Looking at 3,5-Difluorophenylboronic Acid: Impact, Progress, and Possibilities
Tracing the Trail: Historical Development
Organic chemistry never stays still, and the story of 3,5-Difluorophenylboronic Acid gives a window into the progress researchers keep chasing. Back in the early days of boronic acid chemistry, most attention hovered around basic phenylboronic acids. Then came the push for tailored molecules—scientists adjusted the aromatic ring, fiddled with substitutions, and tried all sorts of reaction tweaks. Emerging from all that exploration, the difluoro-substituted version started gaining ground for its unique reactivity and versatility. I remember in grad school flipping through journals filled with cross-coupling studies—boronic acids got plenty of limelight. The difluoro variant hopped onto stage relatively recently, mostly as the need for specialty chemicals grew, especially after Suzuki-Miyaura coupling made efficient carbon–carbon bond formation accessible. These scientific steps shaped 3,5-difluorophenylboronic acid into a go-to building block for medicinal and materials science today.
Understanding 3,5-Difluorophenylboronic Acid
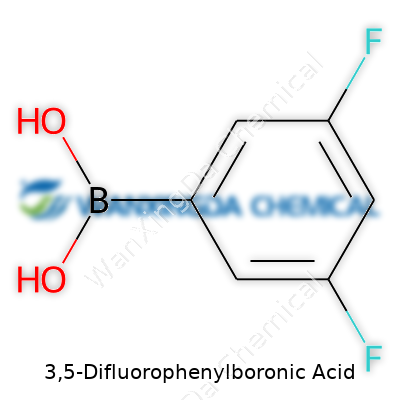
For those who haven't come across it in the lab, 3,5-difluorophenylboronic acid stands out thanks to two fluorine atoms tagging the aromatic ring at the 3 and 5 positions, plus that signature boronic acid group sticking out. This gives it a balance: enough reactivity for coupling reactions but also a unique chemical fingerprint, since those fluorines push and pull electron density in a way that plays out during synthesis. As an off-white or pale yellow powder, it comes in handy not just for classic organic chemistry, but for anyone building up fluorinated drug scaffolds or next-gen polymers. I’ve seen well-labeled jars of it tucked in university stockrooms, signaling a crucial role in research beyond just textbooks.
Tangible Attributes: Physical & Chemical Properties
Telling apart boronic acids often comes down to the details: melting point, solubility, and how they play with water and organic solvents. 3,5-Difluorophenylboronic acid, thanks to its twin fluorine atoms, brings a distinct melting point—typically sitting between 170–175 °C—sturdy enough to survive hot reactions, soft enough to handle without special gear. It’s more soluble in polar organic solvents than in plain water, mostly because those fluorines discourage too much hydrogen bonding. Plus, the acid has a knack for forming stable boronate complexes, a property the Suzuki coupling crew relies on. Unlike completely nonpolar aryl boronic acids, this one dissolves just well enough in DMF or DMSO to make purification and reaction setup less of a headache.
Technical Specs: What Ends Up on the Label
Making sense of a chemical like 3,5-difluorophenylboronic acid usually means reading the fine print. Chemists want to know purity (often above 97 percent for research), CAS number, and whether impurities lurk that could throw off a reaction. Some labels mention the moisture content, since boronic acids suck up water over time, messing with stoichiometry if ignored. Color and appearance aren’t just for show, either. A yellow tint could warn of oxidative degradation. The label rarely tells the whole story, but in my experience, a bottle that comes with a full analytical report—NMR, elemental analysis, even LC-MS—cuts down surprises.
Preparation: Getting from Theory to Flask
Most labs start from a difluorinated benzene (often 3,5-difluorobromobenzene), attack it with a metal-halogen exchange (think n-butyllithium), and then quench the whole thing with trimethyl borate or another boron source. After hydrolysis, you end up with the prized boronic acid. It’s a route that borrows from the classics, though tweaks matter: temperature control, order of reagent addition, and how you handle moisture make or break yield and purity. The method reflects a broader trend—organic chemists still rely on methods crafted half a century ago, but each new derivative demands its own dance steps. Having tried prepping aryl boronic acids, I can say patient crystallization and careful drying beat brute force every time.
Chemical Behavior: Reactivity and Modifications
Reactivity in boronic acids pivots on three things: the aromatic core, the substituents, and the boronic acid group itself. Throwing fluorines on the ring shifts the electron landscape, boosting the acid’s resilience in some reactions and fine-tuning its pairing behavior in Suzuki coupling. I’ve seen colleagues swap the boronic acid group for esters or trifluoroborate salts to improve stability or alter solubility mid-synthesis. Even outside coupling reactions, the molecule gets transformed into sensors or polymerizable units by simple tweaking. In the hands of a skilled chemist, it goes well beyond just “an ingredient”—it’s a dynamic tool shaped by fluorine’s electron-withdrawing power.
Synonyms and Product Names: Speaking Chemist’s Language
Anyone ordering chemicals online knows names can mean confusion. 3,5-Difluorophenylboronic acid picks up plenty of aliases—sometimes just 3,5-Difluoro-phenyl boronic acid, or 3,5-DFPBA, or even by its registry number when suppliers want to keep things straight. Confusion creeps in if suppliers drop or rearrange “di.” Sticking to standardized naming, like IUPAC, and cross-checking the CAS number avoids mix-ups that melt budgets and kill project timelines. In my own ordering mishaps, the registry number often spells the difference between success and a month’s wait for the right powder to show up.
Standards for Handling and Safety
Nobody enjoys emergencies in a chemistry lab, so a bit of planning goes far. Most boronic acids edge on irritation if inhaled or touched, and 3,5-difluorophenylboronic acid is no exception. Proper gloves, eye protection, and decent airflow keep risks minimal, though I’ve seen quick shortcuts lead to skin reactions and bad workdays. Good training and habits protect not only the user but the whole building—no one wants airborne acid dust or poorly stored chemicals cropping up in shared fume hoods. From an operational point of view, labeling, secondary containment, and careful inventory matter more than any regulatory checklist; bad storage ruins good reagents and can spark real hazards.
Practical Impact: Where Chemistry Meets Real Needs
Research labs crave molecules that unlock new transformations, and this boronic acid sits front row in building fluorinated aromatics—a motif popping up everywhere in modern pharmaceuticals and agrochemicals. Fluorines crank up bioavailability, modulate metabolism, and tune receptor binding, making this acid a regular on the medicinal chemistry bench. It's also a workhorse in materials science. Fluorinated monomers make polymers with enhanced resistance, unique dielectric properties, and the kind of performance important in electronics manufacturing. I’ve watched researchers pour months into coupling reactions trying to build target molecules; a reliable supply of specialty boronic acids like this one greases the wheels.
Pushing Frontiers: Ongoing Research and Development
The chemistry community isn’t stopping with old routes and simple uses. Latest research looks at faster, more sustainable syntheses and clever modifications for boronic acid-based drugs, sensors, or catalysts. Green chemistry ambitions push scientists to replace organolithium steps with milder, less wasteful reagents. Peer-reviewed studies keep expanding the known reactivity, stretching coupling methods beyond what Suzuki or Miyaura probably imagined decades ago. The acid’s track record attracts new ideas for environmental sensors, enzyme mimics, and more.
Toxicity: Weighing Risks and Knowing the Unknowns
While boronic acids generally escape the hazard spotlight, the toxicology of many derivatives flies below the radar. Regulators like to see thorough studies before giving broad approvals, and 3,5-difluorophenylboronic acid stands as a case where research remains ongoing. Early data shows low acute toxicity, aligning with its chemical siblings, but fluorinated compounds sometimes show persistence in the environment or bioaccumulate, so skipping proper waste handling misses the bigger picture. I tend to keep a watchful eye on new publications; more data will help pin down safe concentration limits, exposure durations, and waste pathways so labs and industry can close risk gaps before they open.
Momentum for the Future
Boronic acids like 3,5-difluorophenylboronic acid don’t just disappear—demand keeps rising, fueled by everything from new drug platforms to next-generation electronics. If labs and industry can shrink environmental footprints, find higher-yield routes, and grit through safety questions with real data, the molecule promises to stay relevant. My own take is that education, thoughtful supply chains, and investment in greener, scalable processes will do more to keep this tool on the research bench than flashy press releases. Chemists know a reliable reagent can change the pace of innovation; this acid keeps proving its worth in academic and industrial hands alike.
Why the Formula Matters
There’s a certain satisfaction in decoding a chemical formula and knowing exactly what goes on at the molecular level. With 3,5-Difluorophenylboronic Acid, its chemical formula—C6H5BF2O2—holds a particular appeal for researchers and chemists alike. This compound sounds a bit niche, but it has found a comfortable spot on the shelves of chemical supply rooms and R&D labs worldwide.
The Structure at a Glance
The formula might look intimidating at first sight, but each symbol tells a part of the story. The six carbons mark the backbone of the benzene ring, not the kind people picture in basic high school experiments, but one with two fluorines stuck at the 3 and 5 positions. Boron and two oxygens finish the story, forming the boronic acid group. Anyone who’s spent time with organic syntheses knows how groups like these get swapped in and out to build larger molecules.
Fluorine’s Impact
Fluorine’s relatively rare compared to other substituents, and it brings a set of unique properties. Its small size and impressive electronegativity change the way electrons move in the benzene ring. That’s the charm—fluorinated compounds show new patterns of reactivity and stability, which attracts chemists looking to tweak how molecules behave. For pharmaceuticals, agrochemicals, and advanced electronic materials, introducing a difluoro motif can change the game.
What Boronic Acids Bring to the Table
Boronic acids, including 3,5-difluorophenylboronic acid, have become essential tools for Suzuki-Miyaura coupling—a name that probably rings bells in synthetic labs everywhere. This reaction allows scientists to stick two aromatic rings together, almost like snapping Lego blocks. It’s simple in principle, powerful in execution, and has fueled growth in drug discovery and materials science. The ability to attach functional groups precisely has enabled researchers to build more effective drugs, test new catalysts, and even produce organic light-emitting diodes more efficiently.
The Push for Safer, Smarter Chemistry
While the chemistry sounds promising, safe handling is just as important as molecular innovation. Boronic acids can sometimes slip through the cracks of regulatory frameworks, given how new classes of these compounds emerge every year. There’s a responsibility to ensure that anyone using them—grad students, industry chemists, or even hobbyists—does so without risks to health or the environment. Updated guidelines, accessible data on toxicity, and mandatory training can reduce the chances of lab accidents and environmental spills.
Paving the Way for Sustainable Synthesis
Demand for sustainable chemistry grows louder each year. Some boronic acid syntheses involve hazardous reagents and produce high waste. Greener alternatives have started to appear—solvent-free reactions, water as a reaction medium, and more robust recycling of catalysts have become real options. Sharing success stories, publishing protocols that emphasize safety and efficiency, and rewarding labs that cut down on waste can help move the entire field forward.
The Takeaway
The formula C6H5BF2O2 doesn’t just represent a molecule; it stands for an opportunity for innovation. Chemists have figured out how to use small tweaks in structure for big advances in medicine and materials. Efforts aimed at safe use and sustainable synthesis will make sure that these advances don’t come at too high a cost. Sharing knowledge openly and encouraging responsible research goes a long way in shaping the future of chemistry.
A Crucial Building Block in Drug Discovery
For anyone who's spent some hours hunched over a lab bench, 3,5-Difluorophenylboronic acid is more than a line in a catalog—it's a quiet enabler in the world of drug development. Medicinal chemists see compounds like this as tools to stitch together new molecules that push the boundaries of what's treatable. The boronic acid moiety makes it a go-to partner for Suzuki-Miyaura cross-coupling reactions. These reactions matter a lot, not just because they're efficient, but because they let researchers blend pieces of molecular "Lego" to create complex pharmaceuticals.
Take kinase inhibitors, which keep getting attention due to their role in cancer therapies. The insertion of difluoro substitutions often brings better metabolic stability and changes the way the drug interacts with its targets. It's a bit like adjusting a car’s engine to run efficiently on a different grade of fuel—the small tweaks at the molecular level can give a drug longer life in the body or lower its side effects. Over the years, fluorinated phenylboronic acids have become essential ingredients in the toolkit for designing the next wave of targeted medicines.
Powering Agrochemical Innovation
Agriculture leans on chemistry more today than it did in my grandparents' era. Farmers face weeds and pests that refuse to cooperate. To keep crops safe without wrecking the environment, agrochemical companies fine-tune herbicides and insecticides down to the atom. Compounds like 3,5-Difluorophenylboronic acid let them tweak the activity and environmental profile of new formulations. The difluoro group isn't there for show—it flips the biological switch in ways a simple phenyl ring can't. If a new herbicide breaks through because it latches on to a weed enzyme more tightly, odds are good that a chemist made that happen in part with the right boronic acid building block.
Material Science and Electronics
With every generation of smartphones and solar cells, the push for performance boils down to the chemistry in the materials. Boronic acids help in making new kinds of polymers and organic electronic materials. The difluorophenyl group tweaks qualities like conductivity, thermal resistance, and how light interacts with a material. Organic light-emitting diodes, or OLEDs, often owe their glow and efficiency to clever molecular design—sometimes hatched with starting materials drawn from the family of difluorophenylboronic acids. In labs focused on creating molecular sensors or next-gen batteries, this compound offers a head start because of its versatility in building custom frameworks that can trap, transfer, or detect molecules.
Challenges and Responsible Use
As someone who values ethical science, I need to bring up the flip side: waste and safety. Handling boronic acids and fluorinated compounds calls for a steady hand not just in the lab, but all the way through the chemical’s life cycle. Industrial users face questions around disposal and toxicity. The fluorine atoms make these compounds tough, which can lead to persistence in the environment. Researchers look for greener solvents and more efficient reaction processes to reclaim and reuse what would otherwise end up as hazardous waste. University labs can switch to microscale reactions, cut down on stockpiling, and invest in training on chemical stewardship.
Driving Progress One Reaction at a Time
In my experience, the most valuable reagents are the ones that quietly appear in synthetic routes again and again. 3,5-Difluorophenylboronic acid fits that bill. Its value comes from the way it lets chemists craft molecules tailored to solve real-world problems. By keeping an eye on both performance and sustainability, the scientific community can get the most out of this slender white powder—whether it's speeding up a new drug’s journey to market, protecting crops, or building the future of electronics.
Cutting Through the Chemistry Jargon
Talking chemistry outside a lab feels like translating a different language. Most folks, even if they handled acids in high school, rarely think about molecules once the bell rings. Labels like "3,5-Difluorophenylboronic Acid" sound distant until they pop up in news or product labels, or you meet someone designing new drugs with them. Some people meet it through research, others through curiosity about what builds their medicines or electronic gadgets.
The question comes up: what is the molecular weight of this compound? The answer feels straightforward for chemists, but for everyone else, the real meat sits in what that number actually means. This compound’s molecular weight hits 173.94 grams per mole, by the numbers on the periodic table. But facts on their own don’t land a punch without a story.
Why a Simple Number Packs Such a Punch
Every element—carbon, hydrogen, boron, fluorine, oxygen—brings its own count to the total. Chemists run these numbers in their heads like bakers measure flour, but most don’t see how crucial it gets. A single proton here or a neutron there turns a safe vitamin into a toxic mess. This number 173.94 isn’t just a dry calculation; it changes how you ship, store, or even test a sample in the lab. Anyone who’s tried to mix up the wrong batch from a textbook error learns this fast. In chemical research, a faulty weight can mean failed experiments, wasted grant money, and missed deadlines—not just spilled powder.
From Chemistry Bench to Real World
Molecules like 3,5-Difluorophenylboronic Acid don’t exist just for practice. They anchor themselves in the real world, showing up in the background of medicine creation and new materials. Drug designers love boronic acids for making more effective cancer treatments and diabetes drugs. In that industry, every step forward means testing, scaling up, and sharing results in a way anyone, not just PhDs, can understand. Relying on shaky math, or skipping over numbers, lets bad science slip in the back door. People’s health is on the line.
In the hands of everyday researchers and students, exact molecular weights keep the wheels turning. I remember spending late nights double-checking calculations on old scrap paper, anxious about missing a decimal. A wrong weight meant a late-night panic call to a colleague, and once, an entire lab group tossing our batch of benchtop products. Out there in industry, the same error means lost sales, customer complaints, or dangerous products.
Raising the Bar for Accuracy
Better accuracy comes from careful education and easy-to-read data. Universities and journals now demand double and triple checks of every calculation—no shortcuts. Open-access resources empower students and pros to check key facts quickly; fewer locked doors between a wrong answer and public safety. Sharing reliable information links directly to trust, whether you’re a supplier, a teacher, or someone developing the next treatment for a tough disease. Staying transparent, citing trustworthy sources, and going back to basics—checking your work—keeps chemistry honest.
Why Chemistry Demands a Thoughtful Approach to Storage
I’ve spent enough time in both academic and industrial labs to know the smallest mistake in storage can mess up an experiment or put people at risk. Chemical safety reaches past lab coats and goggles—it begins long before anyone measures a single milligram. On the bench, 3,5-Difluorophenylboronic Acid looks like any ordinary white powder. Its subtle properties, though, serve as a reminder that even commonplace chemicals need some respect.
Handling the Real Stuff, Not the Theory
Some researchers roll their eyes at all those admonitions in safety training, but they matter. Boronic acids, including this one, break down if left at room temperature with too much moisture around. Take it from someone who has watched an entire batch go clumpy because the desiccator failed—humidity doesn’t wait. Moisture can prompt chemical changes, impacting reactions or, worse, causing the sample to degrade beyond use. Every time I see those stubborn clumps, I remember how essential it is to dry containers well and never let the cap linger off.
The Importance of a Controlled Environment
Strict storage conditions exist for a good reason. Researchers at leading institutes point out that some boronic acids turn into sticky residue if left exposed. 3,5-Difluorophenylboronic Acid should stay sealed in airtight bottles, preferably in a desiccator filled with fresh drying agent. I always check the color of my desiccant—no one wants water sneaking in on their shift.
A lot of small labs lack dedicated climate-controlled vaults, so a basic refrigerator works as a backup. Low temperature slows down chemical degradation. The key: don’t use any fridge with food or drinks. Cross-contamination isn’t just a health violation; it threatens sample purity. I once worked in a shared university setup where someone put a salad next to a vial of methylated reagent—never again!
Labeling and Rotation Make a Difference
Any time a container gets opened, I mark the date. Researchers trust results only when they know the chemical hasn’t sat around absorbing water for years. Rotating inventory—using older stock first—cuts down on wasted reagents. It sounds dull, but those habits protect science from frustrating dead ends caused by “mystery degradation.” I teach every new lab member to check expiration, date, and condition on every container.
Potential Problems—And Straightforward Fixes
Accidental spills or leftover powder sprinkled on a balance happen more than anyone likes to admit. Cleaning up right away, disposing of wipes or paper towels in a solid chemical waste bin, and washing hands prevents spread. An extra glove or two goes a long way. I always tell colleagues to keep a spill kit handy—this stuff won’t bite like acid, but there’s no reason to take chances.
Storage errors drive costs up and slow research. Every time a reagent fails, the lab not only spends money buying more, but loses time troubleshooting. Building a “storage culture” matters as much as updating protocols. Awareness, daily checks, and shared responsibility transform lab safety from a bureaucracy into basic citizenship for researchers.
Smart Habits Build Better Science
Storing 3,5-Difluorophenylboronic Acid isn't glamorous, but reliability starts here. Proper containment, keeping things dry, cool, and labeled, and never mixing it with food or loose items—these actions let researchers trust their materials enough to push science forward. Labs don’t run well on shortcuts; they thrive on respect for small details. That’s a lesson learned over years, one powder jar at a time.
The Role of Chemical Safety in Everyday Lab Life
Pulling on a lab coat every morning, you never really know which substance will end up spinning in your flask by noon. For researchers, chemists, or just the curious who venture into the world of organic synthesis, chemicals like 3,5-difluorophenylboronic acid pop up more often than a cup of coffee. Recently, there’s more talk around its hazards and handling and this deserves real attention.
What Makes 3,5-Difluorophenylboronic Acid Tick
This chemical belongs to a family prized for their role in Suzuki coupling—a classic way to build carbon frameworks for medicines, materials, and other useful molecules. Its use isn’t exotic; a lot of drug labs, academic benches, and industrial floors have some version of boronic acid stashed on the shelf. Still, a complicated molecular structure with boron, fluorine, and a phenyl ring shouldn’t be dismissed as harmless.
Exposure Risks and Practical Hazards
Once you start thumbing through its safety literature, several risks show up fast. Dust from 3,5-difluorophenylboronic acid can irritate eyes, skin, and the lungs. Accidentally inhaling fine powders or leaving bare skin exposed can trigger discomfort or an allergic reaction. The consequences sometimes go beyond a minor rash or a sneeze. Most aromatic boronic acids aren’t acutely toxic in small amounts, but chronic exposure isn’t something to shrug off either.
In my own grad school days, I’ve seen chemicals with similar profiles spark more trouble from carelessness than from inherent toxicity. A snappy lesson came from watching a colleague brush boronic acid powder off their sleeves—then rub tired eyes half an hour later. That oversight led not to research advances but to a trip to health services. These acids aren’t household bleach, but skin or eye contact is a mistake most newcomers make only once.
Environmental Impact and Waste Concerns
Organic boron compounds rarely break down quickly. Pouring waste down the drain won’t win you friends with environmental officers or regulators. Careless disposal can help small molecules linger in the environment, which over time, could present risks to aquatic life. Most responsible labs now keep proper waste cans and log all their discard. Skipping these steps comes with real penalties, both for lab budgets and long-term safety.
Smart Strategies for Safer Handling
Safety in a chemical lab boils down to habits. Good goggles beat regret every time. Nitrile gloves—always. Fume hoods aren’t decorative—they suck up dust and vapors so your lungs don’t have to filter them. Once, after a marathon synthesis session, my nose itched from working outside a hood. Ever since, I refuse to weigh powder without proper ventilation. The lesson costs a few minutes, not your health. Store this compound carefully, away from moisture and excessive heat, as many boronic acids can decompose or react with water over time.
No chemical loses its risk just because it sits in a university fridge, waiting for a student to brighten a reaction flask. 3,5-Difluorophenylboronic acid is useful, not harmless. Using it right means reading its documentation, wearing gear every single time, running things inside a fume hood, and treating waste as a responsibility you owe the next person who steps into your workspace.