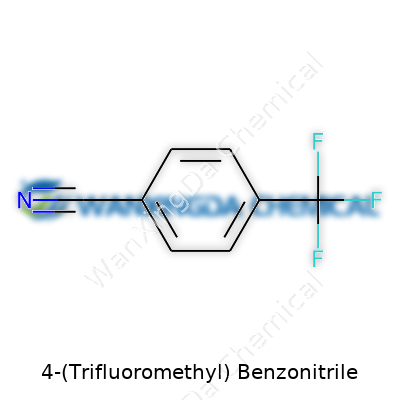
4-(Trifluoromethyl) Benzonitrile: Unpacking a Versatile Chemical from Lab to Industry
Looking Back: Historical Development
The story behind 4-(Trifluoromethyl) benzonitrile reads like a neat illustration of how organic chemistry continues to bring new possibilities to old problems. Chemists first started tinkering with benzonitriles during the growth of industrial organic syntheses in the mid-20th century. As the needs of the pharmaceutical and agrochemical sectors ramped up, so did the interest in designing clever aromatic compounds with novel groups like trifluoromethyl. Introducing this group brought about shifts in both physical properties and bioactivity. Researchers looking to tweak drug and crop protection molecules quickly appreciated the unique role that trifluoromethyl groups could play in stability and reactivity. It wasn’t long after that 4-(Trifluoromethyl) benzonitrile emerged as a solid building block, slotting easily into the toolkits of synthetic chemists around the globe.
What This Compound Brings to the Table
Organic chemists know 4-(Trifluoromethyl) benzonitrile as a colorless to pale yellow crystalline solid, lending sharpness to any laboratory shelf it’s placed on. The trifluoromethyl group adds heft and electron-withdrawing power to the phenyl ring, pushing the compound into interesting chemical territory. Chemists get a two-for-one advantage: the electron-poor aromatic ring hones reactivity, and the nitrile group opens the door to further reactions. Industries look for these types of features because they smooth the way for advanced synthesis, whether someone’s making a drug intermediate or a specialty material. The compound also stands out for its relatively high boiling point and strong resistance to many solvents, characteristics that carry practical benefits for handling and storage.
Physical and Chemical Properties Worth Noting
Anyone who’s spent time around trifluoromethyl aromatics knows that these compounds, including 4-(Trifluoromethyl) benzonitrile, pack a punch thanks to the powerful electron-withdrawing effect of fluorine. A structure like this doesn’t just display a pretty crystalline form – it shows up reliably stable, holding up against light, air, and moderate moisture. Melting points for these compounds generally float in the middle range, which suits processes that call for manageable heating. As for reactivity, the electron pull from trifluoromethyl shapes how the nitrile group interacts in downstream reactions. In the lab, I’ve seen these properties help secure stronger yields in cross-coupling and amination steps, which often trip up less robust intermediates.
Diving into Technical Details
Labelling in chemistry rarely feels flashy, but it has a real impact. With 4-(Trifluoromethyl) benzonitrile, chemists usually note the compound in grams or kilograms, each sample marked by batch purity, moisture content, and handling hazard statements. Label warnings come into play mainly due to its irritant nature to the skin and eyes, much like other aromatic nitriles. Most suppliers ship it under UN classifications, but outside of regulatory language, users gravitate toward real data: purity levels generally above 98 percent, clear expiry timelines, and solvent compatibility notes form the basis for trust and effective use.
How Chemists Pull This Compound Together
A good route to 4-(Trifluoromethyl) benzonitrile often starts from 4-bromobenzonitrile or 4-nitrobenzonitrile, depending on available reagents and cost. The introduction of the trifluoromethyl group usually calls for reagents like trifluoromethyl copper or, with the growth of green chemistry, alternatives like Togni’s reagent. A skilled hand can fine-tune the copper-catalyzed Sandmeyer reaction to drop the trifluoromethyl group right onto a diazonium salt, giving good yields and a relatively clean process. In my own experience in small-scale pharma labs, running this reaction demands respect – the exotherm when you start adding your copper reagent can come on fast, and keeping everything cool proves crucial.
Where Reactions and Modifications Come In
The reactivity of 4-(Trifluoromethyl) benzonitrile means chemists find themselves testing out hydrolysis, reduction, and cross-coupling nearly as often as they do with simpler benzonitriles. Subject it to strong acid or base, and the nitrile group opens up to yield carboxylic acids or amides, which form the backbones of pharmaceuticals and specialty polymers. Metal-catalyzed cross-couplings, such as Suzuki or Buchwald-Hartwig protocols, benefit greatly from the stability trifluoromethyl brings, making this intermediate invaluable. Over the years, labs big and small have shown how such modifications can help stitch together libraries of analogs in search of new drugs or more selective agrochemicals.
Name Game: Synonyms and Common Labels
In the chemical trade, 4-(Trifluoromethyl) benzonitrile goes by a few alternative handles: para-trifluoromethylbenzonitrile, 4-cyanobenzotrifluoride, and sometimes p-(trifluoromethyl)benzonitrile. I’ve seen shipment labels swap between these names according to region or supplier. While it sounds technical, getting used to these synonyms prevents real mix-ups, especially when ordering for a multi-country project where translations and local standards often shift between catalogs.
Zeroing In on Safety and Good Practice
No seasoned chemist skips the safety card with aromatics, and 4-(Trifluoromethyl) benzonitrile warrants the same caution. Eye and respiratory irritation shows up in the MSDS, and spills need to be cleared fast, since the vapor pressure can build up if the workshop lacks proper ventilation. I always keep gloves and goggles in play, backed by a ready supply of absorbent materials for unexpected leaks. Training lab teams on the right protocols pays off too; even with a seemingly innocuous solid, one moment of inattention during weighing can lead to stubborn residues and exposure.
Where This Compound Makes Its Mark: Application Area
Some years back, drug and agrochemical research started pushing deeper into fluorinated aromatics, and 4-(Trifluoromethyl) benzonitrile quickly appeared in patent filings for blockbuster candidates. In pharma, it works handily as a precursor to more complex scaffolds, letting chemists install the trifluoromethyl punch exactly where it matters for metabolic stability. Agrochemical companies run parallel strategies, building more persistent and selective pesticides. The electronics field, keen on fluorinated organics for specialty polymers, finds this compound useful for crafting materials with higher dielectric constants. In short, this one compound rides across several sectors, and its use on real projects shakes up the idea that one molecule equals one use.
R&D: Keeping the Wheels Turning
This molecule stays popular with R&D teams because of its blend of manageable reactivity and the distinct advantages the trifluoromethyl brings. Research groups keep tweaking conditions for greener, more efficient syntheses, both to cut down on toxic reagents and to meet regulatory and sustainability goals. In the last few years, photocatalytic and electrochemical approaches have nudged yields higher with less waste, and I’ve watched teams adopt these methods to shrink batch times and improve overall carbon footprints. That drive for smarter, safer methods seems here to stay, pushed by both economic and environmental incentives.
Digging into Toxicity and Safe Handling
Every new compound needs firm data on toxicity before industry trusts it in larger quantities, and benzonitriles keep researchers alert. Early studies flagged odors and skin irritation as the main issues, but more recent work pushes for acute and chronic toxicity profiles, especially if downstream products end up in food or medicine. Labs pay close attention to waste management, as cyanide-producing degradation — while less likely with trifluoromethyl support — looms as a concern under high-temperature or strongly basic conditions. Ensuring proper fume hoods and containment keeps workers safe, a point often reinforced in workshops and routine audits.
Where the Future Points
The growing push to design sustainable, high-performance chemicals keeps putting 4-(Trifluoromethyl) benzonitrile in the spotlight. Academic and industrial teams alike are exploring better routes to this building block, looking for alternatives to hazardous or expensive reagents. Electrochemical methods, biocatalysis, and continuous-flow systems keep gaining ground, moving the industry closer to less hazardous, more efficient manufacturing lines. There’s also the challenge of scaling: as new applications in electronics, agrochemicals, and pharmaceutical intermediates emerge, the pressure grows to make this compound cleaner, faster, and with even deeper control over impurity profiles. For anyone invested in chemical innovation, following the future of this unique benzonitrile speaks volumes about where sustainable, high-impact chemistry heads next.
Looking Beyond the Name
Science often hides its most interesting truths under thick blankets of jargon and tricky names. Take 4-(Trifluoromethyl)benzonitrile. Most folks see the name and wonder if they’ve stumbled into a chemistry quiz. Still, tucked inside all those syllables sits a chemical with a backbone that’s part of many important materials—from dyes to pharmaceuticals. The formula for this compound, C8H4F3N, might seem like a pile of letters and numbers, but it spells out an entire family tree of carbon, hydrogen, fluorine, and nitrogen atoms coming together in a certain way.
Digging into Its Shape and Why People Care
C8H4F3N means eight carbons, four hydrogens, three fluorines, and one nitrogen hang together in a special arrangement. The benzene ring, which sits at the core, gives strength and stability. Adding a trifluoromethyl group (CF3) and a nitrile group (CN) opens doors for reactivity and other interesting properties. The placement of these groups—CF3 at the fourth carbon, and CN stuck on the benzene ring—makes this molecule more than the sum of its parts.
Big pharma, for one, often reaches for this compound when building drugs. The trifluoromethyl group doesn’t just sound important—it changes how the molecule reacts, making medicines stick around longer in the body or act more powerfully at lower doses. Fluorine has a knack for making drugs less likely to break down quickly, which matters a lot when doctors want steady, predictable results. Beyond medicine, 4-(Trifluoromethyl)benzonitrile lands in dyes, polymers, and specialty chemicals, showing just how wide its reach stretches.
Connecting Chemistry to Real-World Challenges
Chemistry often looks neat on paper. Reality isn’t so tidy. Making this molecule in the lab calls for care: the trifluoromethyl group can prove tough to attach cleanly, especially without waste or risky byproducts. Years ago, I watched a research group lose days chasing yields and trying to scrub away side reactions. Every wasted gram or contaminated batch translates to higher cost, extra cleanup, and more stress for the chemists.
Environmental regulators have also raised questions about fluorinated compounds—sometimes called “forever chemicals” for their dogged persistence in soil and water. Factories that produce or use compounds like 4-(Trifluoromethyl)benzonitrile need to watch waste streams carefully. Banning a chemical tends to catch headlines, but smarter monitoring, better waste treatment, and greener chemistry push for meaningful change. Some researchers have started swapping in alternative reagents or milder conditions to lower the environmental price tag.
Solving Problems and Moving Forward
Better factories, greener solvents, and new ways to attach trifluoromethyl groups safely all push the industry in the right direction. Supporting academic research into sustainable chemical methods helps, too. Open conversations between chemical makers, regulators, and community groups keep everyone honest. Companies open up more about their material flows and waste practices now than a decade ago. Community pressure and clear-headed rules hold a lot of sway.
In the end, a chemical formula like C8H4F3N isn’t just for scientists. It’s a snapshot of how different pieces work together—and a reminder that every link in the chain matters, from lab bench to riverbank. That’s the lesson worth remembering whether you’re a chemist, a local neighbor, or someone who just wants their water clean.
Anyone tuning into the latest wave of chemical innovation will spot 4-(Trifluoromethyl) benzonitrile playing a growing role across several industries. This chemical stands out thanks to its unique structure—the trifluoromethyl bit combined with a benzonitrile backbone—allowing scientists and engineers to get creative in both laboratories and on manufacturing lines. Folks who work in chemistry see real value here, especially for those chasing reliable building blocks for new molecules.
Pharmaceuticals: Starting with the Molecule, Not the Medicine Cabinet
Drug discovery teams draw on 4-(Trifluoromethyl) benzonitrile as a core ingredient to build new therapeutic candidates. By plugging its structure into research projects, it opens doors for crafting medicines with better outcomes. My own time working alongside medicinal chemists showed just how precious these building blocks can be—sometimes that trifluoromethyl group helps make a medicine more stable in the body or helps it slip into cells where it’s needed most. Benign at first glance, the true magic arrives once researchers tweak the molecule for specific targets, making it a favorite for those solving the puzzle of safer, more effective drugs.
Pushing the Boundaries in Agrochemicals
Farmers and growers don’t always see the chemistry behind the scenes, yet 4-(Trifluoromethyl) benzonitrile shows up in the recipe for several crop protection agents. Chemists grab this molecule for building new herbicides, insecticides, or fungicides that can stand up to harsh sun and rain. The trifluoromethyl part of the molecule often toughens up the final product so it doesn’t break down too quickly under real-world conditions. Regulatory bodies watch these substances closely, pushing for effective yet safe substances—so chemists look for starting materials that offer both performance and reliability, qualities this compound tends to provide.
Modern Materials: Pushing Electronics Further
Modern electronics depend on precision parts, from thin film coatings to resilient polymers. 4-(Trifluoromethyl) benzonitrile finds a place in these technologies. Specialist polymers and liquid crystals rely on it for their special properties. The inclusion of fluorine atoms helps these new materials push past the limits of older options—delivering durability or unique electrical behaviors. My work in a materials science lab brought home just how much time and energy goes into finding the right building block for tomorrow’s screens and circuit boards. Although the work stays hidden once the final device lands in your pocket, behind the scenes, these designer chemicals quietly power the next leap forward.
Addressing Supply, Safety, and Sustainability
With demand on the rise, the global supply chain must keep up. Chemists and engineers hunt for routes to make 4-(Trifluoromethyl) benzonitrile more efficiently, cutting out waste and trimming costs. Environmental and workplace safety stand high on the list, given the hazards that can come with toxic solvents or byproducts. Better catalysts and greener methods keep turning up in academic journals, and some companies have moved fast to use these cleaner techniques. Many in the industry urge continued transparency and third-party oversight, which lines up with the push for sustainability that’s reshaping manufacturing worldwide.
Looking Beyond the Chemistry Set
The reputation of 4-(Trifluoromethyl) benzonitrile isn’t just about molecules on a lab bench but about what happens afterward—how it helps bring better medicines, smarter crop protection, and more advanced electronics to people around the world. By focusing on smart design, safer processes, and responsible innovation, chemists give this compound a future beyond the next scientific paper or product line.
4-(Trifluoromethyl) benzonitrile shows up in many research labs and chemical plants that handle organic synthesis, design pharmaceuticals, or chase new materials. It smells sharp, slices through most gloves, and carries a bit of danger if you forget it on the bench. In the world of chemicals, small details about storage make a big difference. Nobody wants an unexpected headache or worse, a lab evacuation. So let's talk about how to give this compound the treatment it deserves on the shelf.
Temperature and Air Control Matter
Experience says cool, stable temperatures keep most aromatic nitriles calm. Storing 4-(Trifluoromethyl) benzonitrile at room temperature often works, but hot summers in a poorly ventilated storeroom can force surprises nobody wants. I always lean on dedicated chemical refrigerators—not freezers, just fridge-level chill—especially when temperature swings could damage the label, the bottle, or the people near it. Ordinary home fridges can turn into a problem if food and solvents mix, so using equipment meant for chemicals matters more than most realize.
Leaving the container open or not sealing it tightly turns a stable powder into an airborne risk. Unlike heaping table salt in a bowl, this compound prefers life sealed up tight, away from moisture and oxygen. Oxygen eats away at certain chemicals and moisture causes clumping, even with those dry solids that look harmless. Silica gel packets tossed in with tight screw caps, in my book, keep the nuisance of clumping to a minimum and boost peace of mind.
Shelving and Labeling Habits
Some labs stash everything flammable in one cabinet, others split things by acids, bases, and organics. 4-(Trifluoromethyl) benzonitrile doesn’t flare up easily, but its organic nature means you shouldn’t box it with oxidizers, strong acids, or strong bases. That’s how fires, toxic fumes, and ruined research follow one sloppy moment. Segregation means reading the label before you stack it somewhere new, not just tossing the bottle on any shelf. Clear, accurate labels help avoid confusion after a late night or interrupted experiment. Permanent ink truly beats masking tape—no question there.
I’ve watched mentors double-check every label each time they reach for something. That kind of caution isn’t just about avoiding a citation—it’s about not losing days of work or risking your life. Mishaps rarely involve just the person who made the mistake but ripple out to everyone nearby.
Well-Ventilated Spaces, Not Forgotten Corners
Stashing this compound in a corner cabinet with poor airflow courts headaches or worse if vapors sneak out. Good ventilation means more than a lid and a small window cracked open. Fume hoods or designated ventilated chemical cabinets pay for themselves the day something leaks, even by accident. Faint odors may not seem urgent, but repeated exposure chips away at your health over years, not just days.
If you spot condensation, crusty bottle necks, or an odd smell, don’t shrug it off. Chemistry rewards the double-check. If nobody’s cleaned that cabinet in months, nobody knows what’s really going on inside. Keep an inventory. Audit your storage every so often. Small routines like this catch problems before they cost time, money, or safety.
Wrapping Up with Real-Life Vigilance
Chemical safety lives in small daily habits: choosing the right shelf, updating a label, checking the seals, and never assuming the person before you got it right. 4-(Trifluoromethyl) benzonitrile isn’t among the most hazardous chemicals, but treating it lightly invites trouble. No single label, sticker, or rule replaces experience and vigilance. This isn’t busywork—this is how good science stays safe and trusted.
4-(Trifluoromethyl)benzonitrile might not sound familiar to most outside chemistry circles, but it’s tucked quietly into work ranging from pharmaceuticals to advanced materials. I’ve watched labs turn to this compound for its reliability in synthesis. The level of purity isn’t just a technical detail. It can make or break downstream reactions, influence regulatory compliance, and affect entire supply chains.
Why Purity Makes a Difference
High-purity chemicals, often marketed as 98% or higher, hold a special place in both research and production settings. Chemists prefer compounds that leave little room for impurities, especially when hunting for targeted outcomes in drug discovery or electronics. A single contaminant nudges reaction yields down or introduces entirely unwanted byproducts. That’s money and time lost in multi-step synthesis. I’ve seen teams forced to trace frustrating glitches back to subpar materials.
HPLC (High-Performance Liquid Chromatography) and NMR remain the absolute standards for checking purity. Analytical chemists pour over these results before ordering large batches. Purity grades affect not only performance but safety—impurities can flare up as unknown hazards in lab and pilot-plant environments. Tooling up with low-grade material undermines the confidence researchers put into their data.
How Grades Differ in the Market
On the market, 4-(Trifluoromethyl)benzonitrile typically shows up in purities of 97% or greater. Industry suppliers label their most popular batches as 98% or 99%. That higher number comes from an extra filtration, distillation, or recrystallization step. For research aimed at FDA submissions or electronics applications, the higher grade almost always wins out. Some research teams order a technical grade—perhaps in the 95-97% range—mainly when cost trumps precision. But even modest contamination from residual solvents or related aromatic species skews tough experiments and muddies analytics. I’ve learned to ask not only for a certificate of analysis but to dig into the methods behind it.
The Risks of Overlooking Purity
Pushing forward with lower-purity material looks tempting when budgets are tight, but shortcuts bring risks. Synthesis can stall, or worse, produce false negatives in sensitive screens, losing momentum for months. I recall several collaborations where shortcuts seemed minor at first. The hidden costs reared up later—extra purification steps, extra checks, more waste. The experience taught me an economy measure sometimes turns into its own problem.
Finding Better Solutions
Big-name labs invest in closer relationships with their suppliers. Regular audits and shared methodologies can shave weeks off troubleshooting. Smaller outfits can build partnerships with analytics labs, directly confirming purity on site. Sometimes, it helps to standardize procurement lists, trimming out suppliers whose documentation lacks real detail. If tighter quality controls feel daunting, sharing best practices across teams gives everyone a leg up. The chemical supply chain isn’t just about procurement—it hinges on trust, verification, and a willingness to speak up over a small impurity that could grow into a big cost.
In the end, the chemistry community benefits when quality standards climb. Chemical purity may start as a technical metric, but it shapes everything from breakthrough medicines to microchips. In my experience, the effort and vigilance always pay off down the road.
In labs working with specialty chemicals, nothing raises eyebrows faster than a material with a mouthful of a name like 4-(Trifluoromethyl) benzonitrile. This compound pops up in research and manufacturing, especially in pharmaceuticals and advanced materials. On the surface, it looks like another organic chemical. But beneath that unassuming label, there’s a mix of concerns every chemist learns to respect.
Looking at Real Hazards
4-(Trifluoromethyl) benzonitrile carries baggage—mainly from its core structure. The cyanide group rings alarms. Cyanides, even outside the classic deadly form, tend to care nothing for human biology. Inhalation or inadvertent contact may lead to skin irritation and respiratory impacts. There have been reports in the literature of organic nitriles metabolizing to toxic byproducts inside the human body. This isn’t some chemical scaremongering; it reflects a history of real accidents and incidents in chemical plants and academic settings.
Adding trifluoromethyl only ups the ante. Fluorinated organics squeeze their way through regular gloves, and they’re reluctant to let go from surfaces. In many cases, these compounds display persistence in the environment. If they spill, cleaning up often means specialized solvents and methods far beyond soap and water. Some may even linger, wasting time and resources as teams work to decontaminate spaces. The trifluoromethyl group also grants unique volatility and sometimes unexpected fat solubility. Accidental exposures become harder to control, and waste requires more thoughtful disposal.
Why Handling Matters
People working with 4-(Trifluoromethyl) benzonitrile often have formal training, but nobody becomes immune to complacency. Routine breeds mistakes. Over the years, I’ve watched senior chemists put on just a half-mask and rubber gloves, thinking familiarity was enough. Still, even careful hands slip up. Wearing a lab coat and goggles only reduces—not erases—risk. Proper chemical fume hoods, chemical-resistant gloves, and a commitment to PPE offer the best bet for staying safe. In my time, splash goggles prevented eye problems after an unexpected evaporation event. No amount of experience replaces that shield.
Real Solutions Show Respect
Workers deserve more than a paper warning. Regular safety trainings grounded in real lab stories teach far better than checklists glued to a cabinet. Making safety gear easy and convenient beats lectures about proper procedure. For example, placing chemical neutralizers within arm’s reach, so they see use before second-guessing. Supervising young researchers to model good behavior matters. In my lab, senior techs led by example. They never missed a double-check of their gloves or the fume hood before popping open a new reagent bottle. This kind of culture beats policy memos every time.
Another angle comes from waste management. The stuff that doesn’t leave in a sealed bottle piles up in hazardous waste bins. There’s plenty of guidance that recommends separating fluorinated organics from other lab trash. Strict protocols keep downstream handlers out of harm’s way. Labs tracking their inventories and regularly disposing of old stock avoid sticky situations involving forgotten bottles and unsafe decay.
Real Knowledge, Not Just Rules
Understanding the risks of 4-(Trifluoromethyl) benzonitrile starts with the science. Anyone handling it needs access to reliable data and a culture that prizes speaking up over cutting corners. Over time, building habits—checking if the fume hood works, swapping gloves at the first tear, labeling containers immediately—cuts down on accidents and close calls. Science careers depend not on sheer technical know-how, but on steady respect for chemical hazards and practical habits to match. That’s the lesson this compound teaches, better than any rulebook.