Ammonium Perfluorooctanoate: Its Surprising Past and Uncertain Future
Decades of Use: How Did We Get Here?
Walking through the tangled history of ammonium perfluorooctanoate, often called APFO or PFOA ammonium salt, is like taking a guided tour of industrial ambition and the slow march of scientific awareness. Back in the mid-twentieth century, nobody batted an eye at a “wonder chemical” that could smooth out the edges of Teflon, resist oil and water, and survive just about anything the world could throw at it. Chemical companies brought PFOA and its salts to market as the backbone for making fluoropolymers, materials that coat frying pans, carpet, even the wiring in your car. Everybody saw the finish line: innovation, convenience, and profits. Almost nobody looked sidewise at what it might cost over the long haul.
How the Chemical Takes Shape
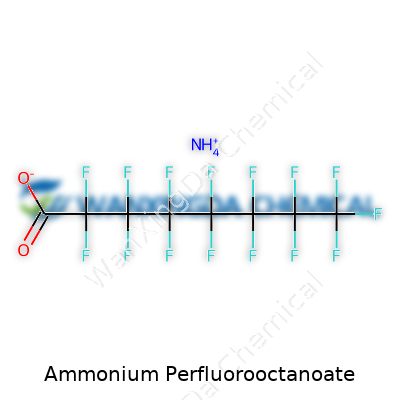
A layperson spots APFO as a white powder, often with a faintly pungent smell. Chemists describe it as a compound with the formula C8HF15O2·NH4. What stands out is that chain of carbon atoms—eight in a row—each linked to a battalion of fluorine. This chain dodges both oil and water, a stubbornness that made it so handy in manufacturing. APFO melts at around 45-50°C, dissolves pretty well in water, and latches onto surfaces with a persistence that's legendary among industrial materials. Regulations demand the labeling state its name clearly—ammonium perfluorooctanoate, or sometimes ammonium salt of PFOA—since nobody wants confusion in applications as touchy as synthesis of high-tech resins.
From Synthesis to Everyday Life
Making APFO starts with perfluorooctanoic acid. The acid reacts with ammonia or ammonium hydroxide to form the salt, a process that's both robust and well-known among industrial chemists. Professionals prefer this salt form because it's easier to handle than the free acid and helps keep certain reactions predictable, which matters during polymerization. In the chemical reactions that follow, APFO delivers its perfluorinated chain, a workhorse that never easily breaks down. After it does its job in the reactor, traces often wind up in waste streams, and that’s where the roots of its environmental persistence grow.
What Other Names Hide This Chemical?
APFO travels under several guises in the literature and commercial records. Folks call it ammonium pentadecafluorooctanoate, PFOA ammonium salt, or simply by its registry number. These names disguise neither its chemical backbone nor its environmental staying power. The catalog of synonyms can be confusing for outsiders, making it tricky to track the compound across different regions or over time. The jargon ensures only those familiar with the field can follow the thread, often leaving the larger public out of essential conversations about risks and benefits.
Safe Handling: Science Versus Experience
Anyone who’s handled APFO in a lab or plant setting knows this isn’t a substance for beginners. Rules call for goggles, gloves, and fume hoods. Companies keep usage tightly monitored, especially since the compound resists nearly all breakdown, slipping through standard filtration and even some incineration systems. Add to that APFO’s ability to bioaccumulate, and the stakes rise quickly. The safer route draws on both careful labeling and a culture of respect for the hazards involved—standards sharpened through incidents and research slowly trickling down from the lab to regulatory bodies.
Behind the Product: Where Does APFO Show Up?
The shadow of APFO stretches across dozens of industries. It’s not a household item, but it’s the linchpin in making PTFE and other fluoropolymers, those glossy, durable coatings on cookware, cable insulation, and waterproof fabrics. Some labs push its chemistry further, looking for new uses or tweaks that might weld its physical strengths to different backbone molecules. A lot of folks working in manufacturing grew comfortable with APFO, treating it like any other tool. This comfort ended up contributing to how widely traces have spread, popping up in streams, wildlife, and—uncomfortably—drinking water around the globe.
Technical Debates and Research Advances
Scrutiny on APFO drove a surge in research that runs hot to this day. Academics and government scientists have spent years measuring how it moves through soil, water, and living things. High-performance liquid chromatography and mass spectrometry track its migration down to parts per trillion, often finding it lurking in places long after production supposedly ended. Questions about how to destroy or contain this chemical drive risk assessment and reengineering efforts in the industry. Every new study chips away at the myth of a simple, neutral workhorse chemical, revealing complex interactions with proteins, membranes, and hormonal systems.
Health and Biodiversity: Red Flags from the Data
After years of complacency, research has filled in grim details. APFO damages immune function, messes with hormones, and potentially raises cancer risks in people with regular exposure. Rats and mice show stunted growth and increased tumors when scientists feed them APFO, leading regulators to set ever tighter limits. Even tiny exposures, over a lifetime, can accumulate, elevating blood levels with no clear route for a quick cleanup. Wildlife—from fish to birds—show similar effects, hinting at a wider ecological threat. More communities started testing for APFO when odd spikes in cancer or immune disease numbers drew the attention of health officials and activists alike.
Looking Ahead: A Chemical at the Crossroads
With growing evidence and mounting public pressure, the world faces a tough choice about what to do next. Some companies have stopped using APFO altogether, figuring the liabilities outweigh the advantages. Others search for replacements—short-chain alternatives, novel polymers, or green chemistry approaches—but face steep technical and economic hurdles. Emerging regulatory bans in the EU, US, and Asia suggest the window is closing for APFO. Remediation remains slow and costly, pushing researchers to invent new methods for extraction or destruction, such as advanced oxidation or high-temperature incineration. It’s clear that walking away from APFO won’t erase decades of deposits in bodies and landscapes.
What Can Make a Difference Now?
If history taught anything, it’s that we can’t afford to ignore persistent pollutants just because they fuel useful products. Tighter monitoring, clearer labeling, transparent supply chains, and investment in safe alternatives all demand attention. Community right-to-know protections and public health interventions need real teeth—especially for neighborhoods forced to bear the brunt of industrial missteps. Direct action from scientists, regulators, industry, and affected people will matter far more than hand-wringing or empty promises about “phasing out.” Everyone—whether sweating in a production plant or pouring water from the kitchen tap—deserves candor and commitment on what chemicals like APFO really mean for health and a livable environment.
Behind Everyday Conveniences
Non-stick pans, rain jackets, fast food packaging—these items make day-to-day life more convenient and comfortable. What goes into them, though, often escapes our attention. Ammonium perfluorooctanoate, sometimes called APFO or C8, helped revolutionize how industries made products repel water and grease. It served as a crucial ingredient in making polytetrafluoroethylene, the chemical behind brands like Teflon. This compound helped cookware shed eggs like magic and fabrics shrug off water.
Why Ammonium Perfluorooctanoate Got So Popular
Factories leaned into APFO for one reason: performance. It let them craft materials that resist sticking, staining, or wetting out. From pizza boxes that don’t wilt under cheese grease to carpet treatments that repel spills, APFO stretched far beyond the kitchen cupboard. Firefighters wore gear that stayed dry longer, and phone wires kept their coatings thanks to this chemistry.
Just as synthetic detergents once replaced soap because they cleaned clothes even in hard water, APFO replaced old-fashioned coatings because it simply worked better. But every shortcut seems to come with a cost.
Real World Costs: Health and Environment
Stories about APFO’s downsides rarely made prime-time news until scientists started digging deeper. I learned about so-called “forever chemicals” after reading a study linking PFOA—APFO’s close cousin—to increased health risks like cancer, liver trouble, and immune changes. APFO persists in the environment and works its way into water, soil, wildlife, and even blood samples from ordinary people. Growing up near a factory town, I remember hearing local worries about water safety, but the big picture didn’t hit me until later.
After major lawsuits exposed companies for hiding research on these risks, many countries began to act. Several banned or restricted APFO use, and big manufacturers scrambled for alternatives. Still, it’s a tough mess to clean up. Once out in the world, many of these chemicals don’t break down for decades.
What Comes Next?
Some folks insist complete phase-out is overdue, while others fight for exceptions. It’s hard to balance the convenience of stain-resistant couches with concerns about polluted drinking water down the road. Better labeling and transparency matter, so people can make up their own minds about what they bring into their homes. Companies hold a responsibility to push toward safer substitutes, and many now invest in new fluorine-free coatings.
Consumer awareness makes a real difference. Friends of mine now check for “PFOA-free” claims on pans and jackets. Restaurants started switching to untreated packaging after community pressure. Governments continue to research and regulate—California, for example, set tight limits for PFAS in water. People who want to go further can support clean up of contaminated sites or ask local officials about drinking water testing.
Rethinking Innovation
The drive to solve problems with new chemicals got us into this tangle, so it’s time to rethink what counts as progress. We can’t pretend away decades of contamination, but supporting safer substitutes, demanding clear information, and learning from past mistakes can help steer us toward better solutions. A wetter raincoat seems like a small price to pay compared to the long-term health of our communities and water.
Real-World Experience Sparks Concern
Stories about chemicals often trigger a split reaction: part of the public immediately worries, and part shrugs it off as another distant industrial issue. Ammonium perfluorooctanoate (APFO), a mouthful of a name, keeps turning up in heated debates for good reason. Used for years in making nonstick cookware and stain-resistant carpets, APFO leaks well beyond laboratories and manufacturing plants. It ends up in water, soil, wildlife, and the human bloodstream.
Everyday folks don’t talk about perfluorinated chemicals over dinner, but the impact sneaks up on us. I grew up near a town where chemical plants pumped out this stuff years ago. Today, people still check their water for contamination, worried that past decisions affect future generations. This isn’t a far-off problem—it's in the backyard for many communities. The Center for Disease Control and Prevention found measurable amounts of such chemicals in most people's blood in the United States. Even if you avoid nonstick pans, you can’t skip what’s in your air or tap water.
Health and Environmental Headaches
The big issue with APFO and related "forever chemicals" hinges on their resistance to breaking down. These molecules stick around—sometimes decades—so exposure adds up. Research ties APFO exposure to a range of health problems. Scientists connect it to cancer, liver damage, weakened immune function, and possible developmental delays in children. That’s not scare talk—it's backed by studies from leading health institutions.
Wildlife doesn't get a free pass, either. River fish living near old manufacturing sites show traces of this chemical in their bodies. The toxin travels through streams, winding through food chains. Birds eat the fish, mammals eat the birds, and on it goes. Even algae, the most basic water life, struggle near high concentrations.
Regulation and Industry Defenses
Some industry voices like to argue that doses make the poison, citing complicated studies to suggest average exposure stays low and risk remains manageable. Regulators around the world started cracking down only after mounting evidence showed harm. Agencies such as the U.S. Environmental Protection Agency and the European Chemicals Agency have already labelled this chemical as hazardous, pressing companies to track, limit, and, where possible, eliminate its use.
Trying to swap APFO for less persistent materials isn’t as easy as flipping a switch. Manufacturers complain about costs, product performance, and lack of direct replacements with similar properties. That said, some companies responded by investing in safer chemical design, proving alternatives aren't science fiction. The push for transparency—full ingredient lists, third-party water testing, honest labelling—often comes from concerned citizens, not corporate boardrooms.
How Change Can Happen
People in affected towns banded together, pressed lawmakers, demanded answers, and eventually forced utility companies to improve water filtering. Health advocates show parents how to reduce risk—simple changes like better water filters, avoiding certain takeout boxes, skipping stain-resistance sprays. It's not the perfect fix, but every little step takes away a bit of exposure.
Shifting industry habits calls for public pressure and steady, science-driven regulation. You can't wish chemicals away, but you can hold polluters responsible and drive demand for safer technologies. Personal experience backs up what studies prove: stubborn chemicals such as APFO don’t vanish, so ignoring them won’t make them less of a threat.
The Gravity of the Material
Most folks outside chemistry circles haven’t heard of ammonium perfluorooctanoate, but those working in certain industries know this stuff changes lives and sometimes not in a good way. It’s been used in making nonstick cookware and waterproof fabrics. The trouble is, it sticks around – in the environment, in the body, and in headlines about chemical safety. The stakes climb high, since even low exposures can build up with time and cause health problems. That alone wakes me up before I unscrew the lid of any drum that mentions it.
Personal Protection as a Foundation
Experience taught me early: don’t skimp on gear. Goggles, gloves, and lab coats sound basic but save hours and maybe years of future regret. Some folks in the plant still try to roll up sleeves to “get more done” – chemical reactions and skin can’t shake hands without leaving a nasty mark. If any is spilled or splashed, a shower must not wait. Anyone who’s had chemical burns or spent the afternoon at an occupational health clinic gets the message quickly.
Air Quality Can’t Be Ignored
I once worked in a lab during a summer where the ventilation system struggled. The difference in how we felt – headaches, tiredness, that scratch in the throat – told us more than any data sheet. Dust masks don’t cut it for something like ammonium perfluorooctanoate. You want a real respirator, ideally a system designed to catch both dust and vapors. If you ever notice a chemical smell, it already means something’s wrong. The solution rests in regular upkeep – nobody should assume ventilation keeps working unless someone checks the filters and makes sure airflow stays steady. Walking into stale air near an open drum is not worth any risk.
Respecting the Waste
I learned from colleagues in waste management that tossing rags or containers in regular trash doesn’t just break rules—it puts neighbors and sanitation workers in harm’s way. Proper disposal means sealed, labeled containers that end up in the hands of hazardous waste pros. The local landfill cannot fix what this compound can do to the water or soil if mishandled. In some areas, regulations now require detailed logbooks to track every ounce. Keeping those current takes time but makes everyone accountable. That push for transparency always pays off, because one overlooked mess can haunt a company and a community for years.
Training Makes the Difference
Hazard training often gets a bad rap in the breakroom, especially after a long shift, but these sessions aren’t just checkboxes. In one of my first jobs, a ten-minute demo on chemical handling saved a coworker from splashing a bottle in his face. Clear signage, emergency eyewashes, and updated procedures help. It boils down to people knowing what to do and practicing it regularly. If leadership shrugs these off, standards sink and risks climb. Putting money and time into training highlights company values—no shortcut beats a workforce that isn’t afraid to speak up or call out a hazard.
Solutions Rely on a Safety Culture
Handling ammonium perfluorooctanoate safely asks everyone to be on board – it isn’t just about PPE or paperwork. Stay curious, ask questions, double-check systems. There’s nothing folks in these industries can take for granted, because the impact lingers beyond the individual. Building a solid safety culture, making it routine, and trusting real-world experience does much more than any checklist could. That way, nobody has to bet their health for a paycheck.
Diving Into the Structure and Its Impact
People may know the name, but not everyone can picture Ammonium Perfluorooctanoate. Try saying that at a backyard barbecue. Chemically, it goes by the formula C8F15O2NH4 and its structure draws attention for more than its tongue-twister name. There’s a long carbon chain, every hydrogen swapped out for fluorine except for a terminal group, wrapping up with an ammonium tag. This is no ordinary molecule. The toughness comes from its eight-carbon backbone decked out with fluorine, about as sturdy a shield as chemistry offers. That’s why it resists breaking down.
Behind the Chemistry: Why Structure Matters
Scientists and manufacturers didn’t pick this structure by accident. The perfluorinated tail—eight carbons loaded with fluorine atoms—repels both water and oil. Add the ammonium end and you get solubility in water. It slides easily into production lines for non-stick pans, fire-fighting foams, and even waterproof clothing. From personal experience as a consumer, I remember old commercials bragging about how stains bead up and roll away. Turns out, that magic owes a debt to this specific chemical framework.
A direct look at the formula C8F15O2NH4 tells a story. Eight carbons, fifteen fluorines, two oxygens, and an ammonium unit pack a punch in stability and surface action. For chemists, that’s a red flag and a green light, depending on the aim. The stability that makes it useful also means it lingers in the environment.
The Long Shadow of Perfluorinated Compounds
Convenience always comes with tradeoffs. After all, seeing a molecule that can withstand acid, base, and heat without blinking should set off warning bells. In this case, real-life health issues stepped into the foreground. Research pointed to links between PFAS substances like Ammonium Perfluorooctanoate and health effects. Some studies have associated exposure with changes to cholesterol, liver enzymes, and immune responses.
I recall reading about community concerns around drinking water, as these compounds don’t just vanish after use. They don’t break down in the body or the environment. They earn that “forever chemical” nickname. Manufacturers across the globe have started phasing them out or cutting back, but contamination persists.
The Push for Solutions
Solutions don’t arrive overnight. Cleaning up water means investing in new filtration technologies, often at great expense. Some towns install activated carbon systems, while researchers race to find cheaper ways to capture or destroy these chemicals. From experience working with community health projects, the frustration circles back to the chemical’s basic design. If we keep building molecules that last longer than civilizations, cleanup costs sprawl into generations.
Innovation needs a new direction—safer surface treatments, biodegradable alternatives, transparency in what goes into common goods. It takes strong regulation, honest discussion, and wide cooperation. Before reaching for a pan or a raincoat, I consider not just how well it works, but what lies beneath the coating. Chemistry never happens in a vacuum, and the structure of Ammonium Perfluorooctanoate keeps reminding us of that reality.
Ammonium Perfluorooctanoate—known to many as PFOA or C8—once lived in the core of a lot of manufacturing settings. It’s hard to talk about nonstick coatings, fast-moving textiles, or some firefighting foams without bumping into PFOA’s storied legacy. Years back, I worked near a factory where nonstick coatings took shape, and the stuff they used didn’t just stay sealed away in a back room. You’d hear stories swirling about old workers and neighbors getting sick or about ponds that grew strangely quiet. Those worries aren’t rumors anymore; the facts are hard to brush aside.
Multiple studies link PFOA to higher risks of cancer, immune issues, and developmental problems. Regulators from the US EPA to European agencies keep ramping up the scrutiny. Nobody wants to admit a product stains the earth and lingers in people’s bodies for decades, but that’s the reality with PFOA. I’ve watched the switch flip—from “industry standard” to “problem chemical”—in just a short stretch of years. There’s nothing abstract about neighbors paying for industry’s convenience with their health.
So do better options really exist? The short answer: yes, but the landscape isn’t simple. Some industries try using shorter-chain PFAS molecules. These chemicals break down a bit faster and appear less likely to pile up in the food chain or in our bodies. For example, C6 chemicals—six carbon atoms instead of eight—have made waves as PFOA replacements. You’ll find them in water-repellent textiles, carpet coatings, and some new firefighting foams. These seem to move through the environment faster, which sounds better, but scientists still wonder about long-term risks.
Other makers pivot toward altogether new chemical families. Silicones, for example, add shine to textiles and offer some water resistance. Some industries have stuck with waxes or other fluorine-free coatings—especially where regulations come tightest. Food packaging firms in Europe, pressured by stricter rules, look toward plant-based barriers including cellulose and other polysaccharides. These don’t off-gas persistent pollutants and leave less of a chemical footprint.
For a guy who grew up seeing factories carry on with “business as usual,” the shift is real. Companies juggle cost, supply chain shifts, and performance expectations. Every alternative has pros and cons—cost and durability come up most often. No one working in a manufacturing plant wants to cut corners on quality just to keep regulators happy. Plenty of us grew tired of hearing, “It’s the only thing that works.” Today, reformulation teams run real-world trials, dig deep on chemical safety data, and tap into academic labs for green chemistry breakthroughs.
Governments and communities play a role: pressure matters. When one city or state bans a compound, industry leaders actually start listening. Companies find budget for R&D that just wasn’t there five years ago. Full transparency in supply chains helps buyers vote with their wallets. Local watchdogs and employees alike can ask for safer ingredients before another chemical legacy gets etched into local rivers or workers’ bones.
Replacing PFOA isn’t all about flawless chemistry; it’s also about listening—to scientists, to neighbors, to workers, and to fresh ideas. The payoff goes well beyond compliance; it comes from putting long-term health before short-term gain. That’s worth a few growing pains and some extra days sorting out new coatings and processes. Industry made this mess; industry can—and should—find a better way out.