4-Hydroxy-D-Phenylglycine: A Critical Building Block and Its Broad Reach
From Roots to Relevance: The Journey of 4-Hydroxy-D-Phenylglycine
4-Hydroxy-D-Phenylglycine did not arrive out of nowhere. Its roots dig deep into a long stretch of organic synthesis history, especially where chemistry and medicine lock arms. Years back, chemists noticed that nature cooked up a few molecules with a sturdy backbone—amino acids dotted with special features like aromatic rings and hydroxy groups. Seeing that, researchers figured the mix of a hydroxy and a phenyl ring glued to a glycine structure could lend itself to big jobs in pharmaceutical assembly lines. Decades of experimentation followed, driven by the chase to construct antibiotics with sharp precision and to engineer molecules that stick around long enough in the body to fight real trouble, not just look impressive on paper.
Looking at the Molecule: What Makes It Stand Apart
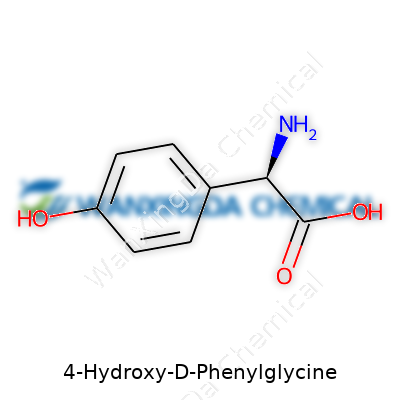
Anyone who has tried to work with amino acids in the lab knows the little quirks set each one apart. 4-Hydroxy-D-Phenylglycine starts with a glycine core and adds on a hydroxy group at the para position of a benzene ring—a decoration that changes the way it reacts, dissolves, and binds to other molecules. This arrangement means the compound is not just another brick in the wall; it offers specific reactions chemists can count on, particularly when making β-lactam antibiotics such as amoxicillin and related drugs. The presence of a chiral center brings an extra layer: you can’t swap D and L forms and expect the same result. Stereochemistry matters, particularly because only one orientation works for enzymes that expect the D-form.
Chemical Personality and Handling
It might not look like much on the surface—just another white powder, moderately soluble in water, holding together until exposed to the right solvents or conditions. But 4-Hydroxy-D-Phenylglycine’s physical and chemical skills run deeper. That hydroxy group invites hydrogen bonding, changing its behavior in reactions or mixtures. Its melting point, optical rotation, and solubility have implications every time a chemist moves it from one flask to another. This is not just trivia—handling requirements trace straight to this profile. Poor storage or shaking up the environment too much, and you lose batch purity.
Labeling and Technical Details: Why Precision Matters
Every scientist who reaches for a bottle of this compound at the bench expects to see technical data that hold up to scrutiny. Manufacturers emphasize enantiomeric purity, precise molecular weight, and water content on labels. Why? The folks downstream—those building critical antibiotics or enzyme inhibitors—don’t have time to guess or check every property in-house. Clear labeling saves hours and prevents mistakes that, in drug development, carry heavy costs. Labs need to trust a listed chiral purity of over 98 percent, knowing small deviations mean big headaches in synthesis and uncertain results in later-stage studies.
Crafting the Compound: Building Blocks and Barriers
The journey from raw materials to finished 4-Hydroxy-D-Phenylglycine usually follows well-trodden routes in organic chemistry. Most syntheses focus on coupling reactions—Strecker, asymmetric hydrogenation, or biocatalysis featuring clever enzymes that enforce the D-configuration. Each path involves trade-offs. Racemic mixtures look easier to make up front, but the effort to separate them eats up resources. Enzymatic routes look green and gentle, but finding robust enzymes (and paying for them) forms a stumbling block. Still, the promise of cleaner products with less waste drives research to refine those paths every year.
Transformation and Chemical Reactions
Once in hand, chemists rarely leave this molecule untouched for long. They might protect the amine or hydroxy groups to control which bit reacts next, all depending on the final target—often a complex antibiotic. Typical modifications include N-acylation, esterification, or introducing bulky groups that prepare the molecule for coupling into larger frameworks. In some cases, selective oxidation or reduction opens doors to derivatives with unexpected biological punch. The chemical reliability and precise reactivity of each functional group guide these transformations, and small mistakes at this step mean a compound won’t fit its intended biological lock.
Aliases in the Field: Synonyms Matter More Than Most Realize
Names in organic chemistry breed confusion if left unchecked. 4-Hydroxy-D-Phenylglycine appears as D-(−)-4-hydroxyphenylglycine, D-4-HPG, or by its cumbersome IUPAC handle. Different suppliers, journals, and patents toss in alternate spellings or abbreviations. A researcher chasing references on this compound quickly learns to search for every synonym to find the buried details. Consistency brings order to research, paperwork, shipping, and even safety planning. Miss the right synonym, and someone might order the wrong compound or miss a key paper describing a crucial reaction.
Staying Safe and Keeping Standards High
A stack of regulations and common sense protect those working with 4-Hydroxy-D-Phenylglycine. Even compounds that never see consumer shelves need careful handling. Gloves, goggles, fume hoods—non-negotiable steps for every synthetic chemist. There’s the dust hazard, the risk of inhalation during weighing, and the possibility of skin sensitization. Safety data sheets matter most in moments of trouble—spillages or unexpected reactivity. Many institutions audit their labs to make sure documentation matches practice. Ongoing safety training builds habits to spot trouble before it grows into accidents.
Shaping Healthcare: The Key Role in Antibiotics
Its claim to fame comes mostly from its time spent in the assembly lines for β-lactam antibiotics—core drugs that protect countless people worldwide. Few newcomers break into that elite club of antibiotic ingredients; 4-Hydroxy-D-Phenylglycine holds firm because its structure snaps perfectly into the backbone of molecules that block bacterial growth. Its effects ripple out from there—affecting everything from the purity of finished tablets to how quickly new antibiotic candidates can be tested. Without it, the process would slow, costs would rise, and frontline antibiotic research could hit a wall.
Pushing Boundaries: Research, Toxicity, New Horizons
Decades of research built a solid understanding of how this compound behaves, but that work never really stops. Toxicity studies chased after any sign that it might harm users or researchers; so far, problems appear mostly with careless handling rather than inherent danger. Scientists keep probing its metabolism in the body, partly because derivatives head straight into the bloodstream as part of active drugs. At the same time, innovative groups work on tweaking its structure to craft fresh antibiotics and even new enzyme inhibitors, hoping to sidestep growing bacterial resistance. These experiments look beyond what’s known, imagining how shifts in molecular features could make tomorrow’s treatments more effective or less likely to trigger allergic reactions.
Looking Forward: Challenges and Opportunities
The world keeps asking a lot from chemical building blocks like 4-Hydroxy-D-Phenylglycine. Concerns about antibiotic resistance press researchers to reinvent parts of classic medicines, knowing the essential ingredients must keep up. Environmental responsibility pushes industry to cut waste during synthesis, nudging chemists toward greener, cleaner processes—even when that means reinventing reactions that have worked for years. Regulatory standards keep ratcheting up as well, reflecting growing attention to patient safety and product quality. But these challenges bring opportunity. Strong R&D communities—across companies, universities, and government labs—collaborate to push the boundaries on efficient production, improved safety, and new therapeutic uses. Every breakthrough feeds back to public health, making this humble-looking molecule a linchpin in the fight for reliable, affordable medicines.
Getting to Know 4-Hydroxy-D-Phenylglycine
People often skip over the complicated-sounding names that come up in pharmaceutical science, but the story behind 4-Hydroxy-D-Phenylglycine deserves a pause. This little molecule plays a big role in the battle against stubborn bacterial infections. Having spent a piece of my career on the research side of antibiotics, I’ve seen how something at the molecular level can ripple out to impact the health landscape worldwide.
The Reason Drugmakers Care About This Compound
Nothing gets folks in drug development more excited than tackling antibiotic resistance. Over the years, germ-fighting workhorses like penicillin have lost their punch against certain bacteria. That opened space for new generations of antibiotics. 4-Hydroxy-D-Phenylglycine turns up as a critical building block in making semisynthetic antibiotics, notably in the glicylcycline and amoxicillin families. In the lab, chemists build on its structure to create newer antibiotics that sidestep bacterial countermeasures.
Imagine cooking — you can’t bake bread without flour. In the same way, medicines such as amoxicillin struggle to exist without this amino acid as a key ingredient. Its structure lets scientists tinker at the molecular level and fine-tune bacteria-busting power. Antibiotics made using this compound target infections from the everyday sore throat to more severe illnesses like pneumonia. Hospitals especially appreciate these advanced drugs for patients at risk of complications from tough-to-treat bugs.
Behind the Science: Why It Matters
People outside pharma circles might not notice the pressure manufacturers face as bacteria evolve. Reports from the Centers for Disease Control and Prevention show that antibiotic-resistant infections take a real toll, costing both lives and money. Hospitals see patients return with recurring infections that once responded to standard treatment. Without that next round of antibiotics built from precursors like 4-Hydroxy-D-Phenylglycine, outcomes can look grim.
There’s also a ripple effect on global supply chains. Some of the world’s largest generic antibiotic facilities rely on a steady source of this molecule. Supply interruptions, whether through export restrictions or production bottlenecks, can leave clinics short-handed. So, the existence of reliable labs able to produce this amino acid isn’t an obscure scientific win — it affects whether someone halfway across the globe gets effective treatment or not.
The Road Ahead: Staying Ahead of Resistance
The challenge never really ends. Drug-resistant bacteria keep coming. That means researchers and companies need to keep refining and improving antibiotics. In my experience, this effort relies on teamwork between governments, drugmakers, chemists, and regulators. Supporting robust research into molecules like 4-Hydroxy-D-Phenylglycine matters.
Funding basic science tends to get crowded out in tight budgets, but reliable precursor chemicals unlock next-generation medicines and keep the antibiotic world turning. Probably every pharmacist and infectious disease doctor will agree: finding fresh ways to fight old germs starts with unglamorous, time-consuming work at the bench. 4-Hydroxy-D-Phenylglycine quietly anchors that work, making a difference for people everywhere who depend on these life-saving medicines.
Why Structure Matters in Biology and Medicine
Chemistry often tells its story straight from the shape of its molecules, and 4-Hydroxy-D-Phenylglycine sets an example worth attention. You get a sense of its impact once you see how a small tweak—a hydroxyl group at the four-position on a benzene ring—completely changes what that molecule can do. It’s not just an obscure part of a lab shelf, but a building block for antibiotics, especially for drugs holding the line against resistant bacteria.
The chemical structure starts with the backbone of D-Phenylglycine, an amino acid that spins the right way in the body (the D, not the L-form, which is more common in nature). Add a hydroxy group to the fourth position of the aromatic ring and you’ve got the power to form new bonds and open up new possibilities. In biochemistry, this translates to better antibiotic activity, stronger molecular recognition, and more targeted interactions.
The Science Behind the Structure
Looking at this molecule, it carries a phenyl group (a simple benzene ring) attached to a glycine structure, where that key carboxyl group and amino group stand out. Drop in a hydroxy group on the fourth carbon of the ring, and you see how reactivity shifts. Nature figured this out before we did: certain bacteria use derivatives of this molecule as components in their own antibiotic weapons. For example, semisynthetic penicillins and cephalosporins, tools in the medical arsenal, rely on this structure to increase activity and resist breakdown by bacterial enzymes.
The specifics of chirality also play a part. D-forms of amino acids don’t behave like their mirror image, and the enzymes in bacteria recognize these differences the same way a lock fits a key. Having the D-form isn’t just some left-field detail. It means the molecule survives in places its mirror image wouldn’t, and activates antibiotic activity that matters in the clinic.
Challenges and Potentials in Application
Getting enough 4-Hydroxy-D-Phenylglycine for production remains a hard challenge. Chemical synthesis brings side reactions and extra purification steps. Attempts to use engineered microbes show some promise, but yields and purity still hit snags. Since this isn’t a glamour molecule found naturally in abundance, industry and academia keep working on better ways to produce it.
Structure-based drug design benefits from detailed knowledge of how this amino acid interacts with other molecules. By altering side chains or tweaking conditions, scientists can create new antibiotics that dodge resistance mechanisms. The approach ties directly back to chemical structure; there’s no shortcut. Every functional group, every twist of the molecule, shifts the way drugs work inside the body and how they hold up against resistance.
Steps Forward for Researchers
Researchers switching to greener, more efficient ways of synthesizing these complex molecules mark a step toward sustainable drug development. Using biocatalysis and engineered enzymes, labs push toward reducing waste and costs. Collaboration between chemists, biologists, and pharmacologists matters more than ever, since the shape and reactivity of small molecules still determine medical breakthroughs.
Recognizing the value of tiny structural tweaks in molecules like 4-Hydroxy-D-Phenylglycine means paying closer attention to every atom’s position. The story plays out in hospitals and industries as new drugs reach patients, shaped by discoveries made at the molecular level.
Walking Through the Maze of Chemical Sourcing
Anyone who’s tried to track down specialty amino acids, especially ones like 4-Hydroxy-D-Phenylglycine, knows it’s not as simple as filling out a shopping cart. This compound, valued for roles in the synthesis of certain antibiotic agents, often pops up during conversations among chemists and procurement teams trying to meet demanding research or industrial requirements. Discovering whether suppliers can truly deliver in bulk isn’t just a matter of typing a name into a search engine.
Demand Outpaces Availability
There’s no denying that research and pharmaceutical circles see real value in molecules like this. 4-Hydroxy-D-Phenylglycine links to the key fragments of well-known antibiotics such as vancomycin. Groups developing these complex drugs tend to pursue scale, but few facilities regularly maintain large on-hand stocks of such niche building blocks. As a researcher in the field, I’ve often had to send more than one inquiry to specialty suppliers, most of whom ask for projected usage volumes and require a lead time for production.
Production Isn’t Simple — and Neither Is Sourcing
Unlike more basic amino acids, reasonably priced mass-market grades of 4-Hydroxy-D-Phenylglycine don’t grace most laboratory catalogs. The compound’s multi-step synthesis — often protected by patents and involving chiral control or fermentation — limits who can make it reliably at scale. Larger facilities in China and India sometimes advertise capability, but it pays to ask detailed questions about batch size, purity, and documentation. Risk always enters with specialty chemicals, as reputation in the chemical trade isn’t fixed by flashy websites but by consistent supply and rigorous quality records.
Quality and Traceability Headline the Concerns
Every procurement manager and bench chemist wants assurance: Is this batch what the certificate says? Quality control for advanced intermediates in pharma can’t slide. Purity, stereochemistry, and trace byproducts all influence whether a shipment meets regulatory or process requirements. Over the years, my teams have learned to demand samples and request third-party analytical data before signing larger contracts. Disappointment can be costly if there’s any deviation because resynthesizing means delays that kill timelines and budgets.
Real-World Lessons: Working with Specialists
Lining up 4-Hydroxy-D-Phenylglycine for a new route or a production run works best with chemical manufacturers who understand the end-use. Relationships with trusted suppliers — usually those who have navigated pharma grade audits — matter more than cold calling dozens of companies. Transparency around raw materials, batch histories, and regulatory records becomes critical. Some procurement professionals look for ISO or GMP certifications to filter their list down to suppliers who take contamination controls seriously.
Solutions: Clear Specifications and Open Dialogue
Early and specific communication about needs helps the whole process. Projects requiring large amounts of such a targeted intermediate benefit from sharing detailed technical documents upfront. To avoid costly surprises, procurement teams ask for samples, signed quality reports, and process validation data. For continued or repeat supply, signing long-term agreements and even investing in partnership batches helps both sides minimize future risks. Demanding transparency and real quality control separates reliable supply from speculative promises.
Understanding Purity in Pharmaceutical Ingredients
Purity stands as one of the cornerstones in pharmaceutical manufacturing, and 4-Hydroxy-D-Phenylglycine demonstrates this clearly. For those who spend their days inside the quality labs, the purity specification often pushes above 98%. This isn’t some arbitrary target. In years spent listening to production floor frustrations, impurities—even in tiny amounts—complicate reactions, impact yields, and ultimately add risk to the whole batch. We’re not just talking about lab theory; any contaminant, even at levels most people would never see, changes the outcome.
Why High Purity Matters Beyond Numbers
Some might see 4-Hydroxy-D-Phenylglycine as just another amino acid derivative, but its applications call for precision. The compound helps build complex molecules like antibiotics, where even a sliver of impurity can disrupt safety, stability, and effectiveness. Regulatory agencies like the US FDA or the EMA set those high purity standards based on hard lessons from past recalls and quality failures. It’s a direct response to real-world problems—lives once put at risk by inconsistent raw materials.
My own experience sitting across from scientists puzzled by unexpected results always led us down the same trail—impurities introduced unknown variables. Take a process struggling to meet emission standards or detect uncharacteristic side products; the problem often comes back to what’s hiding in the raw materials. That’s why a typical specification for 4-Hydroxy-D-Phenylglycine might read 98% or higher. Anything less, and the investigator’s notebook starts filling up with questions about source, testing, and traceability.
Testing and Defining Purity: More than a Percentage Point
Purity for chemicals like this isn’t just about scanning a number on a certificate. Analytical techniques play the real hero—high-performance liquid chromatography, nuclear magnetic resonance, and sometimes mass spectrometry—these tools show what’s really in the vial. Labs set impurity limits, often below 1%, and single out specific contaminants like moisture or residual solvents. Testing every batch is the daily reality for suppliers who want to keep reputations intact. Fewer excuses, more data: this builds the kind of trust that keeps business rolling.
Fixing Purity Gaps: The Role of People and Process
Producing 4-Hydroxy-D-Phenylglycine isn’t just about fancy glassware or high-end reactors. Purity depends on how well teams maintain controls, monitor reactions, and pay attention to cleaning routines. Training never stops, especially as audits tighten and standards grow more demanding. Even outside the production line, distributors and drug makers scrutinize incoming shipments, often retesting even trusted suppliers. Mistakes or shortcuts anywhere in the chain reflect back on everyone involved.
Real improvement comes through openness. Sharing data about impurities, publishing new methods for analysis, and keeping recalls transparent—these approaches build reliability over time. The scientific community thrives not just on hitting numbers but on tracing them back to habits, equipment, and hard conversations about mistakes.
Pushing the Industry Forward
It’s easy to forget how many hands affect that tiny bottle of 4-Hydroxy-D-Phenylglycine that ends up in a pharma plant. Each batch stands as a marker of trust, investment, diligence, and respect for those whose lives will eventually depend on the medicine. The push for higher purity, better testing, and constant oversight sends medicine into the future, sometimes one decimal point at a time.
Understanding the Chemistry in Our Hands
Anyone who works in chemistry, pharmaceuticals, or biotechnology knows the reality of dealing with compounds like 4-Hydroxy-D-Phenylglycine. This substance plays a key role in developing antibiotics and other advanced drugs, so it isn’t just another line on an inventory list. My own early days in a research lab taught me the lesson that every chemical, even those that seem benign, brings its own quirks. Some people treat lab storage like a game—anything goes on any shelf—but in my experience, that’s the straightest path to lost time and ruined resources.
The Right Conditions Matter More Than You Think
4-Hydroxy-D-Phenylglycine demands attention to detail, not only for safety but for maintaining its quality over time. This compound, like many amino acid derivatives, tends to break down or form impurity products if kept at the wrong temperature or in a humid place. I once saw a batch turn to a sticky mess just because someone left it in a storeroom with poor climate control. That mistake scrapped weeks of work and wasted money.
Room temperature might work for sugar or salt, but active pharmaceutical ingredients ask for stricter control. A cool, dry place—think between 2°C and 8°C, much like your average specialized fridge—keeps degradation at bay. Humidity jacks up the risk for clumping, hydrolysis, and even unexpected reactions. Scientists I’ve met who store sensitive chemicals right next to a heating vent learn this lesson the hard way. Moisture and heat never help longevity, just like leaving milk out on the counter instead of the fridge.
The Human Factor—and Simple Fixes
Training matters as much as new equipment or high-tech labeling. Many storage issues start with simple oversight. Clear protocols should become second nature, not just some page in a dusty SOP binder. I’ve worked in labs where ongoing training actually made a difference—people stopped stacking random bottles together, started logging opening dates, and tracked how much of the product had been withdrawn. Labels fade or peel, so logbooks or digital tracking keep everyone honest and the storage process transparent.
Don’t underestimate container choice. Tightly sealed, chemically inert bottles keep out air and protect against accidental spills or contamination. I still remember the frustration after a colleague left a cap loosely screwed on—a little laziness that led to a major headache down the line. Glass or high-quality plastic containers beat cheap alternatives, especially in environments where cross-contamination ruins entire experiments.
The Broader Impact—From Benchtop to Medicine Cabinet
Poor storage affects more than just the compound in the jar. Pharmaceutical companies risk recalls and regulatory penalties because a crucial ingredient didn’t hold up. Researchers may publish data skewed by impure samples, which slows drug development and could impact patient safety. If we respect the requirements up front, batches remain potent and research results stay reliable. That sort of discipline at the storage shelf shows up later in real-world results.
Moving Forward with Responsibility
Every bottle of 4-Hydroxy-D-Phenylglycine on a shelf represents someone’s work, a series of experiments, or a patient’s future treatment. Treating chemicals with respect has nothing to do with fear and everything to do with stewardship. We owe it to ourselves and the broader public to keep storage practices grounded in both science and common sense. Proper storage isn’t rocket science—it’s a basic part of professional pride.