Ammonium Trifluoroacetate: More Than Just a Chemical Formula
Historical Development
The journey of ammonium trifluoroacetate tells something about how modern chemistry evolved alongside new technological demands. Chemists searching for powerful, selective reagents in the mid-1900s found that the influence of trifluoromethyl groups opened the door to unique reactivity. Pairing trifluoroacetic acid with ammonium sources brought ammonium trifluoroacetate into the lab. Since then, the substance has earned a niche reputation. I remember working in an academic lab where a seasoned researcher swore by its reliability in mobile phase preparation for high-performance liquid chromatography. Many analytical chemists recall its impact in improving detection limits and data quality, reminding us how breakthroughs often start with “let’s try something new.”
Product Overview
Ammonium trifluoroacetate stands out for more than its formula, NH4CF3CO2. As a white, hygroscopic crystalline powder, it shows up where precision matters—particularly in techniques like mass spectrometry and protein sequencing that rely on minimal interference. Chemists choose it for its capacity to act as a volatile buffer. It dissolves swiftly in water, methanol, and acetonitrile—common solvents in many analytical workflows. It stays out of the way, so to speak, before evaporating during key steps. This minimal residue feature is far from trivial in sensitive environments where detection errors can wreck weeks of work. More and more, it shows its value outside traditional boundaries, making its mark in research labs and quality control settings.
Physical & Chemical Properties
Handling ammonium trifluoroacetate, one quickly notices its tendency to absorb water from the air. This hygroscopic character means airtight storage matters—not just as a footnote, but as a core lab practice to protect sample consistency. The compound features a melting point close to 123 °C and breaks down when things heat up too far, releasing fumes that include ammonia and hazardous hydrogen fluoride. Its pH in aqueous solutions leans acidic, reflecting the strength of the trifluoroacetate anion. These properties shape how people treat, store, and dispose of it—from bench chemistry to waste disposal protocols. Many in the scientific community favor it over traditional acetate salts for these unique analytical and physical traits, not just for its volatility, but for the clarity it brings to modern investigations.
Technical Specifications and Labeling
Labeling on ammonium trifluoroacetate typically points out its purity, water content, and lot number because these details matter directly for reproducibility. Purity influences background noise in analytical instrumentation, so most labs aim for upwards of 98 percent or better. The best supplies carry documentation that tracks the product from synthesis to shipment. For those working with highly sensitive assays, a difference of even a fraction of a percent can mean hours spent troubleshooting, so the devil truly is in the details. Tracking batch quality represents more than bureaucracy—it builds trust and helps labs spot problems before they interfere with results.
Preparation Method
The process to make ammonium trifluoroacetate typically starts simple but requires care. Mixing trifluoroacetic acid, itself handled with caution, with aqueous ammonia gives the salt in a straightforward acid-base reaction. The end product must go through careful crystallization and drying steps to purge excess moisture—hygroscopicity strikes again—and achieve a quality batch. Small variations in stoichiometry or drying technique can mean the difference between a reliable, dry solid and a tacky mess prone to clumping. It’s not just about following a recipe; routine errors, like incomplete removal of water, risk changing concentrations and undermining the reagent’s performance. Attention to method and process separates a reagent-grade product from something less usable.
Chemical Reactions & Modifications
In the lab, ammonium trifluoroacetate’s roles expand well beyond serving as a buffer. Its ability to introduce a trifluoroacetate group into molecules has helped unlock synthetic pathways unavailable with regular acetate. Researchers modify peptides with it when cleaving resin-bound structures or during protein sequencing, benefiting from its volatility and purity. It dissolves easily and leaves little trace, so synthetic chemists favor it when side reactions with sodium or potassium salts threaten to muddy reaction outcomes. Over the years, investigations into new functionalizations keep cropping up in journals—some exploring better protection groups, others testing milder cleavage conditions. Chemical storytelling doesn’t stop; with every breakthrough, we see how this “simple” salt powers transformations reaching far outside textbooks.
Synonyms & Product Names
Within the literature and on product shelves, ammonium trifluoroacetate goes by names like ammonium trifluoroacetate salt and ammonium 2,2,2-trifluoroacetate. These seem minor, but consider how even small variations can cause confusion, especially in international communications or multidisciplinary teams. Researchers pulling data from various sources must check they’re talking about the same thing—incorrect assumptions slow down work, and inconsistent naming sparks errors in everything from safety documentation to ordering. This issue highlights why standardized cataloging matters, not as an abstract concern, but as an everyday part of reliable science.
Safety & Operational Standards
Safety surrounding ammonium trifluoroacetate can’t be handwaved, especially given the presence of trifluoromethyl groups and the risk of toxic fumes at higher temperatures. Improper handling exposes workers to burning eyes and respiratory tract irritation, so good ventilation and protective equipment become more than poster slogans. Even packaging—tight lids, desiccants, labels confronting risks directly—makes a big difference in preventing mix-ups or accidents. Staff training leads to a workspace where everyone’s watching for early signs of decomposition or exposure rather than cleaning up accidents. Emergency protocols and waste disposal strategies reflect modern regulatory expectations, with trace chemicals disposed of as hazardous waste, not down the drain. This attitude fosters a culture where people look out for each other in an environment that can change fast.
Application Area
The molecule’s most visible role shows up in analytical chemistry—mobile phase additives in chromatography, protein sequencing, and mass spectrometry top the list. Scientists selecting ammonium trifluoroacetate appreciate its clean volatility; you don’t have to worry about stubborn nonvolatile residues trashing sensitive equipment or throwing off trace analysis. In some peptide synthesis and degradation protocols, the salt allows for gentler handling without harsh acids that risk damaging fragile products. Once you look past core analytical circles, more industrial chemists and life-science researchers latch on to its value. Its acceptance grows in emerging areas like environmental monitoring and clinical diagnostics, where clean baselines and low detection thresholds call for exceptional reagents. This broad appeal springs from technical merit, not marketing spin.
Research & Development
Teams in R&D push the reagent in new directions, testing advanced synthesis routes or exploring broader uses in green chemistry. Efforts to minimize ammonia release, promote complete volatilization, or tailor the product’s impact on peptide fragmentation patterns signal a maturing scientific understanding. Best practices emerge through direct experience—learning which impurities have measurable effects, figuring out how to minimize batch-to-batch variation, or even trialing alternative anion-cation pairings. The dialogue between bench chemists and suppliers improves product quality over time, making this salt part of an ongoing story rather than just another lab staple.
Toxicity Research
Toxicity remains a concern that’s sometimes underestimated. Acute exposure studies point to respiratory and mucous membrane irritation, while the long-term effects remain incompletely charted, especially for those regularly handling the salt in poorly ventilated labs. The fluorinated portion of the molecule demands respect: accidental decomposition releases hydrogen fluoride, one of the nastiest substances a chemical worker can face. Throughout my own experience, working with colleagues to double-check containment protocols or share real-world incidents led to better, safer environments, showing that knowledge transfer beats assumption every time. Robust research into handling risks, including in animal studies and simulated spill scenarios, keeps regulations grounded in field reality.
Future Prospects
The future for ammonium trifluoroacetate looks busier—not just in classic applications, but potentially in emerging separation techniques and greener chemistry practices. The demand for analytical methods capable of pushing boundaries pulls interest towards reagents with proven reliability and new possibilities for customization. With ongoing progress in automation, online sample cleanup, and microfluidic devices, the call for purer, more consistent additives grows stronger. Research into recycling or remediating volatile buffers ought to get more attention to reduce environmental impact. There’s also movement in regulatory circles: increased transparency about production and disposal feeds into both public trust and scientific integrity. The story of this unassuming salt reveals that even the simplest chemicals can evolve into something with an outsize influence, guided by shared experience, careful stewardship, and an eye to what might come next.
Peeking Behind the Lab Door
Most people never hear about ammonium trifluoroacetate, unless their daily routine brings them close to a chemistry lab or a scientific supply catalog. Yet, this compound plays a role in the kind of work that makes our technology sharper and our medicine more precise. In my time navigating the intersection of science communication and practical lab work, I’ve watched researchers rely on seemingly obscure chemicals like this one far more often than the average person might suspect.
Chemical Workhorse in Modern Labs
Ammonium trifluoroacetate mainly shows up in analytical chemistry, specifically in high performance liquid chromatography (HPLC) and mass spectrometry. People often talk about the importance of accuracy and sensitivity when checking for contaminants in our food, water, or pharmaceuticals. Professionals in these fields mix ammonium trifluoroacetate into their processes because it can act as a buffer, helping to control the pH and improve reproducibility of results. It does so without clogging things up or leaving behind unwanted residue, which saves time and protects valuable equipment.
Making Complex Analysis Simpler
Years ago, I spoke with a colleague at a biotech startup who barely had enough funding to replace their HPLC columns, let alone toss them out early due to erratic chemical reactions. Consistency means everything for them. Ammonium trifluoroacetate provides a trifluoroacetate component that ionizes cleanly—a real advantage for detecting subtle chemical differences. Anyone who’s spent hours troubleshooting a messy chromatogram knows the relief that comes from switching to a buffer that delivers reliable, consistent results. There’s no dollar value on a good night's sleep after you finally crack a stubborn separation problem.
Problems for People, Not Just Products
No one should ignore the fact that chemistry doesn’t happen in a vacuum. Fluorinated compounds like this one bring environmental and health discussions, especially as more evidence links perfluorinated substances to persistent pollution. While ammonium trifluoroacetate itself stands apart from some notorious persistent chemicals, safety in handling and responsible disposal make up essential parts of the conversation. People in the lab wear gloves and work under extraction hoods for a reason, and regulatory agencies keep an ever-closer eye on chemicals with trifluoro groups.
Solutions Take Everyone
Scientists and companies should invest in safer workplace practices and push for greener chemistry alternatives. Training sessions for young researchers aren’t just for tradition; they foster a culture of respect for both safety and sustainability. At the same time, industry partners need to support moves toward less hazardous alternatives whenever practical. Open dialogue between chemists, suppliers, and regulators can steer the future use of compounds like ammonium trifluoroacetate to balance scientific needs with responsible stewardship of our shared environment.
The Value Hidden in Everyday Science
The impact of something like ammonium trifluoroacetate rarely reaches public debate. Yet, it’s these small building blocks and the choices experts make each day that keep society’s trust in science strong. Whether someone is checking a heart drug for impurities or analyzing traces of toxins in soil, the reliability that comes from understanding and wisely using chemicals like this earns its keep far beyond the bottles on the shelf.
Getting Down to the Molecules
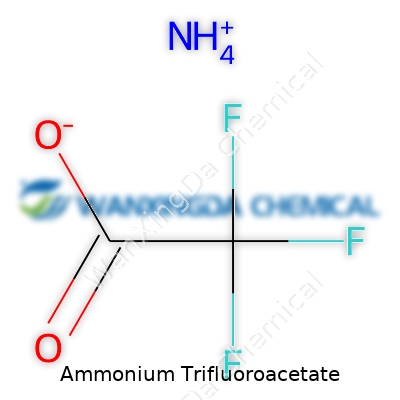
I remember my first run-in with trifluoroacetate salts back in college. It was a time when the periodic table felt like a map with mountains waiting to be climbed. Ammonium trifluoroacetate sounds like a mouthful, but at its core, it’s just a combination of ammonia and trifluoroacetic acid—nature’s chemistry, with a twist. The chemical formula, CF3COO−NH4+ (or, just as simply, C2H4F3NO2) lays out exactly how each atom lines up.
Why the Formula Tells a Bigger Story
Understanding a chemical formula offers more than a simple atomic headcount. This salty marriage of ammonium ion and trifluoroacetate anion spells out real-world features. Trifluoroacetate brings electronegativity from those three fluorine atoms hitching a ride on the acetate backbone, tightening up the structure and making it much less friendly to biological life than plain acetate. That change in structure holds serious weight in lab settings and in any process that involves breaking down or producing molecules using this salt.
I’ve seen chemists reach for ammonium trifluoroacetate for peptide synthesis or as a buffer in mass spectrometry. That’s not a random choice. Take ammonium, for example—a familiar face in everyday cleaning supplies. It binds with trifluoroacetate, a much less common component, and together they make a solid that dissolves nicely but doesn’t leave gunk behind. These features come straight from the formula, reflecting how every letter and number points to a specific role in the lab.
Everyday Impact and Environmental Considerations
Folks sometimes overlook where chemistry meets the street. Trifluoroacetate compounds crop up in industry and research labs, but their chemical toughness can also create headaches downstream. Because of the carbon-fluorine bond’s legendary stubbornness—something the formula unpacks for anyone paying attention—these molecules can linger in water and soil. As a student volunteer in a water quality lab, I saw how persistent these compounds can be. They don't just slip away with the next rainstorm but stand their ground in the environment long after the experiment is done.
That kind of staying power comes directly from the blueprint encoded in the chemical formula. The environmental persistence of fluorinated compounds has already caught the attention of agencies like the EPA and researchers tracking "forever chemicals" in water supplies. Even if ammonium trifluoroacetate itself doesn’t top regulatory lists, its structural cousins do, making it a case worth understanding and monitoring. Good stewardship—something I learned from supervisors who never let us cut corners in waste disposal—means keeping a close eye on how these chemicals travel and what they leave behind.
Finding Smarter Solutions
People working daily with compounds like ammonium trifluoroacetate face a puzzle: keep enjoying the benefits in synthesis, or look for alternatives that don’t hang around in the environment. Some labs are moving toward using less persistent acids or even designing custom buffers that break down faster. It’s a push coming from people who recognize that every number and letter in a chemical formula carries responsibility. Regulations may steer the ship, but personal choices pile up, too—as any scientist swishing out the last bit of solution at the sink knows.
Understanding ammonium trifluoroacetate’s formula isn’t just a question for textbook quizzes; it’s a prompt for anyone working with chemicals to pause and ask, “Where does this lead?” That simple combination of elements carries impact beyond the page.
The Story Behind the Chemical Name
Ammonium trifluoroacetate sounds like a mouthful. In the chemistry lab, it’s a tool that keeps reactions humming. It pops up in peptide synthesis, chromatography workflows, and some pharmaceuticals. Schools and industry folks use it because it’s reliable for very particular jobs. Most of us don’t run across it at the grocery store. Still, interest bubbles up anytime the conversation turns to safety.
What Science Says About Hazards
No one should play fast and loose with a compound just because it has scientific uses. Look at ammonium trifluoroacetate’s building blocks. It draws from ammonia and trifluoroacetic acid (TFA). Ammonia can burn and irritate skin, eyes, and lungs. Trifluoroacetic acid sits among the harsher acids (even at low concentrations, it eats away at tissues and stings noses). The salt form, which is ammonium trifluoroacetate, tends to show lower volatility and behaves more mildly—but not enough to shrug off basic precautions.
Safety data sheets call attention to potential risks: eye and respiratory irritation, skin sensitivity, and possible toxic effects with enough exposure. Studies on rats and other animals link large doses and chronic exposure to organ trouble, although most lab professionals see only brief, low-level contact. Getting ammonium trifluoroacetate on skin can cause redness or burning. Breathing in powder or aerosol can irritate the throat. Not many studies track long-term impact on humans, but no data gives a green light to careless handling.
Why Risk Matters Outside the Lab
With chemicals like this, there’s a bigger debate. Trifluoroacetate compounds draw scrutiny for their environmental stubbornness. The “trifluoro” part means chemical bonds that don’t break easily. This raises flags about persistence in water, soil, and living organisms. In some places, these compounds show up downstream of factories and high-use sites, leading to arguments about waste treatment and whether to tighten chemical regulations.
Best Ways to Stay Safe
Sturdy gloves, lab coats, splash goggles, and a fume hood aren’t just for show. Chemists who mix or weigh the compound rely on these barriers. Nobody wants a cloud of fine chemical dust or a splash in the eye. Spills need quick attention with absorbent material meant for hazardous stuff. Leftover ammonium trifluoroacetate never belongs in the regular trash or down the sink; it calls for a licensed hazardous waste facility.
Good training, clear labels, and respect for the unknowns in chemical safety go a long way. Rules do differ by country. In the U.S., the EPA keeps one eye on the release and disposal of perfluorinated and trifluorinated compounds. The European Union lists similar concerns. In either case, heavy fines and closer inspections now greet companies ignoring the rules.
A Shift Toward Greener Chemistry
There’s a push for safer alternatives as the world learns more about molecules like ammonium trifluoroacetate. Some research teams look for substitute salts or greener reaction helpers, though they face an uphill climb. As new generations of chemists enter the lab, their training leans more toward prevention, not just cleanup. Strict tracking, airtight containers, and real buy-in from leadership help cut risks to workers and the wider community.
Direct experience with these chemicals sticks with you. Reading labels with a critical eye helped me avoid nasty surprises in the lab. Everyone benefits from sharing practical habits and pressing for smarter industrial choices. The ultimate goal: get the perks of advanced chemistry without sidestepping health or the planet’s welfare.
Why Attention to Details Matters with Chemicals
Anyone who’s worked in a lab knows that not every compound deserves equal casualness. Some cause the sort of trouble that can turn a regular day sour in a hurry. Ammonium trifluoroacetate isn’t a household name, but those in research or industrial settings have a good reason to put it on their radar. It’s a sharp reminder that one slip with a small vial can mean real consequences, for people and the environment.
Storage Is More Than Stacking Bottles
Shoving bottles onto a shelf might save a few minutes, but it often costs a lot more down the road. Ammonium trifluoroacetate brings together volatile fluoride chemistry and ammonium’s tendency to go reactive. The safest spot for this compound stays cool, dry, and out of direct sunlight. I’ve seen labs lose expensive reagents because temperature spikes degraded sensitive chemicals—money down the drain and an experiment lost.
Keeping moisture out remains key. Once this compound starts pulling water from the air, purity drops and unpleasant changes follow. A tightly sealed container does more than tidy the lab; it protects investments and health. No one needs the hassle of cleaning up after a chemical that’s gone off just from poor storage habits.
Personal Safety Doesn’t Take a Break
There’s always the temptation to skip gloves or zip through an open bottle for “just one quick use.” Over the years, it’s become clear: shortcuts here pay in skin rashes, respiratory issues, or hours filling out incident reports. Ammonium trifluoroacetate isn’t known for kindness to unprotected skin or lungs, so gloves, goggles, and a proper fume hood become non-negotiable. Eyewash and emergency showers should never gather dust in the corner.
I’ve watched colleagues sneeze for hours after an unexpected whiff of chemical dust. Particulate matter from fluorinated compounds settles deep in the lungs, with possible toxic effects. One good lab rule: if you think a respirator seems overkill, reconsider—your lungs and future self will thank you.
Spills, Waste, and Environmental Impact
Spills happen. The key is not pretending otherwise but planning for the day something tips over. Spill kits tailored for hydrofluoric and ammonium compounds need a place close at hand. Encouraging a culture where nobody feels embarrassed about reporting a mishap does more to keep workspaces safe than any rulebook alone.
As for waste, it doesn't blend harmlessly into the environment. Everything with strong fluorine chemistry asks us to think twice before loosening restrictions on disposal. Every gram poured thoughtlessly down the drain threatens water systems nearby. Following hazardous waste guidelines isn't just box-ticking; it keeps communities safer and often keeps businesses out of legal trouble too.
Solutions: Building Better Habits
Lab culture shapes how people treat risky chemicals. In places where training happens often, accidents drop. Hands-on demonstrations—how to cap a bottle right, where to put a spill kit—stick in memory better than posters nobody reads. Supervisors who refresh protocols and encourage lab workers to ask questions build confidence and safety.
Trust builds not from memorizing every hazard but from knowing the tools and habits that work. Storing ammonium trifluoroacetate correctly and respecting its handling rules turns what could be a daily hazard into a valuable tool. There’s pride in a smooth-running lab, where everyone gets home with nothing worse than a little fatigue.
The Curious Case of Ammonium Trifluoroacetate in Water
People working in research or production know the frustration of an ingredient that just will not dissolve. Ammonium trifluoroacetate comes up often in labs, especially for folks dealing with chemical analysis and organic synthesis. Throw it in water, and you quickly see why chemists favor this salt. It dissolves with little trouble, leaving a clear solution even at different concentrations. For those of us trying to get clean reaction mixtures or reliable HPLC buffers, that reliable water solubility matters more than any catalog description could suggest.
Trifluoroacetate’s strong pull on electrons and the nature of the ammonium ion both help this salt break apart easily as soon as it touches water. I have watched how quickly it can clear up a cloudy buffer, and that alone can save time with filtration and preparation. You do not worry about clogging up columns or fouling precision equipment with gritty residue. It pays off every day for labs running large batches or precise analytical setups.
Other Solvents and Where It Struggles
Water tends to be the home turf for ammonium trifluoroacetate. Try dissolving it in nonpolar solvents like hexane or diethyl ether and you face disappointments—a chalky film, undissolved powder, and wasted effort. Polar solvents such as methanol or ethanol do a decent job, though not quite as smooth or complete as water provides. In routine use, most folks stick with water unless a specific method calls for something stronger.
Impact on the Lab and Beyond
Strong water solubility plays a role in safety and environmental impact. Spill a solution, and cleanup feels much easier, no lingering residues or risky dust suspensions. For anyone scaling up from bench to pilot or full production, it means safer handling and fewer calls to hazardous response teams. Waste streams also become less troublesome, since you can dilute and neutralize aqueous mixtures with less risk.
There is no such thing as a perfect chemical. One thing with ammonium trifluoroacetate is that strong solubility could mean it spreads fast if it enters waterways. Without proper waste management, it has the potential to slip into sewage or rivers. Labs and companies need strong containment plans and routine monitoring. Investing in robust wastewater treatment ensures nobody downstream ends up dealing with unexpected contamination. Paying attention to local regulations and best practices—instead of cutting corners—prevents issues for communities and the environment.
Finding a Balance between Utility and Responsibility
I have seen the frustration that comes with chemicals that fail to dissolve, and the relief when a predictable, water-loving salt like ammonium trifluoroacetate knocks down one more laboratory hassle. The magic is not just in clear flasks; it is also in safer handling and responsible disposal. Every lab can take that extra step: strong solvent-resistant containers, up-to-date spill kits, and clear waste protocols. Sharing experiences through reliable reporting and open data will help chemists and industries keep using this useful salt without adding headaches for anyone else. Real expertise grows from both knowing what a compound does—and from the choices we make after seeing its strengths and weaknesses up close.