4-Vinylphenol: More Than a Chemical, a Cornerstone in Innovation
Historical Development: Tracing Roots and Unlocking Potential
Understanding 4-vinylphenol means recognizing its story starts with curious chemists in the early days of organic synthesis. The journey picked up steam once scientists unlocked ways to produce phenolic compounds from renewable sources like lignin. Early research saw 4-vinylphenol quietly sitting behind its more famous cousins, yet as folks dug deeper into its reactivity and structure, doors opened in both the academic lab and the industrial plant. The rise of interest came alongside growth in the plastics and flavors industries, as new applications started making a clear call for this molecule. 4-vinylphenol slipped out of the research shadows and earned a place in reaction schemes for resins, polymers, and even as a biomarker that hints at spoilage in wine. Over time, this compound became a focal point in both synthesis and analytical chemistry, revealing a usefulness that punched above its weight.
Product Overview: A Look Beyond the Lab Bench
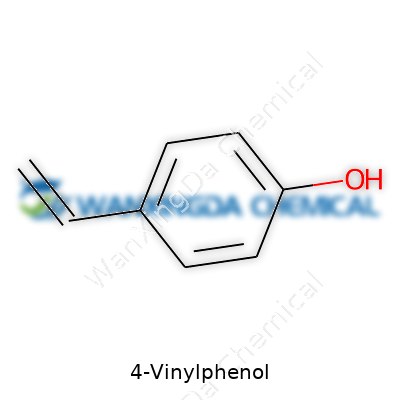
4-vinylphenol stands apart for the way its unique structure—a phenol ring attached to an ethenyl side chain—opens the door to versatility. In everyday terms, anyone handling this stuff is dealing with a core building block that fits into resin production, flavoring studies, and specialty polymers just as well as academic research. Its role as a flavor compound has put it in the crosshairs of both winemakers and food scientists, shining a light on how even tiny amounts shape taste experiences. On the industrial front, big resin and adhesive producers see 4-vinylphenol as a route to crafting specialty polymers with set physical traits. This wide reach makes it plain: interest in this compound goes well beyond pure chemical curiosity.
Physical & Chemical Properties: What Sets It Apart
4-vinylphenol shows up as colorless to light yellow crystals with a sharp, phenolic odor that hints at its robust chemical personality. Its melting point runs around 83-85°C, and it boils off near 215°C—a fact that makes safe handling a priority in any lab. The molecule’s double bond breathes life into its reactivity, letting it jump into a variety of addition and coupling reactions, while the phenolic hydroxyl kicks up both acidity and hydrogen bonding potential. In my work, compounds with this set-up often mean you’re holding a true chameleon, ready to react in several directions. Chemical stability can become an issue when oxygen or heat comes into play, requiring careful storage and planning. Solubility leans toward organic solvents like ethanol and ether, underlining its place in both research and manufacturing workflows.
Technical Specifications & Labeling: Quality Matters
Purity has always ranked as a dealbreaker for anyone buying or studying 4-vinylphenol. Even small impurities can sway reactions or analysis, which drives quality standards up. Most commercial labels flag purity at 98 percent or higher, as trace phenolic byproducts and polymerization leftovers send ripples down the line into finished material properties or flavor profiles. Specification sheets highlight moisture content and storage conditions because of the compound’s reactivity and the push for shelf-stable stock. In practical terms, the chemical’s labeling has evolved along with regulations that demand transparency—from hazard icons showing skin or eye irritation risk to batch codes for traceability. Meeting these demands isn’t red tape; it’s about making sure every vial pulled from the shelf performs with reliability and documented safety.
Preparation Method: Making 4-Vinylphenol Work at Scale
Making 4-vinylphenol in the lab or at production scale tracks the march of progress in modern chemistry. The classic route starts from p-coumaric acid, where decarboxylation stirs things up to produce the vinyl-substituted phenol. Lab techs often lean on mild basic or thermal decarboxylation, sometimes pushing for more advanced catalytic processes as scale grows. There’s an ongoing push to make greener pathways a reality, not just for sustainability’s sake but because process waste and byproduct formation hit profit sheets too. I’ve seen smaller shops use vacuum distillation to collect the product while holding back polymerization risks, and larger producers move toward continuous flow reactors for tighter process control.
Chemical Reactions & Modifications: The Tools in the Toolbox
4-vinylphenol stands out for its blend of phenolic and vinyl moieties, letting chemists tap into crosslinking reactions, hydrophilic modifications, and polymerization all in one place. The double bond jumps into radical and Michael additions with good selectivity, making this chemical a workhorse in specialty adhesive and resin manufacturing. The phenolic part opens sites for etherification, esterification, and even glycosylation, offering an avenue for creating tailored derivatives for medical research or material science. Anyone looking to add function, solubility, or stability can manipulate the vinyl or hydroxyl side—each tweak spinning out new uses and properties. From where I stand, flexibility at this molecular level makes it a favorite in R&D labs, promising room for further exploration as needs evolve.
Synonyms & Product Names: Clear Communication, Fewer Surprises
4-vinylphenol goes by more than one name, and this web of synonyms sometimes tangles up researchers, buyers, or regulators. Look for labels like p-vinylphenol, 4-hydroxy-styrene, or para-vinylphenol—each tracing back to the same core structure but sometimes flagged in databases or regulatory filings under different spellings. Synonym confusion can cause headaches in safety reporting, logistics, or even customs paperwork, so stakeholders everywhere stress clear communication and documentation at every handoff.
Safety & Operational Standards: Respect the Risks
Working with 4-vinylphenol means taking its hazards seriously. Skin contact brings immediate irritation, while airborne dust or vapor ought to set off alarms in any laboratory or production space. Regular chemical hygiene routines—gloves, goggles, fume hoods, and airtight storage—turn from checkboxes into daily habits where this compound gets handled. Regulatory agencies tag it as hazardous, with strict limits on exposure and disposal practices. From my own time among solvents and reactive intermediates, every bottle comes with a respect for the unknown: batch-to-batch variations, new findings about chronic health impacts, and compliance checks that hold teams to a shared standard. Those standards matter because safety lets innovation happen without turning tomorrow’s solution into today’s health scare.
Application Area: Where 4-Vinylphenol Shows Its Teeth
Everywhere I look—food analysis, wine science, resin synthesis, or polymer research—4-vinylphenol makes an impact. Winemakers track it as a sign of spoilage, learning that even at trace levels it brings strong medicinal or phenolic off-flavors. In adhesives, it helps engineer resins with set chemical resistance. Polymer chemists appreciate its ability to create crosslinked networks hardy enough for special coatings or biomedical devices. Analytical labs use it as a reference marker, measuring fermentation flaws or process yield. In my own experience, its value comes as much from being a marker that tracks invisible changes as from being an ingredient itself.
Research & Development: Finding the Next Breakthrough
In recent years, 4-vinylphenol has become a magnet for researchers looking to tweak properties or pursue green chemistry. Many teams focus on modifying its side chains for higher stability or new activity, moving beyond bulk materials to targeted applications in sensing technologies or healthcare materials. Some labs dive into bio-based synthesis, using plant waste to swap petroleum inputs. Others explore how to anchor 4-vinylphenol in next-gen polymers for medical implants or selective membranes. I see a community that keeps building on small insights, using this familiar compound to solve problems that stretch far beyond its roots.
Toxicity Research: The Risks Under the Microscope
Toxicologists put 4-vinylphenol under the spotlight thanks to its phenolic core. Reports flag risks on skin or eye contact, and animal tests have pointed out acute toxicity at higher exposure levels. That has pushed regulatory limits lower over time, and safety data sheets keep evolving as more findings trickle in. Long-term impacts on humans still need filling in—most evidence remains animal-based or occupational study-driven. Still, every new result means updated training materials and stricter monitoring in labs or plants. Staying ahead of these findings can feel like a race, but it protects workers and users while science pushes toward safer alternatives or improved handling practices.
Future Prospects: Looking Forward, Building Smarter
4-vinylphenol stands at the intersection of tradition and the next wave of sustainability. Green chemistry trends favor natural feedstocks and safer reaction conditions, driving research toward enzymatic production and plant-based starting points. Industry interest leans into new blends and copolymers for specialized electronics or biocompatible materials, hoping for edge cases where the unique reactivity spells out fresh opportunity. Regulatory pressure for cleaner processes and traceable supply chains will only grow, so keeping up with sustainable sourcing stands as a business and ethical move. As people learn more about its safe uses, 4-vinylphenol will shape innovation across food, pharmaceuticals, and materials science, not as a relic but as a continuing engine for creative problem-solving.
4-Vinylphenol isn’t something most people talk about over dinner, yet it pops up in more places than you’d think. I’ve spent years digging into the chemicals behind products we use every day, and this one deserves a spotlight. At its core, 4-Vinylphenol comes from the breakdown of certain natural substances—often when microbes go to work on plant material, notably in food and beverages. But food and drink tell only part of the story.
Role in Food and Beverage Flavor
In wine and beer, 4-Vinylphenol often sparks debate among professionals. Some winemakers and brewers dread its presence; it can bring a punchy medicinal or smoky aroma that throws off the balance of a good drink. This becomes more noticeable in white wines or lagers, where delicate profiles rule. From my own experience with home brewing, just a hint of off-flavor from contaminated barrels means starting over from scratch. The presence of 4-Vinylphenol, usually stemming from wild yeast called Brettanomyces, acts as a fingerprint for unwanted spoilage—but some beer styles, especially a handful of Belgian ales, actually celebrate the spicy, clove-like flavors that come from similar chemicals. Judgment calls depend on the maker’s goals and regional taste preferences.
Hidden Impact on Everyday Products
Beyond the world of craft beverages, I’ve found 4-Vinylphenol at work in a range of industries. It’s a key building block in manufacturing specialty resins and polymers. Think about coatings on electronics, specialty adhesives or inks. The reason for its popularity? Its phenolic nature means it can add thermal stability and toughness to the final product. So when electronics run hot or coatings on outdoor equipment face the sun day after day, 4-Vinylphenol’s inclusion helps them last longer. Structural engineers and designers care about these chemical details far more than most consumers realize.
Perfumers and flavor chemists also keep an eye on 4-Vinylphenol. In very precise amounts, it brings a smoky, spicy note that adds dimension to fragrances and flavors. Some of the more experimental perfumers aim for that unique complexity, blending the chemical’s edge with woodsy or leathery tones. From lab bench to boutique perfume counter, its influence deserves respect—delicate handling goes a long way to avoid overwhelming other notes.
Safety and Sustainable Questions
Every chemical in our world presents questions about safety. Regular exposure to high levels of phenolic compounds can irritate skin, lungs, or eyes. For decades, workplace guidelines have targeted such risks with practical controls, ventilation, gloves, and training. On the regulatory front, agencies set limits on exposures and residues, especially in food-contact materials. That’s shaped how manufacturers approach their formulations, pushing for careful tracking and, where possible, safer alternatives.
From the perspective of responsible sourcing, interest in renewable routes to 4-Vinylphenol rises every year. Biotechnologists now look to enzyme-based or fermentation pathways, moving away from petroleum-derived raw materials. This change isn’t just about ticking a “green” box; it cuts down on environmental impact over the chemical’s lifecycle and reduces reliance on fossil fuels.
The Road Ahead
With all eyes watching the intersection of chemistry, safety, and sustainability, manufacturers search for smarter uses and better control measures for 4-Vinylphenol. Research continues into greener production, food safety practices get tighter, and creative makers experiment to find just the right amount to enhance flavor or fragrance instead of ruin it. In my view, respect for such a small molecule comes from understanding both its risks and the roles it quietly plays, from the factory floor to the dinner table.
Anyone who works with chemicals knows that some substances call for extra respect. 4-Vinylphenol is one of those. As a writer with a background in chemistry labs and more than a few hours logged in the trenches of industrial research, I’ve seen firsthand where shortcuts on safety can land you. When it comes to handling this compound, keeping your guard up does more than check a box—it keeps you and everyone around you out of the hospital.
What Makes 4-Vinylphenol Worth Worrying About?
4-Vinylphenol sits on shelves in small bottles, and on paper, it doesn’t scream “danger” louder than some classic organic chemicals. Still, this compound can irritate skin, eyes, and the respiratory system. Even minor spills can test your attention to detail. People have gotten chemical burns or ended up with inflamed airways from playing fast and loose with basics, and those experiences stick in your mind long after the fume hood is cleaned up.
The Human Side of Precaution
I remember my first encounter with 4-Vinylphenol. I noticed the acrid odor before I even uncapped the bottle, and that was a real wake-up call. Learning to suit up properly in gloves—nitrile, not latex—felt clumsy at first, but it didn't take long before it became second nature. Lab coats and goggles aren't just for show, either. It’s easy to get careless on a late shift, but if you skip protection once, you might regret it. Even the most experienced chemists can underestimate splash risk, especially in a rush.
Why insist on a fume hood for weighing and transferring this stuff? The vapors irritate, and nobody has time to recover from a coughing fit in the middle of a busy day. I’ve seen colleagues tough it out, convinced the bottle was “just open for a second.” Red, watery eyes and an afternoon headache tell you everything you need to know about that approach. There’s no glory in ignoring good practice. A hood with strong airflow pulls those fumes away, making your workspace a lot more comfortable and safe.
Fact-Driven Solutions to Common Hazards
Putting real-world fixes ahead of empty protocols works. Good ventilation matters most. I’ve worked in older labs where the hood's airflow turned out to be below safe standards, and trust me—the difference isn’t subtle. Spill trays, absorbent mats, and tight-sealing containers can save your skin, literally. Proper disposal counts, too. Pouring leftover chemicals down the sink is still too common, and it's a habit that needs to die out. Waste streams exist for a reason, and regulations back up why pouring these organics into the sewer invites long-term damage to water supplies and communities.
Training is where the solution really takes shape. A written protocol won’t save you if nobody takes it seriously or updates it when procedures evolve. Using hands-on practice and peer mentorship keeps everyone sharp. I’ve learned more from watching a careful colleague than any stack of written warnings could ever teach me. Mistakes happen when corners get cut, usually when someone gets too comfortable or complacent.
Building a Stronger Safety Culture
No single rule covers every possible scenario, but a strong safety culture roots out trouble before it starts. I think back to supervisors who kept an open-door policy, encouraging questions on handling weird reagents, not just the obvious hazards. Real safety means making sure everyone feels they can speak up if they see something off, without getting dismissed. That kind of atmosphere stops problems in their tracks.
Handling 4-Vinylphenol safely isn’t just about personal protection. It’s about respecting chemistry and the people who work with it. The lessons I’ve picked up—watch for fumes, suit up, dispose responsibly, never get casual—aren’t complicated, but they’re non-negotiable. That’s how you come home in one piece. That’s the real bottom line.
Unpacking the Formula
4-Vinylphenol carries the chemical formula C8H8O. The structure shows a benzene ring holding a hydroxyl group attached at the para position, with a vinyl group opposite it. In simple terms, that vinyl group features two carbon atoms, double-bonded, which creates a reactive site. In chemistry classrooms, students often sketch this as a phenol ring with a vinyl arm sticking out at carbon number four.
More Than Just a Molecular Structure
Spotting 4-Vinylphenol in the real world happens more than most realize. This molecule pops up in everyday encounters, especially if you enjoy a glass of wine or certain aged beers. Yeast, under some conditions, transforms p-coumaric acid into 4-vinylphenol. This chemical often ends up influencing the aroma, swinging between medicinal, smokey, or spicy depending on concentration. Nobody forgets the sharp, bandage-like scent; those who taste a spoiled wine usually wonder where that odd note comes from.
Why It Matters
Some of the biggest debates in oenology and brewing circle back to molecules like 4-vinylphenol. Both professionals and enthusiasts run into it more than they’d like in drinks marketed as craft or vintage. Consumers sometimes walk away when these flavors overpower everything else. A grower or vintner only gets a few shots every season, so knowing exactly what’s in their barrels or casks counts for everything.
Beyond food and beverages, 4-vinylphenol also finds a place in polymer science. The vinyl group can link into bigger chains, making this compound valuable in specialty resins and plastics. Lab technicians often care about its purity and structure, since even trace contamination brings unwanted quirks to synthetic materials.
Risks and Challenges
Focusing too much on flavor ignores the chemical’s other sides. In the wrong environment, this molecule can act as an irritant—researchers handling pure samples need gloves, proper ventilation, and respect for lab safety protocols. A single spill in the wrong place becomes a teaching moment for anyone paying attention.
No chemical walks around causing trouble by itself. The process, conditions, and microbial balance shape whether 4-vinylphenol even shows up and in what quantity. Food scientists, for instance, have learned to track the bacteria and yeasts most responsible. Lactic acid bacteria in sour beers, or wild yeasts like Brettanomyces in wine barrels, work behind the scenes.
Better Decisions for Future Batches
Brewers and winemakers eager to limit off-flavors often fine-tune fermentation and storage conditions. Sterile equipment, proper sanitization, and careful screening of yeast strains put the odds in their favor. In the lab, chemical engineers design targeted synthetic routes that avoid unnecessary byproducts, including stray 4-vinylphenol.
Understanding the formula and structure turns out to be a lot more than a chemistry quiz question. Small tweaks—switching up the method or keeping a tighter process—can mean a drink worth sharing or a batch best forgotten. For those of us who appreciate a good glass or a durable plastic, tracking the details behind this little molecule helps everything come together just right.
Understanding What’s in Your Lab
4-Vinylphenol sits on a shelf in the average chemist’s storeroom, but before you know it, you’re reading its label twice. Coming from working with a range of chemicals, each with its peculiar quirks, you learn early: dismissing safety with a compound like this is never an option. It smells sharp, even through plastic, and the tiniest whiff reminds everyone it isn’t just another benign liquid.
Why Treat 4-Vinylphenol with Respect?
People in labs talk a lot about PPE, ventilation, and safe handling. The conversation tends to heat up around chemicals that do more than stain a lab coat—4-Vinylphenol’s toxicity sits pretty firmly in that category. Just touching it without proper gloves or inhaling its vapors brings risks. Long-term exposure isn’t well researched, but current knowledge points toward irritation and possible chronic effects. For those reasons, just stuffing the bottle in a drawer and locking the door counts as a rookie mistake.
Basic Storage Practices—And Why They Matter
Every storage decision starts with temperature. 4-Vinylphenol, from my experience, fares best tucked away in a cool, dry place. Direct sunlight changes its chemistry and can speed up the polymerization process. That’s why you pick a spot with stable, low light, and definitely no heat sources nearby. A simple metal cabinet with a solid door, away from radiators and windows, goes a long way.
Chemicals never mix magic; they react. Acids, oxidizers, and bases near 4-Vinylphenol just invite trouble. Returning from a short vacation once, I found an entire shelf reorganized because someone thought alphabetizing mattered more than separation by hazard class. That week turned into a sorting marathon. Lesson learned: segregation isn’t just for paperwork, especially with organic compounds that react with air, acids, or oxidizers.
The Right Containers and Labels Save More Than Time
No one in a serious lab pours 4-Vinylphenol into a mystery container. The smart move has always been amber glass bottles with screw tops, with labels showing not just the name, but purchase date and relevant warnings. I’ve seen spills from capped plastic bottles that cracked under pressure, ruining the shelf and turning a ten-minute job into a half-day cleanup.
Ventilation: Not Just a Guess
Vapors hang around, even from tightly closed bottles. Choosing cabinets with venting options lets any stray fumes escape without filling the storage room. No one wants to walk into a cloud of mystery smells first thing in the morning. At my old workplace, we installed small vent fans in our chemical cabinets—simple, but effective.
Building Safer Habits Pays Off
Regulatory agencies don’t come up with storage rules for their health. Many countries require compliance with strict standards, not to annoy chemists, but because past accidents make a solid case. I still remember stories of faculty getting injured over a mislabeled bottle or a forgotten open cap. These stories usually end with hard new rules, sometimes after hospital visits.
Not everything in the lab demands the same attention, but 4-Vinylphenol proves that taking shortcuts rarely saves much in the long run. Practical caution, regular checks, decent storage gear, and a bit of old-fashioned respect for unknowns make a safer workplace, for professionals and for students just starting out.
Health Hazards That Deserve Attention
Few people have heard of 4-vinylphenol, but it turns up in places that touch daily routines, from food processing and flavoring to certain polymer industries and lab settings. Science shows that breathing, swallowing, or even touching this compound can lead to real health risks, yet public awareness lags miles behind its use.
Workers in chemical plants get this stuff on their hands or inhale vapors, often without knowing much about long-term consequences. Short-term exposure can burn skin or eyes and irritate the respiratory system. Chronic breathing of aromatic chemicals like 4-vinylphenol increases the risk of allergy-like symptoms, asthma, and even changes in lung function. Studies in rodents suggest that high doses could possibly damage organs such as the liver or kidneys, which should alarm anyone regularly working with the compound.
Some will say, “I don’t work in a factory, why worry?” But traces of these chemicals show up in processed foods, tobacco smoke, and urban air, given their widespread use and production. Low-level contamination, especially in food packaging or storage, can add up over time. Think about kids eating snacks that have sat too long in plastic wrappers; even trace exposure at a young age can impact health years down the line.
Why Oversight Falls Short
Regulation tends to lag behind manufacturing trends. Agencies may set exposure limits but enforcement in small workplaces often proves spotty at best. Many chemical plants worldwide still lack proper ventilation, personal protective gear, or regular monitoring equipment. As a result, many workers only learn about health hazards after symptoms crop up.
I once spent a month job-shadowing in a plastics processing plant. Gloves and goggles piled in dusty corners instead of being worn. Over lunch, two workers casually talked about nosebleeds and persistent coughs since starting work. Management dismissed complaints as allergies, never mentioning chemicals. This left lingering doubts in my mind about the costs we sometimes ignore to save a few pennies on safety. Such neglect threatens family health far beyond the factory gates.
Paving the Way for Safer Practices
Science already maps safer alternatives and engineering controls. Improving ventilation, using closed systems, and replacing outdated processes can slash airborne concentrations. Where substitution isn’t possible, providing fitted respirators and regular checkups for staff becomes non-negotiable. Training workers to understand chemical risks brings lasting change far beyond posters or manuals. In my experience, open discussions on health hazards lead to far greater vigilance than rulebooks ever will.
On the consumer front, transparency means a lot. Labeling products that might contain hazardous residues empowers shoppers to seek safer alternatives. Industry giants must track and disclose chemical content up and down their supply chains. It’s not enough to rely on “acceptable limits.” We have to demand fewer hazardous materials in consumer products, especially those used in foods and packaging.
Health Over Convenience
By shifting focus from short-term profits to long-term wellbeing, businesses create healthier communities and improve trust. Modern chemistry brings incredible advances, but it also demands new responsibility. With knowledge comes the ability to change how chemicals like 4-vinylphenol shape our lives, at work and at home. Balance means finding ways to innovate without dismissing the human cost behind every new compound entering our world.