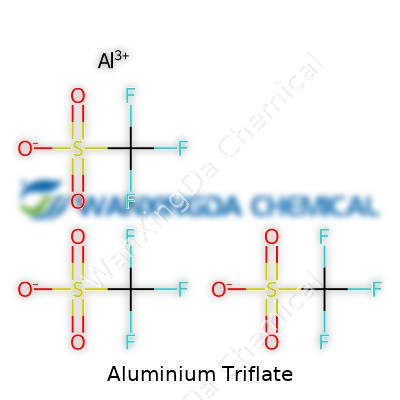
Aluminium Triflate: Behind the Scenes of a Modern Lab Staple
Historical Development
Over the last few decades, aluminium triflate has become a common name in research labs and specialized industries. Its journey didn’t start with a headline; the compound quietly crept out of the background once chemists started looking for stronger, more versatile Lewis acids. Early discoveries in the mid-twentieth century focused on aluminium compounds for applications like catalysis. As technology advanced, the demand for robust, non-coordinating anions like triflate pushed aluminium triflate forward. I’ve seen shifts in lab protocol as scientists realized the power triflate brought to organic synthesis and electrochemical applications. Now, the compound sits on the shelves of labs that value both adaptability and reliability, a testament to decades of growth in the search for better reagents.
Product Overview
Aluminium triflate, often abbreviated as Al(OTf)3, is sought after because it brings consistency and strength to the table. Many labs today keep it on hand thanks to its role as a strong Lewis acid, aiding everything from catalyst formation to electrochemical studies. The triflate group stands out for its electron-withdrawing nature, which leaves aluminium ions more accessible for interactions with reactants. In practice, this means smoother reactions and often higher yields. Speaking from experience, there’s a sense of trust you develop for chemicals that just do their job, and aluminium triflate has become one of those.
Physical & Chemical Properties
Aluminium triflate doesn't turn heads with flashy appearance—it usually looks like a white, slightly granular powder. Even though it seems unassuming, it boasts remarkable chemical resilience. Solubility in polar organic solvents like acetonitrile and nitromethane makes it a popular choice for chemists working across different fields. Its stability, even under mildly damp conditions, separates it from other Lewis acids that tend to break down. In a world where so much work gets lost to impure reagents, aluminium triflate’s purity and moisture tolerance often tip the scale in its favor.
Technical Specifications & Labeling
Labels on aluminium triflate containers usually show its chemical formula: Al(OTf)3 or Al(CF3SO3)3. Purity tends to range high, often 98% or above, since contaminants can play havoc with sensitive reactions. Packaging aims for airtight environments, often amber bottles or moisture-resistant containers, since triflate’s low affinity for water helps preserve its shelf life but doesn’t give it immunity. Labelling details hint at precision—batch number, manufacture and expiry dates, and proper hazard codes, all reminders that lab safety starts with good communication on a bottle.
Preparation Method
Manufacturing aluminium triflate draws on well-known reaction pathways. The main way involves reacting aluminium chloride or aluminium sulfate with triflic acid under controlled temperature and solvent conditions. These reactions release gases and demand exacting control—my own run-ins with exothermic errors taught me that much. Filtering, drying, and repeated recrystallization often follow, as cleanliness at the synthesis stage stands between a clean result and an unexpected side reaction weeks later. The multiple steps reflect the value placed not just on efficiency, but on the peace of mind that comes from reliable chemistry.
Chemical Reactions & Modifications
Triflate’s use as a Lewis acid ramps up in organic chemistry, especially in reactions needing a mild touch but a strong effect. Aluminium triflate can activate carbonyl compounds, drive cycloadditions, and even spark glycosylation reactions. Modifications often stem from blending aluminium triflate with co-catalysts or other metal triflates, adjusting reactivity to fit specific research needs. The adaptability here shows what happens when chemists refuse to settle—using the right blend widens the field for total synthesis of complex organic molecules. My encounters with catalyst development taught me just how often subtle tweaks open new avenues nobody saw coming.
Synonyms & Product Names
Chemists assign aluminium triflate a range of shorthand names, usually favoring “Al(OTf)3” in publications. Longer forms like “aluminium trifluoromethanesulfonate” appear on shipment manifests and regulatory documents. Industry registers and chemical suppliers sometimes use commercial names or codes, but what matters in any setting is precise identification—mistaking chemicals with similar roots creates uncertainty nobody wants in tightly controlled science or engineering settings. Familiarity with aliases keeps lab work honest and productive.
Safety & Operational Standards
Even seasoned chemists treat aluminium triflate with respect. Dry handling is key; many labs require gloves, goggles, and fume hoods when working with triflate salts. Exposure to skin or inhalation of dust can irritate tissues. Waste management gets attention due to its persistence and possible effect on aquatic environments. Protocols recommend keeping aluminium triflate isolated from basic or strong reducing substances, as unintended reactions can trigger hazardous by-products. I've learned that open dialogue in the lab, ongoing training, and clear labeling form the triple backbone of handling not just aluminium triflate but any reactive salt with teeth.
Application Area
Aluminium triflate has carved a niche in a few worlds. Its use in organic synthesis as a Lewis acid helps with things like Friedel-Crafts acylation, alkylation, and glycosidic bond formation. The battery sector also leans on its ionic conductivity to push boundaries in non-aqueous electrolytes, securing a spot in research on next-generation energy storage. Pharmaceutical labs count on it for selectivity in making complex scaffolds. The same strength that supports innovations means that any failure in quality can bring huge setbacks. My own projects have benefitted from its role in shifting bottlenecks—seeing progress speed up thanks to a single additive brings home just how far small innovations can go.
Research & Development
Ongoing research circles around advanced synthesis routes, cleaner production, and tweaks to catalytic efficiency. Scientists also look for ways to recycle or repurpose used triflate complexes, acknowledging resource limits and environmental impact. There’s ongoing work on its use in polymerization catalysts and electrochemical devices. Journals reflect a growing fascination with new materials, energy solutions, and the underlying electron dynamics that make aluminium triflate so effective. I’ve watched talented colleagues find new tricks for an old chemical, underlining the lifelong nature of learning in real science.
Toxicity Research
Aluminium compounds sometimes spark debate about long-term exposure, environmental load, and bioaccumulation. Aluminium triflate isn’t considered highly toxic at typical lab concentrations, but chronic exposure data still raises unanswered questions. Respiratory and mild skin irritation stand as the most common short-term problems. Environmental monitoring studies look for persistence, especially since large-scale industrial discharge can pose risks. Responsible stewardship arises from these studies—researchers who understand the limits and risks of their tools handle them more thoughtfully. My own safety habits sharpened as awareness of low-level toxicity risks grew alongside expanding chemical toolkits.
Future Prospects
Next steps for aluminium triflate seem to head in two directions: driving efficiency in organic synthesis and developing green applications. As labs keep chasing more selective, less wasteful reactions, the flexible nature of triflate promises to help. Energy tech stands on the edge of new breakthroughs—better electrolytes could transform batteries and capacitors, and aluminium triflate’s unique properties keep it in the conversation. Ethical manufacturing and recycling methods are also gathering steam. Having seen how fast chemical trends can change, I tend to hedge bets; nothing stays king forever, but the ongoing effort to harness the best trait of each compound is where science really grows.
Aluminium Triflate: More Than a Mouthful
Sometimes, the chemicals with the most complicated names make the biggest difference. Aluminium triflate, with its hefty name, hides its impact behind chemistry lab benches and industry doors. As someone fascinated by how small tweaks in a chemical recipe change the course of an entire technology, I think aluminium triflate deserves more attention for what it quietly delivers, especially for future electronics, batteries, and chemical processes.
Fueling Advanced Batteries and Clean Energy Dreams
Aluminium triflate’s main claim in the past decade comes from its role in advanced batteries—lithium-ion, but also zinc and supercapacitors. Battery researchers keep searching for ways to get more power with longer life while keeping size and cost under control. One thing that limits a battery’s performance is the quality and stability of the electrolyte, the liquid or gel that lets ions move between the battery’s inner parts. Aluminium triflate, thanks to its strong molecular bonds and unique structure, dissolves smoothly into these electrolyte mixtures.
Some trials with lithium and zinc batteries show that aluminium triflate helps reduce side reactions, meaning the battery parts degrade slower and cycles last longer. Greater safety also results from its resistance to breaking down at high voltages. Compared to older salts—some with nasty byproducts or limited thermal stability—aluminium triflate keeps unwanted reactions in check. This detail might not make the headlines but makes electric cars safer and phones less likely to overheat.
Pushing the Boundaries of Organic Synthesis
Lab chemists, including myself on a handful of projects, turn to aluminium triflate in synthetic routes needing a tough and reliable Lewis acid. Lewis acids help push chemical reactions forward, speeding up or even tinkering with the exact outcome of a process. In specialty pharmaceuticals and fine chemical labs, the need for precise reaction conditions keeps growing. Aluminium triflate brings unique properties to the table, such as high solubility in organic solvents and compatibility with delicate reaction partners.
My own work with coupling reactions showed how stubborn some organic molecules can be. By shaking aluminium triflate into the mix, we reached conversion rates no other acid could match, cutting down on leftovers and cleanup time. This boosts lab efficiency and slashes waste, which matters for both budgets and environmental impact.
Industrial Benefits Without the Usual Downsides
Industries chase efficiency at every stage. Aluminium triflate, compared to older standbys, packs more bang per molecule in many cases. It doesn’t produce corrosive byproducts that can eat through equipment, and its stability means less risk of runaway contamination or unplanned downtime. For factories producing electronic parts, OLED displays, or high-purity chemicals, cutting out these headaches brings down maintenance costs.
Regulators and local communities watch for toxic byproducts and hazardous waste. Aluminium triflate’s behavior tends to draw fewer red flags than alternatives like aluminium chloride or perchlorate salts, which can raise alarms for groundwater contamination or explosive risk.
Looking Ahead: The Case for Innovation
As research pushes deeper into new battery types, greener chemical routes, and reliable electronics, the demand for well-behaved reagents grows. Still, there’s room for honest evaluation. The cost of manufacture and sourcing of aluminium triflate isn’t trivial. Academic papers and industrial reports note its price can slow adoption, especially for budget-strapped labs. Chemists and engineers actively work on scalable ways to recycle or regenerate these specialty salts, or even to design cousins with similar benefits and easier production cycles.
Aluminium triflate might not spark public excitement, yet its chemistry feeds directly into safer cars, smarter phones, and cleaner production. For anyone invested in sustainable progress, its journey offers proof that even small chemical shifts can ripple through entire industries and touch daily life.
The Formula That Shapes a Compound’s Future: Al(OTf)₃
Aluminium triflate shows up in labs and advanced manufacturing more often than most people would guess. Its chemical formula, Al(OTf)₃, isn’t something you see in everyday conversations, but this detail—combining one aluminium atom with three triflate ions—holds a story that reaches far beyond the world of dull textbooks. Triflate, known chemically as CF3SO3−, gives the compound unique traits unlike the typical salts from high school chemistry sets.
Why the Formula Matters
Those who dig into chemistry might notice that triflate-based salts aren’t just powdered curiosities. Al(OTf)₃ stands out because of the fare of options it offers. The triflate ion brings stability and non-coordinating properties to the table, which means it doesn’t tie up aluminium in tricky bonds. In practice, this matters for work in non-aqueous chemistry—settings where water isn’t welcome, which covers much of battery research and advanced materials science.
I remember working on a research project where the regular suspects—chlorides, sulfates—kept getting in the way, creating unwanted reactions or turning simple processes into hours of extra work. Swapping to a triflate salt turned a week-long purification haze into a straightforward afternoon grind. The chemistry just cleaned up.
Backing Up the Claims
Researchers have found aluminium triflate to be a top choice for making high-performance electrolytes, especially in the search for better energy storage. A published study in the Journal of the Electrochemical Society put the spotlight on Al(OTf)₃ as an effective candidate for rechargeable aluminium batteries. The non-coordinating flavor of the triflate anion lets aluminium ions stay mobile and available, which keeps these systems efficient.
It doesn’t stop with batteries. In organic chemistry, using aluminium triflate as a Lewis acid can move along tricky transformations—making hard-to-achieve reactions not just possible, but practical. Its stable structure resists water better than many competing compounds, so those in humid labs aren’t sent back to the drawing board when the weather turns sticky.
Addressing Environmental and Safety Concerns
Greater popularity brings more scrutiny. Any compound that carries fluorinated groups, like the triflate in Al(OTf)₃, deserves a close look. Fluorinated chemicals don’t always break down easily in the environment and can be persistent in soil and water systems. Awareness and transparency about these features help researchers make better choices. Current evidence points out that the stable structure helps keep reactivity in check, but waste management needs honest attention. Any lab working with Al(OTf)₃ takes care to collect and treat its waste, rather than pour it down the drain.
Finding Smarter Practices
Initiatives to create greener, safer triflate salts continue. Chemists try less harmful fluorinated groups or tune the synthesis conditions to lower emissions. Support for these solutions from both academic and industrial settings speeds up their arrival in labs across the globe. These changes echo every time a student or researcher pulls a bottle from the shelf and weighs out a few grams, knowing a safer process backs up their work.
Beyond the Label: Why Aluminium Triflate Storage Matters
People often overlook how everyday routines in research hinges on chemistry’s finer details. In my experience, aluminium triflate jumps off the shelf as both a useful and sensitive reagent. Unlike bulk chemicals tossed in the corner, this one shapes how you treat the whole storage system.
What Happens in the Real World
In university labs, I learned an important lesson fast: this compound hates water like cats hate baths. Open a bottle on a humid day, cap it just a bit late, and you’re risking unpredictable, even hazardous, results for your next run. That bite of humidity can mean reactions go sideways or worse, expensive failures. According to the American Chemical Society, triflates show high hygroscopicity - they grab water from the air and never let go. Just imagine coming back to a lumpy, sticky mess; it’s happened to colleagues who let comfort win over caution.
Simple Storage Isn’t Enough
Shelves in the back of a dusty storeroom don’t cut it. Here’s what worked in shared labs: using tightly sealed bottles, ideally those with a PTFE-lined cap. We’d double-bag the container, stick it into a desiccator packed with fresh silica gel, and track the dates pretty closely. Exposure creeps up when protocols slip, especially with rushed handling. I noticed that chemists respected materials like this more after one bad batch lost through lazy storage.
Temperature matters, too. Room temperature works, but heat brings risk. Don’t shove triflate near steam pipes or radiators—heat speeds up the breakdown or moves moisture quicker. Refrigeration helps, but only if you keep condensation in check each time you pull the container out.
Why Protocols Can’t be Shortcut
Lab accidents don’t always mean explosions and alarms. Sometimes, they’re just bad data, wasted weeks, or pricey re-orders. The National Institutes of Health highlight that improperly managed moisture in the workplace often leads to degraded reagents with sneaky, hard-to-spot changes in chemistry. It isn’t enough to expect that a “keep dry” sticker will do all the work.
From an environmental and safety angle, spills reach a new kind of headache with fine powders like this. Proper packaging and training keep everybody safer, not just the folks handling the triflate. Some university labs take it a step further, logging each weigh-out and storage update for future users. That level of transparency weeds out mistakes and makes new students take those “silly” details more seriously.
Real Solutions for Reliable Results
The best changes I’ve seen always start simple. Build a routine—swap out old desiccants, keep the caps on tight, and never weigh out near the fume hood with fans blowing. If budgets allow, switching to dry boxes with humidity monitors takes things up a notch. Some departments invest in automated tracking so supplies don’t vanish or spoil, saving costs over the long haul.
All these steps come down to respecting the little things in chemical storage. Aluminium triflate doesn’t care about excuses or shortcuts. Build sound habits, keep it dry, and you protect your work, your lab mates, and your investment.
Looking at the Risks and Realities
Aluminium triflate pops up in labs and specialty manufacturing, often flying under the radar compared to more common chemicals. Questions about its safety keep surfacing, and for good reason. Too many chemicals get glossed over until stories about exposure surface, followed by the scramble for documentation and regulated controls. I’ve spent years around specialty materials, listening to chemists, reading regulatory bulletins, and seeing the uncertainty that creeps in whenever a new compound enters the supply chain. If you’re going to have a chemical with “aluminium” and “triflate” in the name, it’s smart to ask hard questions about what handling it actually means for safety and health.
Research on aluminium triflate itself stays pretty thin, which always makes me nervous. Most chemists go looking for safety data sheets or guidance from regulatory bodies. You’ll find references to related aluminium salts. The triflate part refers to the trifluoromethanesulfonate group, which offers strong, stable ionic properties. It gets plenty of attention in battery research and as a catalyst in organic synthesis. At this point, the compound hasn't cracked major headlines for causing accidents or widespread illness, but less publicity doesn’t mean no risk.
What the Facts Say
Looking at its relatives, aluminium compounds often spark worries about long-term exposure and bioaccumulation. Common aluminium salts sometimes pop up in concerns about possible links to neurological effects. Triflates, on the other hand, haven’t earned much of a reputation for human toxicity, but the presence of fluorine as a component makes me take notice. Some fluorinated compounds, particularly those in perfluorinated categories, don’t break down easily and can have serious environmental persistence. That said, not all fluorinated molecules behave the same.
I haven’t uncovered any major health incidents tied directly to exposure from aluminium triflate in laboratory or industrial work. For most workers, the main exposure routes would involve dust inhalation or accidental contact. As with many reactive chemicals, bare skin and eyes need protection. Breathing it in as a fine powder, or getting it in the eyes, brings typical chemical risks—think irritation, coughing, or possible allergic reactions. Lacking thorough long-term animal testing or controlled human exposure trials, many regulators err on the side of calling it an irritant and recommending minimized contact.
Environmental and Workplace Perspectives
Waste handling makes up a big part of the conversation around newer or specialty chemicals. Disposal rules often treat aluminium triflate alongside other metal-containing and fluorinated materials—usually with the assumption that, unless you know exactly how it’s broken down or neutralized, you handle it as hazardous waste. Proper containment stops dust from getting airborne and cuts the chances of contaminating lab benches or water sources. Any spills get treated as a cleanup priority. Flipping the package over and tossing it in the regular trash or down the drain won’t cut it if you care about worker health or the environment.
Training matters. I’ve watched well-meaning researchers get careless with lab gloves and masks because some compounds don’t “sound” as threatening. If your workplace uses aluminium triflate, make sure everyone who handles it knows the real risks and wears the right protection—N95 masks for dust, chemical-resistant gloves, goggles, and lab coats. Spending ten extra minutes on a safety briefing beats risking days out sick.
Improving the Safety Culture
Every time a new material lands in the shop or research space, the team benefits from a cautious approach. If you don’t know the chronic effects, treat the chemical with respect. Push for more research on long-term impacts, ask suppliers for the latest safety documents, and keep the local environmental officer in the conversation. In the end, it comes down to active curiosity. Instead of assuming the absence of evidence means the absence of danger, stay proactive. That way, health doesn’t get sacrificed for the sake of productivity—or unknown risks hiding in plain sight.
Beneath the Lab Coats: Aluminium Triflate’s Surprising Roles
Spend enough time in research labs and you start noticing how some chemicals quietly drive breakthroughs. Aluminium Triflate falls into this crowd — not flashy, yet quietly essential to progress in places most folks never think about. This compound, often called a Lewis acid, grabs the spotlight in chemistry circles for its ability to turbocharge certain reactions. But its usefulness reaches much further.
Electronics and Batteries: Keeping Pace With Innovation
The electronics world rarely takes a breather. Behind every new smartphone or electric vehicle, teams race to squeeze out better performance from batteries and capacitors. Aluminium Triflate has grown popular as an electrolyte additive in lithium-ion batteries. Unlike cheaper salts that corrode parts or break down under stress, it keeps working and doesn’t play rough with sensitive battery components. Researchers point to its stability in demanding conditions, giving batteries a much longer life without the drama of swelling or unexpected fires. Over time, technology that offers better safety and longer-lasting batteries could help cut waste and push green tech further ahead.
Chemical and Pharmaceutical Manufacturing: More Than Just a Catalyst
Plenty of everyday products rely on tough syntheses. Take pharmaceuticals — the methods for assembling complex drugs can’t afford uncertainty. Here, Aluminium Triflate works as a strong acid catalyst, helping to bond carbon atoms smoothly in processes like Friedel-Crafts reactions. I’ve talked to chemists who swear by it when other acids leave them with unwanted byproducts or fail to get a clean yield. This reliability cuts down the steps needed to purify finished products and, in a world of ever-shorter patent lives, shaves precious time from drug approval timelines.
Fine Chemicals and Fragrances: Precision Makes a Difference
Ever wondered how your favorite perfume keeps the same crisp scent, bottle after bottle? The answer lies in careful control during synthesis, which often calls for powerful catalysts. Fragrance houses and specialty chemical makers look for ways to attach groups to aromatic rings without producing a mess of byproducts. Aluminium Triflate gives chemists that fine touch, leading to consistent results in flavors and fragrances that keep customers loyal. In a business where reputation stands on repeatability, this chemical pulls more weight than most imagine.
Sustainability and Future Potential
With the drumbeat for greener manufacturing getting louder, there’s no ignoring Aluminium Triflate’s credentials. It can often drive reactions in water rather than hazardous organic solvents. That trait lets factories lighten their environmental footprint and pass stricter audits. It’s not a silver bullet, but it supports a push toward cleaner chemistry. Forward-looking firms and startups alike, especially in Europe and Asia, show growing interest as they chase efficiency and less toxic waste.
Gaps and Opportunities for Change
No chemical comes without hurdles. Cost and safe handling tips top the list for companies thinking of making the switch. Some still stick to old-school acids out of familiarity or to cut costs upfront. Industry groups and academic partnerships could do more to share safe methods and real-world data — not just glossy marketing pitches. Lowering the price of greener chemicals like Aluminium Triflate through better production methods would speed up adoption, especially for smaller businesses that want in.
Final Thoughts
The industries using Aluminium Triflate might not make headlines, but they shape plenty of devices and products most people rely on. In the drive for safer, smarter manufacturing, this behind-the-scenes player will likely keep finding roles across chemistry and tech for years to come.