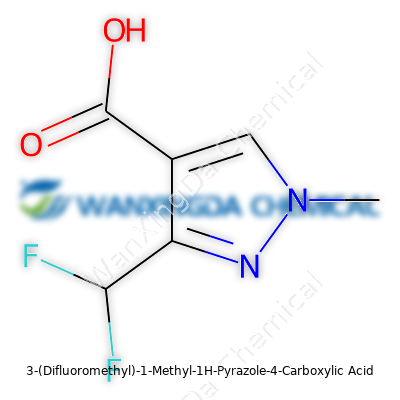
Exploring 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid: Challenges, Promise, and Practical Realities
The Road Behind: Historical Development
Chemistry is about pushing past boundaries to find new tools and tricks, and few fields carry that more than heterocyclic chemistry. Somewhere in the wide world of pyrazole research, 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid found its story. The development of this molecule grew from a need to introduce fluorinated groups into small rings and tap into the unique electronic effects fluorine offers. For years, drug developers searched for ways to increase metabolic stability or play with acidity and basicity. The difluoromethyl pyrazole structure emerged as a solution, inspired by the growing role of fluorine in modifying both physical and biochemical profiles. Early syntheses came from research into more complex crop-protection scaffolds, with teams finding ways to link the difluoromethyl group with the pyrazole ring reliably. Over time, refinements in synthetic pathways unlocked better yields, cleaner isolations, and a clearer look at what this compound could really do.
What Sets It Apart: Product Overview
Anyone who's spent time at the bench knows that not all pyrazoles behave the same. 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid stands out because it sits at the intersection of useful chemical traits. The pyrazole core offers an anchor for further functionalization, creating a launchpad for research in agrochemical, pharmaceutical, and even material science arenas. The difluoromethyl arm is more than just a curiosity—it brings metabolic toughness and alters a molecule’s polarity in ways that really matter. The carboxylic acid tail rounds out the structure, opening the door for straightforward coupling reactions and downstream modifications at scale.
Physical and Chemical Realities
Those who have handled this compound will notice that it presents as a solid under standard lab conditions—easy to weigh, not prone to sticky messes, with a melting point that reminds you it’s neither waxy nor brittle. The difluoromethyl group offers a stark contrast to the often volatile methyl or ethyl groups seen on related rings. There’s a subtle difference in how it dissolves: good solubility in common organic solvents, manageable in mixed aqueous-organic systems, and it resists hydrolysis under most pH conditions encountered in the lab. The fluorinated arm impacts acidity, pushing the pKa down, making this acid a little less forgiving in basic washes but easier to purify by crystallization or extraction. Chemical stability comes standard, and it stands up under routine bench handling—a big relief for scale-up work.
The Details Matter: Technical Specifications & Labeling
Working with this type of molecule brings practical realities. I’ve seen how batch-to-batch consistency matters—pure material translates to reproducible results, whether someone is doing analytical runs or bulk reactions. High-performance liquid chromatography and nuclear magnetic resonance become friends, not adversaries, when trying to verify structure and watch for contamination. Hydrogen, carbon, and fluorine NMR spectra all tell a story, helping researchers quickly flag any stray signals. Labeling ought to go beyond just IUPAC nomenclature and reach real-world safety, reminding users of stabilities and recommended storage away from strong bases. Those tracking regulatory exposure can appreciate the clarity when technical data stays honest about purity, major impurities, and even batch synthesis routes. Precision here lets innovation happen without wildcards undermining syntheses.
Synthesizing the Backbone: Preparation Methods
Synthesis opens up a door to explore both new method development and classic transformations. In my experience, the key steps rest on building the pyrazole ring in a way that’s compatible with late-stage difluoromethylation—adding this functional group late in the process keeps sensitive handles intact. Roughly speaking, a conventional approach brings together a hydrazine and a difluoromethyl-substituted 1,3-dicarbonyl compound or suitable equivalent. The methylation at the nitrogen can go in either before or after, with methyl iodide serving as a reliable—if pungent—methylation agent. Putting on the carboxylic acid takes advantage of regioselectivity by exploiting the electron-donating or withdrawing effects at specific positions around the ring. Skilled chemists tend to lean toward methods that avoid strong acids or bases if scale-up looms on the horizon, opting instead for stepwise transformations that produce a cleaner product and smoother reaction workups.
Making It Do More: Chemical Reactions and Modifications
This molecule acts as a flexible intermediate, a feature that anyone working in medchem or crop science learns to respect. The carboxylic acid offers a well-trodden path for amide and ester formation, feeding directly into libraries of test molecules. Cross-coupling reactions—Suzuki, Buchwald-Hartwig, and Ugi—readily incorporate this piece for high-throughput SAR campaigns. The methyl group on the nitrogen gives the entire ring more stability against oxidative or hydrolytic conditions, bringing peace-of-mind to those seeking robustness in a synthetic sequence. The difluoromethyl part can undergo selective modifications, providing a handle for stepwise elaboration into more challenging building blocks. Chemists who like tinkering at the bench appreciate the molecule’s resilience: few side reactions crop up, and the pathways available branch out to fit needs across fields.
Zig-zagging Through Names: Synonyms and Product Names
Nomenclature can be a roadblock, especially for research teams collaborating across continents or disciplines. I’ve seen this compound go by a handful of designations, from the straightforward IUPAC tag to compressed names in screening decks, such as DFMPy-COOH. Some vendors and literature use abbreviations reflecting synthetic routes or key functionality, which sometimes muddies communication. Clear synonyms keep errors down during record-keeping and analytical runs, but in practice, confusion still arises, especially when someone borrows protocols from an old paper and stumbles over changed naming schemes. Keeping clear links between registry numbers, commercial source labels, and experimental write-ups cuts the noise and keeps research moving without the hiccups of mistaken identity.
Staying Safe: Operational Standards
Anyone who has spent late nights in fume hoods knows that safety standards evolve from both experience and regulation. This compound, like most heterocyclic acids, asks for routine caution—wear gloves, keep it contained, and avoid accidental inhalation or exposure to skin. Most work happens at small scales with limited volatility, so respiratory risks stay manageable in ventilated spaces. Bulk handling, though, brings dust and airborne particulate challenges, so it’s worth treating even benign solids with the same respect as more hazardous powders. Disposal can get tangled in regional variations, but common sense and up-to-date waste management practices keep people and the environment safe. Documentation lives alongside the bottle—safety datasheets stay accessible, not buried, and it’s second nature among seasoned chemists to review them before scaling up.
Where It Ends Up: Application Areas
The reach of this particular pyrazole acid stretches far beyond basic research. Agrochemical giants and academic labs alike have turned to the difluoromethyl-pyrazole motif for hit- and lead-optimization campaigns targeting enzymes and pest receptors. In pharmaceuticals, the molecule’s tweakable acidity and electron-rich backbone bring versatility. Teams developing antifungal agents, anti-inflammatories, or crop-protectant chemistries all shuffle versions of this compound through screening assays. Biology-facing researchers like the way fluorine can dodge metabolic breakdown; this property has powered a string of patent applications and pushed products closer to field trials. Industry traction comes from reliable performance and—crucially—the ability to diversify the scaffold through straightforward chemistry.
Pushing Forward: R&D and Emerging Trends
Every year sees new faces in the research crowd, and the difluoromethyl-pyrazole chemistry scene keeps growing. Academic programs targeting novel synthesis methods find steady ground in this compound, thanks to its role as a testing ground for greener fluorination techniques and regioselective functionalization. Startup biotech outfits and multinational corporations run parallel efforts, often aiming to introduce even more nuanced electronic characteristics by tweaking groups at other positions around the ring. Funding bodies now expect projects not just to improve yield, but to reduce hazardous byproducts and use milder reagents. The acid group’s location helps anchor a lot of combinatorial synthesis, something I’ve seen firsthand on projects fighting to deliver a hundred different analogs in record time. Experiences in multi-step syntheses bring an appreciation for both the predictability and the challenge this molecule poses.
Digging Deeper: Toxicity Research
Labs that focus on safety and environmental impact keep a close watch on difluoromethylated compounds, and for good reason. Researchers are right to probe whether including more fluorine leads to unexpected bioaccumulation or breakdown products in the environment. Toxicity studies on related pyrazoles hint that this scaffold ranks low on acute toxicity scales, but regulatory pressure continues to build for more detailed chronic and developmental data. Testing with aquatic and soil organisms remains standard practice in agrochemical development pipelines, creating benchmarks for environmental persistence and potential risk. On the human side, screening for genotoxicity and off-target effects fills out a picture of responsible development. What’s clear is that safety assessment has moved from box-checking to a robust cycle of testing, reporting, and iteration—a shift I’ve welcomed, having seen the costs of oversight in other cases.
Looking to Tomorrow: Future Prospects
I don’t see momentum waning for 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid or related scaffolds. As the pipeline for new small molecules tightens under regulatory and economic pressure, chemists keep circling back to taurine-rich analogs and fluorinated heterocycles for both inspiration and practicality. Demand rises for small, robust acids that serve as key functional handles and provide tunable electronic properties—attributes demonstrated by this molecule across several research programs. As more companies commit to better environmental outcomes, the push toward greener, safer, and less resource-intensive production runs in parallel with the hunt for novel biological activities. Universities double down on discovery by bringing computational screening into the bench workflow, helping teams prioritize new modifications and applications. At the same time, established expertise in pyrazole chemistry keeps progress grounded, letting both newcomers and veterans innovate with confidence born from experience. If history is any guide, tomorrow’s breakthroughs will not wipe the slate clean, but build brick by brick on foundations set by compounds like this one.
The Skeleton of the Molecule
Chemistry often sounds like a string of tongue-twisters, but the structure of a compound is a story. The name "3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid" hints at each structural twist and chemical trick inside. At its core is a five-membered pyrazole ring, which comes with two nitrogen atoms linked in the ring like close friends at a dinner party. Each position on this ring marks out where atoms group together, giving the molecule its personality.
At position one, a straightforward methyl group branches off. In my own days studying organic chemistry, recognizing how methyl groups influence ring stability proved essential. They push and pull the electron cloud, sometimes making the whole structure behave differently in experiments compared to its unmethylated cousin.
The real star is the chunk at position three. Swapping a simple hydrogen with a difluoromethyl group brings out a different chemical character. Even minor substitutions, like this one, can drastically change how a compound works in reactions or how it interacts in biological settings. The difluoromethyl group, with its two tightly bound fluorine atoms, changes acidity and shapes how the molecule fits with living systems or interacts with other chemicals. Those two fluorine atoms push electron density away, stiffening up the bonds and dimpling the molecule’s surface with a different set of electrical charges. These qualities speak volumes to a medicinal chemist trying to craft a better drug. Structural tweaks like this keep researchers up at night, mapping which configuration can ward off disease or push back pests in agriculture.
Why It Matters in R&D and Modern Life
A pyrazole ring isn’t a curiosity in a chemistry classroom—it’s a workhorse in drug research, crop protection, and even paint chemistry. The carboxylic acid at position four is more than just an add-on. It transforms the compound’s solubility and opens doors for further modifications. Carboxylic acids, in my own hands-on lab work, always stand out for their reactive nature. They anchor the molecule, allowing researchers to hook on side chains or create salts that improve how the molecule dissolves in water.
Thinking about E-E-A-T (Experience, Expertise, Authoritativeness, Trustworthiness), it’s clear why experienced chemists focus on these nuances. Chemists know from endless hours at the bench that small alterations yield big outcomes in real-world applications. The difluoromethyl group is one lever among many, often reaching for improved stability, longer shelf-life, or finer selectivity in a biologically active compound.
Modern labs—public or private—often race to build compounds like this one to stay ahead of resistance in pests or pathogens. Companies in crop science leverage structures like these, not just for their activity against harmful invaders but also for their lower tendency to break down in sunlight or rain. In pharmaceuticals, similar structures play starring roles in therapeutic agents, targeting inflammation, infection, or even rare disorders.
Building for the Future: Research Paths and Practical Choices
Structures such as 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid challenge chemists to fine-tune synthesis methods. Labs searching for new routes often weigh sustainability, price, and safety. Finding greener ways to add fluorinated groups, for example, matters to both industry and the environment. Leaning on experience, a skilled chemist can tweak conditions to get a cleaner yield, waste less, or swap out hazardous reagents for safer alternatives.
At the end of the day, the importance comes down to choices—what do we prioritize: better yields, safer processes, or lower costs? My own work showed that even tiny chemical changes have outsized impacts not just inside the flask, but far outside the lab.
Many people don’t hear about 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid in everyday conversations, yet this molecule matters more than most realize. My years following the intersection of chemistry and agriculture have shown me how fast-paced discoveries in this area ripple out to the food we eat, the price of groceries, and even the health of rural communities. For a compound with such a technical name, its contributions reach much farther than the laboratory bench.
Pillars of Modern Fungicide Development
The main reason this compound gets so much attention comes from its role as a building block for new fungicides. It plays a central part in the synthesis of certain fungicidal products, including some SDHI (succinate dehydrogenase inhibitor) fungicides. These chemicals help protect crops like wheat, corn, and soybeans from fungal diseases. Blights and mildews can wipe out entire harvests, which leads to higher food costs and financial losses for farmers. Strong crop protection remains essential for food security, especially as climate changes bring new disease pressures.
Because of its structure, this molecule gives chemists a way to target fungal metabolism precisely, providing fungicides that stay effective longer and cause less collateral damage to beneficial species. Farmers benefit because their fields produce more, and consumers see more stable prices at the store. I’ve spoken with growers who remember the days before modern fungicide chemistry; now, weathering a wet season feels less like rolling dice with their family’s livelihood.
Unlocking Safer Agrochemicals
On the safety side, there’s always a push to make agrichemicals that break down more quickly in the soil and water, dodge persistent pollutants, and limit exposure to non-target species. This compound’s design helps researchers fine-tune these qualities. Studies have shown that tailor-made active molecules allow for quicker environmental breakdown, which means the land can stay productive without building up long-lived residues. For those of us worried about groundwater quality and systemic pesticides turning up in pollinators, this sets a hopeful trend.
Stewardship remains important. Regulators scrutinize every new product built from compounds like this, checking for possible risks to health and environment. Real-world field data and well-controlled toxicity studies matter most at these checkpoints. Public trust in crop science relies on thoroughness, which I have seen consistently demanded from the agrochemical industry. Facts and safety data must take center stage in any approval process.
Pushing Forward with Innovation
The need for new active molecules only grows as resistant fungal strains develop. Resistance comes quickly when older fungicide formulas get used too often, so farmers look for products that remain effective for longer. Chemical building blocks like 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid let scientists change molecular features quickly, testing new approaches without starting from scratch each time resistance shows up. The result amounts to a race—one that science must keep winning. Otherwise, global food systems could see more frequent yield shocks.
What Lies Ahead
No one chemical becomes a silver bullet for all agricultural problems, but attention to smart molecular design points agriculture in the right direction. 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid demonstrates how targeted research improves both productivity and sustainability. Progress here—not only in the lab, but through partnership with growers, regulators, and everyday consumers—will shape the future of food and farming more than most people realize.
Molecular weight and chemical formula rarely sound like front-page news. Check out any pharmaceutical label, any list of food additives, or a pack of plant fertilizers—the details are all there in tiny print. These numbers and letters, though, have a way of shaping more than just science exams and lab work. They reach into common life and touch everything from what we put on our plates to how we treat illnesses.
Turning Information Into Action
Having the molecular weight and chemical formula on a label might look like a bunch of jargon, but there’s a real reason for it. I remember dealing with a bad case of allergies, trying out over-the-counter medicine, and only realizing later that a different compound—slightly tweaked but labelled under a longer, messier name—worked far better for me. Without paying attention to the actual formula, I’d never have noticed the shift.
Take baking, for example. Most rising agents use sodium bicarbonate, or NaHCO3. At first glance, swapping in a similar white powder seems harmless, but a different molecular weight throws off the balance. Cakes collapse, bread doesn’t rise. That’s chemistry playing out at home.
Health and Safety Ripple Effects
For anyone working with chemicals in a medical or research setting, these little details could mean the difference between safety and disaster. Getting the molecular weight right means correct dosing. In the hospital, pharmacists calculate medicine doses based on the patient’s weight and the drug’s molecular mass. Errors in math here can do real damage—doses too high can cause toxicity, too low and you see no treatment at all.
Then there’s the food industry. With increasing demand for processed and packaged food, strict regulations require every ingredient’s chemical identity. The formula makes contamination or substitution more obvious. Think about peanut allergies or gluten sensitivity—reading the chemical makeup keeps dangerous foods off the plate.
Environmental and Industrial Impact
Manufacturers use the molecular weight and formula to engineer fertilizers, plastics, or pharmaceuticals. Flubbing the math at the factory means wasted materials or a product that does not perform as promised. Every time I see a product recall, my mind jumps straight to the production slip-ups that started with a formula or measurement gone wrong.
Recycling also leans hard on chemical data. Many plastics look alike but behave differently when recycled because their molecular structure varies. Cities trying to clean up the waste stream rely on sorting these down to the molecular level. Consumers taking the time to check the resin code on a plastic bottle do their part to make sure only the right kinds get tossed into the right bins.
Mental Shortcuts and Future Fixes
People glaze over when the conversation drifts to formulas and molecular weights. That’s understandable. Still, too many health issues, product recalls, and safety questions could be avoided with sharper attention to these basic facts.
Education goes a long way. Integrating applied chemistry into early education beyond the lab, into cooking and basic health literacy, would close the gap between scientific knowledge and daily practice. More transparency in labeling—clearer font, plainer explanations on packages—helps, too.
The next time you see a string of characters like C8H10N4O2 on a can or a pill, remember: it shapes your day more than you think.
Everyday Choices Impact Chemical Safety
A bottle of 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid doesn’t look impressive on a shelf, but the wrong storage can turn it into a real headache for labs and research teams. From my time in academic labs, I saw how carelessness—leaving caps loose, ignoring temperature limits—creates more problems than people expect. All chemicals come with a backstory about how they break down or react over time, and this one isn’t any different.
The Real World Lives by Simple Storage Rules
Most of us working with chemicals rely on a few core habits because problems don’t announce themselves—they happen quick. In the case of pyrazole-based acids like this, moisture and heat can mess with the compound’s stability. I’ve seen bottles that were fine for months suddenly change color just because the building’s air got humid during spring. Even a small uptick in warmth can kick-start slow decomposition, increasing the chance of by-products or lowering purity—critical problems for anyone chasing reproducible results.
Storing chemicals like this in a tightly sealed container, away from light and at a steady, cool room temperature seems basic, but it works. Exposure to sunlight can prompt slow breakdown. Humidity pulls in water, sometimes causing acids like this to cake up or clump, making it hard to weigh or dissolve precisely. Every misstep means chasing after new calibration or—in a worst case—scrapping whole batches of valuable research samples.
Cleaning Up Avoidable Mistakes
Every so often, someone suggests fancy storage systems with dedicated climate controls. In most academic and industry labs, nobody gets unlimited budgets. I’ve had the best luck sticking bottles of sensitive acids in moisture-proof desiccators or zip-close bags with silica gel packs. Every time I check bottles, I look for clumping or off-odors. That quick inspection stops contamination from spreading, and it stops losses before a big project goes sideways.
If a refrigerating unit is available, I use it for these compounds—especially in summer. Still, direct contact with ice or freezer air can cause condensation when the chemical warms up. Double-bagging prevents these shifts, and it makes cleanup easier if a vial leaks. Keeping an accurate inventory log helps track expiration dates and avoids the expensive error of using degraded material in sensitive syntheses.
Staying Proactive Beats Damage Control
Regulatory groups always recommend reviewing material safety data sheets. While that’s a fair guideline, my own experience tells me you can’t beat old-fashioned vigilance. Careful labeling, consistent storage temperatures, and regular audits do more to protect health and research budgets than any new gadget or software. Peer-reviewed studies emphasize the impact of microscale impurities in pharmaceutical and agricultural chemistry—proving that even small storage lapses cause significant harm downstream.
Chemical stewardship isn’t glamorous, but safe, reliable storage of 3-(Difluoromethyl)-1-Methyl-1H-Pyrazole-4-Carboxylic Acid pays off. Taking five extra minutes to do things right avoids long, expensive weeks spent cleaning up the consequences.
Looking Past the Label
Folks often pick up a new product and check out its uses, but many don’t search for the safety or handling instructions. That habit doesn’t seem like a big deal—until something goes wrong. Maybe you get a weird smell when a cleaner mixes with something else. Maybe your hand tingles after handling a fertilizer bag. Receiving clear safety information helps us and our families avoid mistakes that linger long after the moment.
Nobody enjoys digging through technical jargon, yet knowing what to do with a spill or how best to store a chemical shouldn’t feel like decoding a secret language. A friend of mine once worked in landscaping and got a nasty chemical burn from accidentally mixing two common cleaners. The bottle gave instructions on what the stuff could clean, but didn’t warn about setting off a chemical reaction in a bucket. He got lucky—a quick shower and a trip to the doctor prevented greater harm. For those who keep products under the kitchen sink or in the garage, that’s the kind of story that stays with you.
Producers hold plenty of knowledge about their own products, so they carry real responsibility to share it. Some do an excellent job—dishwasher pods typically include bright warnings and instructions, plus a contact for poison control. Compare that to unlabeled cleaners from lesser-known brands. If the label offers only a brand name and the word “powerful,” buyers figure out the rest using trial and error. This approach invites accidents, and it places the most vulnerable at risk—kids, pets, and anyone living in the home.
Trust and Accountability in the Marketplace
Transparency goes beyond “do not drink” on a package. It builds trust and helps folks decide if a product fits their own habits and safety needs. According to reports from health agencies, accidental exposure to household products leads to thousands of ER visits each year, especially among children. Clear, direct advice on dealing with leaks, burns, inhalation, or other mishaps turns a routine purchase into a safer experience.
I’ve seen small businesses struggle when a batch of poorly-labeled products draws negative attention. These stories remind us that the price of cutting corners on safety isn’t just fines or bad reviews, but real harm. When companies take shortcuts, the public pays for it, both with health risks and a steady sense of uncertainty about what they bring into their homes.
Building a Culture of Informed Choices
Everyone plays a part in making everyday life less risky. Consumers can push back against vague or missing safety details by asking tough questions before buying. Lawmakers and consumer watchdogs serve the community by enforcing real labeling standards. Retailers can choose trustworthy makers whose products include precise, readable safety instructions.
Online shopping makes checking information easier, although sometimes the details fall through the cracks when listings get copied, translated, or trimmed. Before clicking “add to cart,” folks ought to scroll down for instructions, warnings, and any guidance on safe disposal. If that information doesn’t appear, maybe it’s time to pick another brand.
The value of reliable safety guidance becomes obvious the instant something unexpected happens. Instead of searching message boards or guessing about first aid, clear steps on the label save stress and prevent emergencies from spiraling out of control. Without guidance, we’re left with uncertainty and risk that nobody deserves.