close call and a real injury in the labs I’ve worked in.
Building Strong Safety Culture
Chemistry only gets safer when everyone buys in. Even experts can forget basics under deadline pressure. Regular reminders, sharing near-misses, and honest feedback help keep safety at the front of everyone’s mind. In my career, small habits — like double-checking containers and watching for signs of wear on gloves — add up to fewer injuries and more productive work.
Better Solutions Through Training and Design
No one solves chemical hazards with gear alone. Real improvements follow clear protocols, thorough training, and an open door for questions. Designing labs for safe traffic flow and accessible safety gear stops problems before they start. People do their best work when they don’t have to second-guess their safety every time they handle something like 3,5-difluorophenol.
The Reality Behind Purity Claims
Chemistry isn’t just done by people in white coats. People in many walks of life encounter chemicals, sometimes unexpectedly—whether in a research lab, supporting a school science project, or running a business that touches manufacturing. 3,5-Difluorophenol comes up in more fields than most folks realize. This compound draws attention because of its special role in organic synthesis, pharmaceutical work, and even agrochemical development.
Much of the discussion about this chemical circles back to how pure it actually is when you get ahold of it. Labs usually look for purities above 98% to avoid any trouble during reactions. Impurities aren’t just annoying; they can seriously derail results or even create risks nobody bargained for. Years ago, I worked in a university lab focused on drug discovery, where one unlucky bottle with a low-purity intermediate wiped out a week’s reactions—and more importantly, shot down our chances to repeat the experiment until new stock arrived.
Major chemical suppliers typically offer 3,5-Difluorophenol at a stated purity above 98 percent, catering to everything from academic research to commercial applications. Documentation, in my experience, reads like gospel: analysts check for residual solvents, related phenols, water, and inorganic ash. A pure sample keeps byproducts to a bare minimum, supporting reliable downstream reactions. Precision here is vital, not just for scientific accuracy, but in some fields, for regulatory or safety reasons.
What Do People Actually See?
So what does 3,5-Difluorophenol look like to the naked eye? The common answer: a white or nearly white crystalline solid. That matches what most researchers report across global chemical catalogs or share in hands-on experience. It doesn’t draw much attention sitting in a vial—just a small handful of off-white crystals, pretty typical for many substituted phenols.
Some folks may run into slight differences, especially if their storage isn’t ideal or the compound came from a less reputable supplier. Bad storage can nudge the white color toward a light yellow or cream, which could mean slowly forming impurities or minor oxidation. For a critical experiment, or in regulated industries, even that subtle color hint should ring warning bells—some labs may screen the material again with thin layer chromatography or NMR.
Anyone handling chemicals quickly learns that the absence of an obvious color doesn’t guarantee purity, but it’s not a bad first check. Crystallinity, not powderiness or stickiness, points toward a cleaner sample. Physical feel can sometimes betray too much moisture or contamination. Transparency from suppliers, plus documentation like a certificate of analysis, empowers users to dig a little deeper before risking valuable time and money.
The Value of Quality and How to Protect It
Protecting the quality of a compound like 3,5-Difluorophenol goes beyond just buying the top-purity brand. Storage in tightly closed containers away from heat and light keeps it in good shape. My own mishap with leaky lids taught me the cost of ignoring those details—it doesn’t matter if the original material is 99 percent pure if it degrades before use. A little vigilance up front saves big headaches down the line.
Chemists often trade advice about which suppliers deliver consistent batches and who seems to cut corners. Open communication about experiences, both good and bad, keeps the community one step ahead of surprise setbacks.
Practical Solutions Going Forward
As work with 3,5-Difluorophenol and similar compounds ramps up, keeping a core focus on purity and honest evaluation of the physical sample goes a long way. Requesting up-to-date analytical data, storing materials carefully, and investing in small confirmatory tests before crucial syntheses all help build a reliable workflow. The simplest early reviews—checking that a sample stays white and crystalline—give clues nobody should ignore.
Looking Beyond the Label—Why Safety Matters
3,5-Difluorophenol doesn’t exactly pop up on the list of household names, but in the realm of chemical manufacturing and research, it serves some important jobs. With a reputation for being corrosive, flammable, and potentially harmful if mishandled, it deserves more than just a quick glance before being shoved onto a lab shelf or loaded onto a truck. Stuff like this calls for a level of respect that everyone in the pipeline—from makers to carriers—can’t afford to ignore.
Handling and Storage: Paying Attention Isn’t Optional
I’ve worked around labs long enough to see the mess left behind by shortcuts—corroded shelves, leaking packaging, and confusion about what went wrong. For a chemical like 3,5-Difluorophenol, those small moments of care help prevent much bigger disasters. Moisture and heat turn it unpredictable; air-tight, corrosion-resistant containers help keep it stable. Nobody wants to hear about a spill that caught fire or a storage area that quietly filled with fumes.
I remember a time a leaking bottle ended up corroding a storage rack—luckily, the facility’s system caught it before it turned into an emergency. That wake-up call drove the team to rethink our storage habits. Glass containers with sturdy PTFE-lined caps did the trick. Putting those bottles in a segregated cabinet—away from acids, oxidizers, and bases—made sense, especially since certain chemicals play very badly together.
Transport: No Room for Improvisation
Moving 3,5-Difluorophenol doesn’t work like tossing boxes into a van. Flammable substances with nasty vapor risks belong in sealed containers that can handle a rough ride. Secure packaging matters just as much as keeping temperature and humidity low. Stacking boxes can crush cheap bottles, so a shock-absorbing crate can spare you from cleaning up a sticky, toxic mess.
Laws around Hazmat transport require labels—clear, legible, and weather-resistant. The right paperwork helps responders make sense of what they’re dealing with if something goes sideways. I’ve seen shipments held up just because documentation was missing or didn’t match the product inside, causing confusion and delays that companies don’t need.
Looking at the Bigger Picture: Training and Oversight
No checklist beats experience. Teams that handle chemicals well tend to get hands-on training and regular refreshers. I’ve seen what happens when training drops off—the slip-ups usually pop up fast. It helps when everyone involved, from storage supervisors to truck drivers, knows what to expect and what to do if something leaks.
Facility managers get peace of mind from routine inspections, not just for the bottles themselves, but the rooms and equipment around them. Ventilation matters, too; cutting corners might save cash in the short run, but one chemical cloud can shut a lab or warehouse for days.
Better Habits, Safer Outcomes
Safety with chemicals like 3,5-Difluorophenol shouldn’t rely on the rare expert knowing every rule—it’s about everyday discipline. Good storage containers, well-planned warehouse space, solid transport procedures, and ongoing education give everyone a stake in safe handling. The alternative has always been expensive, messy, and sometimes tragic. With stakes this real, cutting corners never pays off.