Looking Deeper into 4,4'-Difluorobenzophenone: From Origins to Future Impact
The Rise of 4,4'-Difluorobenzophenone: How We Got Here
Chemistry often traces its big moments back to the dogged search for materials that stand up to the world’s harshest demands. 4,4'-Difluorobenzophenone emerged on the chemistry scene as a quiet key player in the broader push for advanced engineering plastics. The route started from classic benzophenone, an old chemical staple. Add two fluorine atoms at the four positions and suddenly the molecule takes on a new life – ready to fill gaps that early chemists didn’t know existed. Polyetherketones and their derivatives, with their signature toughness and chemical resistance, wouldn’t work without raw ingredients like this fluorinated benzophenone. It’s not nostalgia that makes this development stand out, but the way it pushed polymer science forward. The material arrived not with hype, but with substance, impressing researchers eager to push boundaries in aerospace, electronics, and beyond.
Stepping Inside the Molecule: What 4,4'-Difluorobenzophenone Is and Can Do
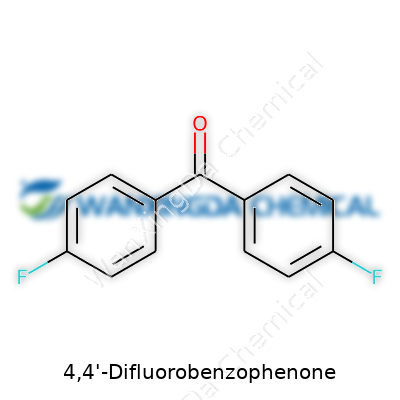
Holding up a vial of 4,4'-Difluorobenzophenone, you’d see a pale, flaky solid, not much to distinguish it from other crystalline powders at first glance. But that’s where the similarity stops. The compound packs a punch at the molecular level, thanks to the placement of those two fluorine atoms. Chemists see benzophenone as a starting block; swapping out hydrogens for fluorines cranks up the stability and shakes up the way the molecule reacts. It doesn’t just step into the role of a building block — it does so with real impact, letting materials made from it outlast, outperform, and outshine the competition. The chemical backbone is nothing fancy: a ketone core with phenyl rings flanked by fluorines at strategic points. In the right hands, it’s this simplicity that enables transformation.
The Backbone Facts: Physical and Chemical Terrain
You don’t need to be a chemist to notice that fluorine changes everything. 4,4'-Difluorobenzophenone holds up under serious heat — melting at temperatures well above what you’d see with the original benzophenone. With a boiling point that pushes the boundaries, this compound seldom gives in to volatility or breakdown, even when applications take things above the comfort zone for most organics. Toss in solvent resistance and strong belts of chemical stability, and it’s little wonder why polymer companies flock to it. Even with its higher melting point, it still dissolves in several organic solvents, which keeps processing manageable. These hard facts translate directly into tougher, longer-lived plastics used in places where replacement just isn’t an option.
On the Label: Technical Realities
In the trenches, a chemist wants details: purity matters, contaminants can spoil an entire batch. Producers typically ensure 4,4'-Difluorobenzophenone reaches customers with purity levels that don’t leave room for doubt. Color, melting point, and even trace water content draw scrutiny, since the wrong impurity can undermine a whole line of polymers down the road. Packaging usually aims to protect against moisture and light, echoing decades of lessons learned from busted batches and lab mishaps. Those who use it know the stakes, from drum labeling to transfer lines in the factory — everything is about keeping the molecule reliable, repeatable, and understood by those who open the container next.
Chemistry in Action: Making and Shaping the Molecule
Getting from raw starting materials to a clean batch of 4,4'-Difluorobenzophenone leans heavily on skilled synthesis. Many routes aim for efficiency, but most settle on nucleophilic aromatic substitution, pitting fluorobenzene against benzoyl chloride or related benzoic acid derivatives in the presence of clever catalysts and bases. This approach lets the right bonds snap into place, pushing the fluorines exactly where they’re needed. From there, a handful of purification steps remove unwanted byproducts and leave the prized ketone ready to perform. Labs seeking new flavors or modifications often experiment with reaction tweaks, protecting groups, or alternative fluorination strategies in pursuit of better control, higher yields, and even cleaner end products.
Reactivity and Customization: Pushing Beyond the Standard
Once you have 4,4'-Difluorobenzophenone, the doors swing open for a surprising menu of modifications. This isn’t a molecule content to rest on its initial structure. The carbonyl group and activated aromatic rings welcome further chemistry, from reduction and halogenation to cross-linking and polymer formation. Some research groups have explored how to dial in new properties by shifting the position or count of fluorine atoms, or by chaining the core benzophenone with other functional groups. Real progress often bubbles up when someone pushes beyond the “usual” — and with this molecule, the range of possible chemistry proves that out.
The Many Names: Navigating Chemical Synonyms
Chemical names can trip up even the best-prepared scientist. In literature and supply catalogs, 4,4'-Difluorobenzophenone sometimes appears under a mix of formal and common synonyms — from 4,4'-Difluorodiphenyl ketone to p,p'-Difluorobenzophenone. Knowing each name and its variations keeps confusion at bay, especially when tracking down safety data or hunting for the right grade. Companies and regional catalogs tack on their own identifiers, further underlining the need to cut through the clutter and make sure the molecule matches the need.
Staying Safe: Handling and Safety Realities
Chemists and line operators don’t get a free pass on safety here. Like many fine powders, 4,4'-Difluorobenzophenone can irritate the respiratory tract if inhaled or cause trouble with skin contact. Labs and factories stay alert, relying on gloves, goggles, and careful workspace ventilation to tamp down risks. Material safety data sheets over the years have mapped out emergency steps and outlined proper disposal, reflecting both regulatory requirements and the real-world lessons from accidents, near-misses, and ongoing toxicity research. You come to respect the substance — not out of fear, but out of a hard-earned trust in standard protocols. The right guardrails, drilled into staff and revisited each year, make for workplaces that aren’t just compliant but confident.
Where It Matters: Applications in the Real World
Step into any industry that craves resilience, and traces of 4,4'-Difluorobenzophenone likely turn up under the hood. Poly(aryl ether ketone) plastics, like PEEK and PEK, rely on its unique structure to create lightweight parts that survive heat, friction, and corrosive chemicals. Aerospace engineers use these polymers in connectors and insulation that withstand brutal cycles of temperature and stress. Electronics designers value its insulating and mechanical strengths, helping them pack power into smaller, tougher packages. Even down in oil exploration gear, where equipment faces pressure and chemical attack, the molecular backbone forged by 4,4'-Difluorobenzophenone keeps tools running longer, needing less maintenance. Decades of product testing show this molecule turns abstract chemistry into durable, day-to-day reality.
Advancing the Science: Research and Development Paths
Research on 4,4'-Difluorobenzophenone doesn’t stand still. Teams push boundaries by engineering even tougher derivatives, blending in new atoms or subgroups to fine-tune properties. I’ve seen researchers tweak polymerization conditions or explore blendable variants for 3D printing, aiming to bring precision plastics into low-volume, high-performance use cases. Tuning crystallinity, investigating copolymer combinations, and mapping degradation under decades-long simulated use fills academic journals every year. These experiments go beyond curiosity — they respond to relentless market pressure for lighter, thinner, greener materials in everything from medical devices to satellites.
Health and Safety Research: Toxicity Takes Center Stage
No one who works with chemicals forgets the human and environmental questions. Early uncertainty about the risk profile of fluorinated benzophenones prompted deeper studies. Toxicological testing maps the molecule’s impact at the cellular and organism level, measuring acute and chronic risks. Animal models and cell-based assays have flagged the need for continued vigilance: while acute toxicity remains low compared to heavier industrial chemicals, chronic exposure stories drive regular reviews and safety training. Environmental fate and the slow creep of persistent organics into waterways and soil sparked debates on regulation and waste management. Smart companies invest in ongoing monitoring and waste stream controls, sending a message that industry doesn’t have to mean neglect.
The Road Ahead: Opportunities and Challenges
Innovation rarely moves in a straight line, and 4,4'-Difluorobenzophenone’s journey still leaves room for surprise. Material scientists hunt for substitutes with even lower environmental impact and higher recyclability, while weighing the loss of performance that makes this compound special. Regulatory scrutiny of fluorinated substances continues to climb, forcing chemists to track downstream effects, disposal methods, and long-term environmental footprints. At the same time, unmet needs in sectors like energy storage, membrane technology, and medical devices dangle major prizes for those who adapt or rethink traditional chemistry. Those of us who track these changes know real progress will take honest work — tight partnerships between chemists, engineers, health experts, and policymakers, grounded in world-class science and an eye for practical results.
Chemicals With a Purpose
Walk into any electronics store or open up the inside of a car dashboard, and you’ll see plastics everywhere. There’s a reason some plastics hold up in heat, cold, and over years of use, while others crack or fade. Certain chemicals set the tone for these differences, and 4,4'-Difluorobenzophenone helps make that possible. By adding specific properties to polymers, this compound has shaped how engineers build our electronics, cars, and even medical devices.
The Backbone of Tough Plastics
No one wants an airplane window that grows brittle over time. This is where specialty plastics come into play. Polyetheretherketone—often called PEEK—leans on 4,4'-Difluorobenzophenone for its reliable performance. As a monomer, this chemical sets the stage for plastics with exceptional strength, resistance to heat, and a low chance of reacting with chemicals it touches. That means a medical implant can stay safe in the body, while connectors in electric vehicles can face the heat and vibrations of daily use.
Big Role in Electronics and Machinery
The growth in handheld devices, wearables, and smart appliances continues to feed demand for materials that don’t just melt or deform the moment they get a little warm. 4,4'-Difluorobenzophenone lends itself to the creation of polymer films and coatings that insulate circuits or add tough, thin layers to sensitive parts. Battery makers know that certain plastics stand between a safe phone and a potential meltdown, relying on this one molecule to manage that balance.
Green Chemistry: The Elephant in the Lab
Any chemical with a foot in major industries deserves scrutiny. Safety matters—not just for people who turn 4,4'-Difluorobenzophenone from a lab recipe into a tanker shipment, but also for the people who end up handling these plastics every day. Incidents involving contaminated water or accidental worker exposure underscore a need for tight controls. Environmental and health agencies in the US, Europe, and Asia ask companies to follow strict guidelines, especially with chemicals that could break down in unpredictable ways.
Solutions That Stick
The push for cleaner chemistry keeps gaining pace. Chemical suppliers and big manufacturers watch for regulations and come up with ways to recover or recycle waste—turning off the tap before toxins reach streams or landfill sites. Creation of closed-loop recycling systems and safer alternatives for solvents stand out as strong steps forward. Some labs study biodegradable substitutes or new processing tricks that trim emissions without losing the heat resistance or strength plastics need in demanding jobs.
A Personal Take
Experience tells me the reputation of a product can hinge on small details in its recipe. A smartphone case that shatters under pressure feeds customer complaints and costs a company down the line. My own interest in environmentally sound design always reminds me to dig into what’s beneath the surface. Materials built with 4,4'-Difluorobenzophenone don’t just make things last; they’re a reminder that every material choice comes with a tradeoff—balancing performance, health, and environmental impact. Watching companies steer toward transparency and new methods gives hope that chemistry can keep pace with society’s rising standards.
Breaking Down a Double-Ring Molecule
When I first started learning about organic chemistry, I couldn’t help but marvel at how two simple rings could give rise to all sorts of interesting properties and uses. 4,4'-Difluorobenzophenone is one of those molecules that stands out to chemists and industry professionals for a reason. Here, you’re looking at a molecule with a core that connects two benzene rings with a carbonyl group. What matters is where those rings pick up their fluorine atoms—both at the “4” positions, which means directly across from where the rings meet the central carbonyl.
Why Chemists Care About Structure
Molecules like 4,4'-Difluorobenzophenone don’t just look good on paper. The presence of fluorine swaps out hydrogen, and the shift shapes the behavior of the whole molecule. In this case, those two fluorines, one on each benzene ring, push the molecule toward increased chemical stability and make a big impact on its reactivity. Chemists rely on these tweaks; they’re not decorating molecules for the fun of it.
Anyone who has made or used a polymer that needs to resist heat and chemicals will have run into benzophenone derivatives. Swapping in fluorine at these specific spots changes how the material behaves. Polymers that include 4,4'-Difluorobenzophenone resist solvents better and hold up under heat, and this isn’t random chance—it’s the result of molecular engineering. Fluorine forms stronger bonds with carbon, making the rings tougher and less reactive to things that would typically break down organic molecules.
Industry Use and Environmental Impact
Out in the real world, you’ll see this molecule most in the manufacture of high-performance plastics. Think about electronic parts, aerospace components, or anything that demands strength while facing heat or chemical exposure. Having worked in labs where plastics crack, warp, or degrade, I’ve come to appreciate how attention to molecular structure solves practical problems. Engineers and scientists love fluorinated benzophenones for their reliability.
But the use of fluorinated compounds isn’t without rash consequences. Fluorinated chemicals have drawn global scrutiny because some can build up in the environment and the human body. Not all fluorinated molecules cause the same issues, but the industry must keep its eye on responsible manufacturing and disposal. As someone who cares about both chemistry and the planet, I look for ways to minimize unnecessary use of persistent fluorinated substances. Close monitoring, development of less persistent alternatives, and transparent reporting are crucial here. Nobody wants the benefits of better plastics to come at the cost of contaminated water or health risks down the line.
Solving Tomorrow’s Problems Today
Building safe, durable materials is a balancing act. Scientists studying 4,4'-Difluorobenzophenone and its relatives need to run ongoing risk assessments, refine processes, and keep learning from fresh research. New recycling and waste treatment technologies help address the challenges of persistent synthetic chemicals. By thinking through the way molecules like 4,4'-Difluorobenzophenone are made, used, and discarded, the community can find ways to enjoy technical progress without sacrificing environmental integrity.
Responsibility in the Lab Isn’t Optional
Every chemical tells a story, and 4,4'-Difluorobenzophenone has its own hard-learned lessons. This compound pops up in research labs, polymer manufacturing, and specialty chemistry projects. Its applications drive innovation, but the flip side never loses importance: safety. I’ve seen seasoned chemists handle powerful oxidizers like they’re just one more bottle on the shelf, only to end up regretting loose gloves or ignored ventilation hoods. With 4,4'-Difluorobenzophenone, a little neglect goes a long way – mostly not in your favor.
Goggles and Gloves Are Non-Negotiable
From day one, lab instructors hammer home the basics: eye protection, nitrile or neoprene gloves, and a well-fitting lab coat. My early days in the lab, I watched a classmate dismiss those rules while handling aromatic compounds. Minor splashes turned into real health scares – rashes, coughing fits, missed work. Exposure to fluorinated compounds doesn’t bring instant disaster, but long-term effects on skin and lungs can add up fast.
Use of a well-maintained fume hood isn’t just protocol. It’s a line between a routine experiment and breathing in something that lingers in your system long after the flask is emptied. Airborne particles from powders like 4,4'-Difluorobenzophenone seep into unprotected space quickly. No closed-door meeting can undo ignored precautions during lab prep.
Clean-Up Isn't Just Housekeeping
Daily routines around chemical waste keep the entire lab safer. I’ve watched spills of organofluorines hit open benches, followed by someone’s hasty wipe. Disposing of contaminated paper towels like household trash only spreads danger from bench tops to garbage bins. Approach these spills with containment in mind: use chemically resistant pads, place supplies in sealed bags, and label waste for specialized disposal. Some folks think one careless toss saves time, but the real cost arrives with ruined gloves, irritated skin, and surprise regulatory visits.
Training Makes a Difference
Labs move quickly, deadlines pile up, and sometimes new staff skip proper onboarding. I once started at a site where even basic personal protection training came second to productivity. Mistakes followed, safety was compromised, and the culture never fully recovered. Introducing clear training about 4,4'-Difluorobenzophenone – its risks, symptoms of exposure, and emergency flushing procedures – sets a tone that lasts beyond a quarterly refresher.
Solutions: Culture Over Compliance
Safety comes from more than lists on a clipboard. An open culture, where one person speaks up about a missing pair of gloves or a shut fume hood, does more to protect a team than sporadic audits. Peer accountability helps; so do easy-to-read signs at storage cabinets, regular practice runs for spill response, and a manager who enforces not just the rules, but the reasons behind them.
Respect for 4,4'-Difluorobenzophenone doesn’t develop out of fear. It comes from understanding real outcomes of inattention, seeing the results of good habits, and knowing every step – from weighing powder to final waste disposal – matters. The safer the lab, the better the science.
Melting Point Sets the Stage
Few people give a passing thought to the melting point of a compound like 4,4'-Difluorobenzophenone. Every chemist I know pays close attention, though, and for good reason. The melting point—typically reported to hover around 84–86°C for this compound—does more than signal when a solid turns to liquid. It’s a badge of purity. If a batch comes off the lab bench with a melting range above two degrees wide, something’s wrong. Contaminants, byproducts, or just leftover solvents throw the numbers off. Pure samples tend to melt sharply, and that sharpness is a quick reality check for anyone scaling up or investigating a new synthesis route.
I remember my postdoc days, hunched over a melting point apparatus, watching crystals turn to liquid in a tiny capillary tube. Hit a narrow melting window, and research leaps forward. Get a broad or depressed range, and you’re stuck troubleshooting. With 4,4'-Difluorobenzophenone, the chemistry world expects precision. The compound often serves as a key intermediate for making specialty polymers, advanced resins, and high-performance materials. The raw thermal properties of these materials hinge on every step of their manufacture, but that initial melting point readout sets the tone for everything downstream.
Purity Isn’t Just Academic
Across the globe, labs and process engineers ask: How pure is that 4,4'-Difluorobenzophenone? Years ago, I watched a colleague lose weeks chasing yield, only to realize impurities were sandbagging his crystallization. This molecule’s purity influences not just melting point, but also color, solubility, and final polymer performance. Published reports show that commercial samples, when analyzed by HPLC or NMR, usually achieve above 99% purity. That level isn’t a luxury; it’s essential if someone wants uncontaminated reactions or electronic applications like optoelectronics, where stray atoms spell disaster.
Impurities cause more headaches than many people guess. Sometimes they get baked into the final product, degrading performance. Sometimes they poison catalysts or set off chain reactions during processing. I’ve seen talented scientists set up experiments with impure batches, only to watch their hard work unravel. A vendor’s certificate of analysis can assure, but nothing beats running your own checks—especially before an important synthesis or scale-up. TLC, melting point checks, NMR, and elemental analysis caught more contaminants than I care to remember.
Moving Toward Solutions
So how do we sidestep these pitfalls? Recrystallization stands out as a tried-and-true technique. Push the limits—try antisolvents, swap cooling rates, get creative. Purifying 4,4'-Difluorobenzophenone takes more patience than brute force. For those at industrial scale, advanced chromatography steps or continuous crystallization rigs step up the game. Certification helps, but I always recommend periodic re-testing; sometimes vendors change suppliers, and specs quietly drift.
In my experience, sustained collaboration between researchers, procurement staff, and quality control keeps these issues from blowing up projects. Communicating about critical properties like melting point and purity, setting up routine analytical checks, and being ready to rework or reject suspicious batches builds resilience into both lab and plant environments. Every successful development—a stronger composite, a clearer optical filter, or a more reliable adhesive—starts with getting simple properties right. That’s not glamourous, but it’s the kind of detail work that underpins every innovation built with 4,4'-Difluorobenzophenone.
Essential Knowledge for Safe Storage
I’ve spent enough time in research labs to see the difference that good chemical storage habits make. Take 4,4'-Difluorobenzophenone – a solid used in polymers and as a building block in specialty chemistry. Keeping this compound safe isn’t just a matter of habit. It speaks to the heart of lab safety, cost, and environmental responsibility.
The Risk of Cutting Corners
Tucked unsafely in a cabinet, even a plain-looking powder like this can spell trouble. Moisture, light, and air all trigger changes, sometimes subtle, but often damaging. Small mistakes—like screwing a cap back loosely or not labeling a bottle—can mean ruined materials, expensive disposal, or even exposure to hazardous dust.
Labs that skate by on ‘temporary’ solutions usually end up paying the price. Old labels get hard to read. Lids degrade. You might think a minor spill just means loss of product, but clean-up eats into time and budget, especially once regulators get involved.
Real-World Storage Tactics
Working with 4,4'-Difluorobenzophenone, I’ve learned that the right container makes all the difference. A tightly sealed glass bottle stands up well to its chemical characteristics – no unwanted reactions, no leaching. Synthetic alternatives sometimes soften or react, so glass often wins out. I always remind new lab members to double-check for cracks or chips before pouring anything in or out.
Temperature also matters more than you’d think. A cool, dry storage spot protects from moisture that could clump or contaminate. The stuff stays free-flowing, making weighing and transferring simpler. Humidity, especially in older buildings or tropical climates, can sneak in and ruin a batch before anyone notices. So desiccators still play a major role, even with robust ventilation systems.
Light can mess with stability over time. It just doesn’t make sense to keep something valuable or reactive in the open. Opaque bins or cabinets with solid doors limit premature degradation, keeping compounds in peak shape for longer. I’ve seen teams use homemade fixes like wrapping bottles in foil, but built-for-purpose cabinets work best for larger stocks.
Labeling: More Than Just a Name
Clear, current labeling goes way beyond courtesy for the next person. Hazards, arrival dates, and expiry periods aren’t luxuries. I recall finding an unmarked bottle in shared storage—a real headache that could have led to a dangerous mistake. Good labeling cuts confusion, lowers accident risk, and keeps audits painless.
Reducing Waste, Saving Money, Boosting Safety
Institutions face strict scrutiny over chemical inventories. Missteps bring regulatory fines, waste disposal headaches, or severe cleanup costs. Smart storage preserves the quality of 4,4'-Difluorobenzophenone for the long run, reducing unnecessary purchases and avoiding expired stockpiles.
From experience, the most robust labs are those where safety feels routine and proactive. Precautions don't just protect people—they protect research, budgets, and reputations. Effective storage of even the humblest chemical pays dividends no spreadsheet will ever capture.