Benzoic Acid: A Look at Its Journey and Real-World Impact
From Early Curiosity to Industry Backbone
People stumbled onto benzoic acid centuries ago while tinkering with gum benzoin, a resin pulled from tree bark and coveted for its fragrance. Even in the 1500s, early chemists noticed unusual crystals forming when they heated this resin, though a real breakthrough took hold in the 19th century when German scientists figured out how to isolate benzoic acid in its pure form. This single discovery unlocked a line of products and industries that have changed daily routines, made food last longer, and spurred innovation in countless fields. Over time, commercial production leaned toward oxidizing toluene, which drove the cost down and opened up new bulk markets. Watching how such a simple molecule shaped preservation methods, plastics, and even pharmaceuticals says a lot about how small, persistent scientific efforts can change how billions of people live and eat.
A Clear Picture of Benzoic Acid
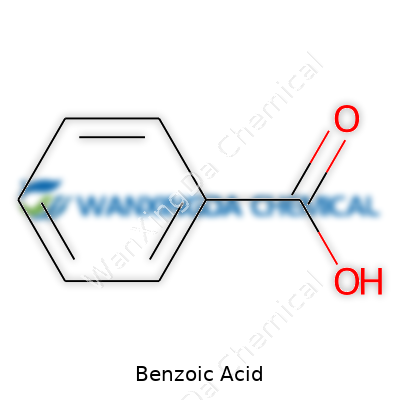
What stands out about benzoic acid is its solid form: white, flaky crystals with a slightly sweet aroma, unlike harsh industrial chemicals. Its chemical formula, C7H6O2, hides a versatility that few might suspect from something so mundane. Around room temperature, it doesn’t melt unless you apply real heat—over 120°C. Its solubility in water feels limited, but once in hot water or a bit of alcohol, it dissolves much more easily. This property alone shapes how manufacturers store, handle, and apply benzoic acid in day-to-day products. In a lab, that balance of stability and reactivity means chemists use it as a building block for even more complex molecules. The pure form carries a distinct tartness, a trait that shows up in the tiny doses added to food as a preservative, keeping mold and bacteria at bay for longer stretches.
Getting Technical with Labeling and Quality
Meeting strict technical guidelines matters every time benzoic acid heads from a plant to a shelf. Purity usually stays above 99%, since even minor contamination could trip up anything from pharmaceuticals to food safety. Labels carry not just purity but warnings about dust inhalation, storage temperatures, and incompatibilities with strong oxidizers. Detailed batch records and analysis back every shipment, especially in jurisdictions where consumer safety regulations run tight. In my experience, facilities that cut corners with benzoic acid quality face real headaches—anything from spoiled products to withdrawn batches. Simple as its chemistry may seem, getting benzoic acid safely from factory to consumer requires a disciplined, precise approach at every link in the supply chain.
Common Preparation and Old-Fashioned Chemistry
Labs and factories don’t always follow textbook methods, but one of the most common routes to benzoic acid today involves oxidizing toluene—a major ingredient in gasoline and paint thinner. By introducing enough oxygen under catalyzed conditions, toluene sheds hydrogen atoms and forms benzoic acid, which workers then purify and crystallize. Before this route took over, people used saponification of benzoin resin and even unique fermentation methods, but these older approaches became rare as demand surged and costs dropped. This shift away from artisanal production made benzoic acid affordable enough to slip into everyday products. Growing up in a small community with home canning traditions, I saw how food preservatives like benzoic acid made a difference in keeping pickles, jams, and juices fresh long after summer ended—proof that shifts in production affect real-world eating habits.
Benzoic Acid in the Wild: What Reactions Teach and Enable
Simple as it looks, benzoic acid plays a part in dozens of chemical reactions. By reacting with alcohols, it forms esters, which bring a pleasant scent to perfumes and flavorings. If you add strong bases, it becomes a salt, benzoate, which dissolves well and acts as a go-to preservative in acidic foods and soft drinks. In a factory or research lab, you might see it converted to benzoyl chloride—a useful ingredient in dye, plastic, and pharmaceutical manufacturing. These chemical tweaks unlock doors: benzoic acid itself seldom ends up in a final, shelf-ready product, but its derivatives show up in spots as diverse as wound ointment tubes, soda cans, and even everyday cleaning supplies. By changing one atom or reshuffling a group here or there, chemists shape an enormous range of consumer goods.
This Acid by Any Other Name
People might spot benzoic acid under all sorts of labels: E210 on food ingredient lists, benzenecarboxylic acid or carboxybenzene in chemical catalogs, or even drily as "benzeneformic acid." Each name signals a different world—food production, research chemistry, even environmental science, where trace amounts show up in soil and water monitoring. While these synonyms can trip up new learners, understanding what each name signals helps sort out whether something’s going into a soft drink, a prescription cream, or an industrial tank.
Staying Safe with Acidic Tools
Benzoic acid’s reputation for relative safety rests on using it the right way, in the right amounts. Too much exposure to dust can irritate airways, and for factory workers, gloves and masks provide real protection against skin or lung irritation. For the everyday person eating foods preserved with benzoic acid, typical doses fall far below safety thresholds set by health bodies. The trouble usually starts in workplaces that slack on ventilation or ignore routine cleaning, so management sets the tone by investing in worker protection and sound training. My background in food safety regulation impressed on me that regular audits and clear labeling on bulk containers kept both staff and end users informed—an overlooked, but essential step in risk management.
A World of Uses Across Fields
Benzoic acid made its greatest mark in food preservation, but its reach stretches a lot further. Rubber manufacturing depends on it as a retardant; it slows how fast rubber cures, giving product makers better control. Cosmetics use its esters for scent and feel, lending lotions and creams a subtle fragrance without overpowering the senses. Modern medicine leans on benzoic acid as both an ingredient and an intermediate—think antifungal ointments or cough syrups. Even plastics like polyester trace a thread back to this unassuming acid. Years as a quality control manager taught me to look for upstream chemical origins whenever a product performed better or worse than expected. Over and again, the footprint of benzoic acid popped up where people least anticipated.
Today’s Research: Beyond the Status Quo
Current research circles back to examining how benzoic acid behaves in combination with other additives, especially as food and beverage companies look for cleaner labels and natural preservatives. Advanced detection methods give a leg up in tracking even trace levels, keeping tabs on cumulative exposure. In pharmaceutical R&D, benzoic acid’s simple structure offers a model for new drug candidates, especially as researchers probe drug metabolism, absorption, and shelf stability. Environmental science takes its own interest in benzoic acid, using it to monitor breakdown rates of organic pollution or as a marker for industrial discharge. As more restrictions land on synthetic additives, development of green production methods for benzoic acid gains ground, with researchers turning to renewable feedstocks—part of a broader shift to cut reliance on petroleum-based raw materials.
Digging into Toxicity: Facts and Real-World Risks
Debates about benzoic acid toxicity often stir up strong opinions, but the science stays pretty clear: at authorized low levels in food, the risk remains low for most healthy adults. Animal testing has shown that massive, unrealistic doses can stress internal organs, but those situations fall far outside typical dietary patterns. There is some concern about benzoic acid forming benzene, a known carcinogen, in certain acidic conditions with the presence of ascorbic acid (vitamin C), mainly in poorly stored soft drinks. Regulatory agencies set strict upper limits to address these edge cases. Keeping intake well below these thresholds stays the norm almost everywhere. Still, anyone in food safety learns that small subgroups, like children or people with rare metabolic issues, need extra attention, so guidelines usually err on the conservative side to protect vulnerable folks.
What Lies Ahead for Benzoic Acid
Looking ahead, benzoic acid faces both opportunity and scrutiny. Alternative preservatives—a rising trend as consumers push for “clean” food labels—compete for the same shelf space, but benzoic acid stands strong thanks to its proven, dependable track record. More sustainable manufacturing will likely reshape where benzoic acid comes from, as new methods use plant-based inputs or leverage fermentation instead of chemical synthesis. In industrial and pharmaceutical chemistry, benzoic acid will remain a faithful stepping stone, since its value as a simple, reliable base for other compounds can’t be easily replaced. As research deepens, expect new uses in advanced materials, biomedical devices, or even as a probe in environmental monitoring. Innovation often blooms in the margins, and for benzoic acid, a long history of utility points to a future where it keeps surprising people across every field touched by science and manufacturing.
Open your pantry or medicine cabinet and chances are, benzoic acid has already made its way into your life. People may not give much thought to the fine print on food packaging or the dense ingredients lists on personal care products. The truth is, this simple white powder shows up far beyond chemistry labs, shaping the way we eat, clean, and care for ourselves.
Food on the Shelf—Not by Accident
Walk into any grocery store, and the invisible battle between spoilage and preservation rages on every aisle. Benzoic acid lands right in the middle of that fight. Acting as a food preservative, it helps slow down the growth of bacteria, yeast, and mold. Without some sort of barrier, jams, sodas, and pickles would grow fuzzy almost as fast as they’re opened. The US Food and Drug Administration caps its level in foods to keep things safe, but most consumers probably never notice it’s there.
The impact? Fewer spoiled goods, less waste, and shelf-stable foods that can travel longer distances. That means local stores can stock tropical flavors or global imports, which would be impossible without preservatives like benzoic acid. Food manufacturers rely on it not for profit but for practicality—it simply works. It keeps production lines running smoothly by cutting down on returns and complaints, and it allows people to enjoy strawberries in December without worrying about whether the jam has gone bad overnight.
Packed in Personal Care
Benzoic acid steps out of the kitchen and into the bathroom. If you read the ingredient list on lotions, shampoos, or even toothpaste, you might spot its name. Beyond fighting spoilage in food, the acid helps keep bacteria and other germs at bay in products that sit on shelves or counters for weeks. No one wants cosmetics that go sour or creams that grow strange colors. This ingredient means you don’t have to think twice about your favorite moisturizer going off after a few uses.
Pharmacy Staples
Pharmacies don’t miss out either. Ointments and creams for athlete’s foot or other fungal ailments often rely on benzoic acid. Its mild antifungal and antibacterial traits make it a dependable, economical choice. The world doesn’t need endless new chemicals if a tried-and-true compound gets the job done with minimal health risks. Medical professionals prefer clear, well-understood ingredients over unknowns, and benzoic acid’s track record speaks for itself.
Safety and Public Concerns
Everything on a label comes with questions. Over the years, benzoic acid faced scrutiny. Some worry about potential health effects, especially with long-term consumption or in combination with certain other additives. If heated with vitamin C, it can form benzene, a known carcinogen. Regulators keep a tight eye on how it’s used. Following rules about concentrations and combinations dramatically reduces any risk.
Companies would do well to stay transparent. Keeping ingredients and concentrations in clear language builds trust. More importantly, science-backed regulation and routine safety reviews give everyday shoppers the assurance that no ingredient, no matter how old-school, gets a free pass.
Better Solutions Start with Questions
There’s room to keep pushing for safer food and consumer goods. Research into alternatives continues, but sometimes the old solution works better than the untested new one. Phasing out preservatives where better cold chains or packaging exist makes sense. Using innovations like “clean labeling” helps those who want to limit chemical additives. Balancing shelf life, safety, and consumer demand for simplicity will always require smart, ongoing dialogue among food scientists, regulators, and the public. Benzoic acid isn’t leaving shelves any time soon—but questioning its place keeps everyone safer and more informed.
Why People Care About Benzoic Acid
Take a look at the back of most popular soft drinks, jams, and even pickles, and you’ll probably spot benzoic acid or its cousin sodium benzoate. This food additive helps keep mold, bacteria, and spoilage at bay. Food safety standards developed over the last century stick to using preservatives like benzoic acid, especially in foods with longer shelf lives. People want their food to last longer and stay fresh, and benzoic acid delivers on that promise.
Understanding How Much is Safe
Regulators around the world, including the US Food and Drug Administration and the European Food Safety Authority, have given benzoic acid a green light within certain limits. The FDA says foods should not have more than 0.1% benzoic acid by weight. Actual levels found in most products usually sit well below this cap. Scientists determined this amount after studying how people’s bodies react. After eating foods with benzoic acid, the body breaks it down and gets rid of it in urine, so it doesn’t build up over time.
Potential Health Concerns
Some people worry about possible effects when consuming large amounts of benzoic acid. Studies found that extremely high doses—much higher than those allowed in foods—could cause problems in lab animals. Nausea, stomach irritation, and allergies sometimes come up as concerns from folks who believe they’re sensitive to preservatives. Another hot topic has been the creation of benzene, a known carcinogen, when benzoic acid mixes with vitamin C in the presence of heat and light. The FDA and food makers have taken steps to limit this reaction, tweaking formulations to keep benzene levels extremely low, far beneath levels thought to raise cancer risks.
Who Might Want to Cut Back
Kids and people with specific allergies or medical situations may want to watch out for foods loaded with preservatives, including benzoic acid. Some health professionals suggest those with asthma or hay fever seem to notice more sensitivity, reporting symptoms like hives or breathing trouble, though clear links remain rare. For most healthy people, occasional intake—even a regular can of soda—stays far from risky territory.
Finding the Balance
Some shoppers prefer eating fewer processed foods, taking “no artificial preservatives” labels seriously. Others take comfort that their favorite drinks or fruit spreads won’t go bad right after opening. For many, it’s about balance—enjoying the convenience of modern foods while understanding what’s added to them and why.
Learning to read labels helps make those decisions easier. Nutritional transparency has improved with regulations requiring clear ingredient lists. If benzoic acid raises personal concerns, plenty of preservative-free or fresh alternatives exist. Cheesemongers, bakers, and farmers’ markets rarely use these additives.
What Works for You
Everyone draws their own line about what feels safe to eat and drink. As science digs deeper, it pays to follow updates from credible health authorities, doctors, and registered dietitians. My own kitchen sees both store-bought and homemade foods, and I appreciate the convenience preservatives offer, especially on busy weeks. Still, nothing beats the taste of produce straight from the garden, and it never hurts to pause and check what’s in the things we eat every day.
Simple Stuff, Real Risks
Benzoic acid pops up all over the place, from foods to personal care products. This white, powdery compound gets a rep for being simple and easy to handle, but treating it like any generic chemical risks bigger headaches than most people realize. It’s tempting to tuck it on a shelf and call it a day because that’s what some old-timer in a shop told you once. Thing is, ignoring proper handling can put both health and product quality at risk.
Moisture and Clumping Ruin Your Day
Benzoic acid absorbs moisture from the air, and this turns it into a lumpy mess that’s hard to work with. Clumping ruins batch consistency, making it impossible to measure or mix accurately. Toss in humid storage, and you might find mold growing in food additives or granules sticking in machines and bagging lines. Dry storage wins the day. Cool, sealed containers with a tight-fitting lid keep water out. Just like flour in a bakery—leave the bag open and it cakes up, messing with today’s recipe and tomorrow’s too.
Why Air Quality Isn’t Just Hot Air
Dust matters. Benzoic acid dust drifts loose in a workspace and hits your nose or lungs. It’s not as notorious as some solvents or fibers, but repeated exposure, especially in tight places, can bring on respiratory irritations and mild allergies. Decades in labs and small production rooms have shown that a well-ventilated storage area lets workers avoid sneezing fits or worse. Nobody wants to cough their way through a shift just because someone skipped keeping the lid shut.
Don’t Mix with the Wrong Crowd
Keeping chemicals apart isn’t always about drama or big explosions. Benzoic acid reacts with strong oxidizers, leading to heat and, in rare cases, hazardous byproducts. I remember a facility where careless stacking put cleaning products and food preservatives together—one small spill was all it took for a nasty mess. Segregation by compatibility, a habit in every serious operation, prevents chemical cross-contamination and potential hazards. Color-coded bins and clear labels go a long way, even for staff changing shifts in a big warehouse.
Strong Containers, Smarter Safety
Always go for containers made of materials that hold up, such as plastic or glass. Metals can corrode with prolonged contact, shifting small bits into the benzoic acid. Someone once stored a bag in an old tin—it rusted and crumbled, contaminating half a year’s supply meant for preservation. Tamper-evident seals keep out both moisture and careless hands. No one likes dumping an entire stockpile over one avoidable mistake.
Common Sense Beats Overkill
Locking up benzoic acid doesn’t call for elaborate climate controls unless you deal with bulk or pharmaceutical grade. In most businesses, it’s about a dry, shaded space that workers keep tidy and free from reactive chemicals. Regular checks make a difference. If a container looks bloated or leaky, replace it right away. Respecting storage basics protects both the product and the people who handle it daily. Experience shows most storage disasters start small—a cracked lid, a missed label, or a forgotten container in a damp corner. Addressing these small things saves money and headaches in the long run.
Smarter Habits for Safer Work
Routine attention and well-organized shelves matter more than fancy equipment. Training every worker to spot problems and fix them quickly has saved me, and plenty of others, from setbacks. Good storage habits spread by example—once a team sees simple steps keep operations smooth, corners stop getting cut. Benzoic acid may seem low on the threat scale, but treating it with proper respect keeps everyone and everything running safely.
What’s in Your Food Preservative?
Benzoic acid shows up on countless food labels, mostly in preserved foods like jams, sodas, and pickles. Flipping a jar around in a grocery store, folks often wonder if it belongs in the same group as fresh-squeezed lemon juice or more with artificial dyes. I’ve seen more parents asking this question while scanning ingredient lists, trying to figure out what exactly they’re serving for dinner.
The Roots of Benzoic Acid
Benzoic acid has deep roots in nature. Certain berries, like cranberries and lingonberries, contain it, and so do cloves. It sits in the background of many plants, usually working as a natural defense. Eating raw cranberries brings you a dash of this compound, whether or not the label mentions it.
Most benzoic acid on store shelves doesn’t come pressed out of berries, though. In manufacturing, companies make it by oxidizing toluene, a chemical found in crude oil. This method produces large quantities at a fraction of the cost of harvesting plants. When companies want to add it to soda or packed cookies, synthetic benzoic acid offers consistency and easier quality checks. That doesn't automatically make it evil, but it leaves the term “natural” slipping away from the picture.
Why Should Consumers Care?
It’s not just a matter of curiosity. Drawing lines between natural and synthetic has a real impact on people concerned about clean eating. From my experience, some shoppers try to weed out anything made in a lab, sticking to ingredients that actually grow somewhere. Others trust the historical use of a chemical like benzoic acid more than they trust the latest “all-natural” label, figuring the science keeps food safer for longer.
Public health agencies, including the US Food and Drug Administration, give benzoic acid the green light as a food preservative, within set limits. Experts know exactly how much is safe over a lifetime. On the flip side, research has linked very high amounts (above what’s legal in foods) to some mild negative effects, but these risks remain unlikely at regulated levels. Some small studies have flagged issues for people sensitive to benzoates—especially those with asthma or certain allergies.
Finding the Middle Ground
For people earning a living in food manufacturing, synthetic benzoic acid solves a problem—they can keep large batches of food from spoiling, lower the odds of dangerous bacteria, and avoid cost spikes that would hit shoppers hardest. On the other hand, for those choosing food for their own table, transparency matters. Clearer labeling would help folks understand if that benzoic acid started life in a berry patch or a chemical plant.
One win would be stricter guidelines for the use of words like “natural” on packaging. If a preservative comes from a synthetic source, the label should say so, leaving fewer surprises for careful consumers. Food companies could also look into more sustainable ways of extracting natural benzoic acid, making it easier for people to choose with their values in mind.
Eating preservative-free isn’t always realistic. Not everyone can pick berries from the backyard or make jam from scratch. Still, more open conversation about where ingredients come from helps families weigh the benefits and tradeoffs. Whether benzoic acid sits on the natural or synthetic side of the fence, consumers should have the facts before making up their own minds.
What Lasts, and Why That Matters
Pulling an old container of benzoic acid from the back of a lab shelf always sparks a little hesitation. Unlike milk, it has no sour smell to clue people in when age starts to catch up with it. Manufacturers usually stamp a two- to three-year expiration date on the labels, and many chemists treat that as gospel. Yet benzoic acid is a hardy compound. The stuff’s a staple in food preservation and has been used as an antimicrobial agent since the 19th century. It fights spoilage in acidic foods, and that same property helps it resist spoilage in its pure chemical powder form.
The Simple Truth About Decomposition
Chemically speaking, benzoic acid doesn’t fall apart easily. Its structure isn’t vulnerable to the slow breakdown you see with lots of organic compounds. Lab tests have shown that, as long as moisture and direct sunlight stay out of the picture, benzoic acid can keep its chemical punch for years past what’s printed on the bottle.
Humidity poses the real risk. Clumpy powder after exposure to moisture tells its own story: benzoic acid starts to cake and loses its ease of use during weighing and measuring. Even then, as long as it hasn’t dissolved and formed new compounds, its preservative action stays pretty stable. From personal experience, a sealed jar stored in a cool, dry place in my school chemistry storeroom still worked fine five years later. The crystals looked unchanged, and infrared spectra confirmed the same functional groups as the day it shipped in.
Aging, Safety, and Best Practice
The issue with using an old batch centers on reliability instead of safety. The FDA and European Food Safety Authority have cleared benzoic acid for food use, but both agencies stress the role of shelf life mainly to guarantee predictable results. Degraded benzoic acid seldom becomes dangerous in itself. Its biggest risk lies in slipping a degraded preservative into a recipe or formulation—and winding up with spoiled food later on, or a failed lab reaction. The loss is in effectiveness, not in the development of toxic byproducts.
Lab workers and food processors can sidestep most problems with some simple habits. Store benzoic acid in airtight containers, away from sunlight and heat, and always steer clear of working over an open bag. If the powder clumps, tests with a melting point device or a quick TLC (thin-layer chromatography) run reveal the purity. In most cases, pure benzoic acid sticks to its textbook melting range even after years in storage.
Solutions and Mindset Shifts
People often treat chemical expiration dates the same as those slapped on a bottle of milk: a hard stop instead of a suggestion. Reframing these dates as a prompt to check quality—rather than dump a perfectly usable chemical—can save money and reduce waste. Solid lab habits trump blind loyalty to dates, especially with a rock-steady compound like benzoic acid.
As more labs look for ways to reduce environmental and financial waste, it makes sense to test, not toss. Infrared spectroscopy, melting point checks, or even simple qualitative food-grade effectiveness tests give benzoic acid a fair assessment. If it passes, it still has a job to do.