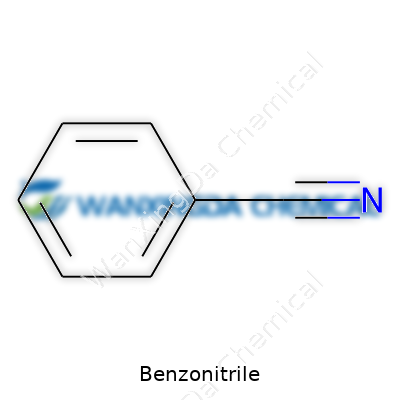
Benzonitrile: A Closer Look at an Unsung Chemical Player
Historical Development
Benzonitrile didn’t arrive overnight. Chemists tracing it back usually start with the story of the 19th century, a time flush with discovery and relentless curiosity about the benzene ring. Friedrich Woehler and Justus Liebig, those legendary pioneers, nudged benzonitrile into textbooks for the first time. Back then, isolating a compound like this from bitter almond oil felt revolutionary. Benzonitrile showed that aromatic chemistry could reach farther than anyone thought. For a little while, its practical uses lagged behind its academic intrigue, but modern organic synthesis caught up fast. This molecule stopped being a bench curiosity and started finding real meaning in specialty areas, especially in the laboratories developing dyes and pharmaceuticals between the old world wars.
Product Overview
Benzonitrile wears a lot of hats in the chemical world. At first glance, it's a colorless liquid with a faint, almond-like smell. But beyond its scent, it’s tough and reliable—a real solvent for many organic reactions. Its role in the laboratory feels similar to that of an old, sturdy tool: not glamorous, but completely essential for chemists chasing new molecules or refining old ones. A lot gets done with benzonitrile, from serving as a raw material in agrochemical synthesis and making active ingredients for medicine, to enabling the production of high-performance polymers. While not flashy, its reliability keeps it stocked on the shelves of every serious lab I’ve ever set foot in.
Physical & Chemical Properties
Taking a closer look, benzonitrile doesn’t hide what it’s made of. It comes with a boiling point just under 200 degrees Celsius and holds up well under pressure in most glassware. Its chemical formula, C7H5N, says “simplicity” but inside, the triple bond of the nitrile group gives it a stubborn, almost unbreakable personality. It mixes well with plenty of organic solvents, and holds enough polarity that reactions in it happen cleaner than in a lot of other options. It’s not explosive, not especially flammable, and if you spill it, it won’t disappear in a puff. Instead, it lingers—a reminder to respect even the most well-behaved chemicals.
Technical Specifications & Labeling
In the lab, technical specifications aren’t read for entertainment. You check purity, water content, and allowable impurity levels because mistakes cost time as much as money. High-purity benzonitrile often means better yields and fewer unknowns popping up on a spectra. Labeling tries to keep up, translating regulatory speak for the working chemist: hazard symbols warn about toxicity, batch numbers track what went wrong if something fails, and expiration dates keep old stock out of critical reactions. The people setting those standards have earned their spot from hard lessons and routine audits. It doesn’t make the process exciting, but it does keep it honest.
Preparation Method
Making benzonitrile on an industrial scale uses more finesse than most folks imagine. Classical chemists would start with benzyl chloride, treating it with ammonia and heat. These days, approaches can look more modern, leveraging copper-catalyzed reactions that trim side-products and waste. Skilled workers keep things tightly controlled—temperature, pressure, reactant feed—because mistakes spill over quickly. Each run demands careful handling of ammonia and control over byproducts. Watching a well-run process feels a bit like watching a skilled craftsman who knows exactly when to step in and when to let the setup run itself. The product rolls out looking ready for bottle or barrel.
Chemical Reactions & Modifications
Benzonitrile’s chemistry opens pathways for makers and tinkerers. That triple bond in the nitrile group lets it slide into all sorts of reactions, from Grignard additions to catalytic hydrogenations and reductions to amines or even aldehydes if you zig when you should zag. Every organic chemist remembers at least one trick involving benzonitrile—if not for its reactivity, then for what it teaches about carbon-nitrogen chemistry. It stands out for its straightforward participation in synthetic sequences. Modifying it builds routes to more complex molecules, laying the foundation for countless research breakthroughs and even a few commercial hits.
Synonyms & Product Names
Chemists can get creative with names—benzonitrile wears several faces: phenyl cyanide, cyanobenzene, and in historical papers, sometimes just “nitrile of benzene.” Popular labels try to keep it simple, but regional preferences and legacy code numbers still sneak into documentation and bottle labels. For any professional, learning these synonyms becomes a matter of avoiding confusion, especially with global collaboration and constant regulatory updates. In practice, the core stays the same, even if the name at the top of the bottle shifts a little.
Safety & Operational Standards
I’ve seen more than a few folks underestimate what benzonitrile demands for safety. Exposure can lead to headaches, confusion, or worse with enough inhalation or skin contact. The old advice from supervisors rings true: work in a fume hood, wear gloves, watch where you splash. Newer rules go further, with training certifications and digital recordkeeping. Lab safety culture now expects eye protection by default and backup spill kits at arm’s reach. Waste disposal rules treat it seriously, recognizing its persistence in water and the need for responsible stewardship. Good practice in the lab means fewer accidents, less downtime, and maybe even a better night’s sleep.
Application Area
Benzonitrile doesn’t hog the spotlight, but nearly every major chemical industry leans on it. In pharmaceuticals, it’s an intermediate for antihistamines and blood pressure drugs. In agrochemicals, crops grow a little stronger thanks to synthesis steps rooted in this humble liquid. Textile and dye manufacturers take advantage of its solvency, and the polymer world needs it for resins that won’t give up under stress. Materials scientists look for purity here, knowing that trace impurities can change the color or strength of their end product. The tiny improvements benzonitrile enables up and down the supply chain add up, supporting advances that consumers rarely notice but always benefit from.
Research & Development
Benzonitrile keeps rewarding researchers ready to push boundaries. In the last decade, work on catalytic C-H activation and green chemistry has dragged it back into journal headlines. Teams work on better catalysts that save energy, minimize hazardous waste, and extract more value from each batch. The molecule’s straightforward structure makes it an attractive candidate for molecular modeling and new reaction pathways—great for students and seasoned researchers looking for tight control and reproducibility. The story continues to evolve, as industry and academia collaborate more than ever, pushing for answers to complex synthesis questions, be it in drug discovery or advanced materials.
Toxicity Research
Plenty of debate surrounds chemical safety, and benzonitrile brings real issues. Studies show that swallowing or inhaling high concentrations poses problems, targeting the central nervous system and presenting risks similar to other aromatic nitriles. Long-term exposure in workers draws scrutiny. Regulatory bodies keep testing for chronic effects, and toxicology teams update risk assessments with every new animal or cellular study. Technical work continues on improved protective measures and better detection in workplace air. It’s a reminder for anyone—from student to veteran researcher—to pay attention to emerging data before brushing off the old hazards as routine.
Future Prospects
Benzonitrile’s future rides with innovation in sustainability and precision synthesis. Biotech groups are searching for routes that use enzymatic or biobased inputs, hoping to shrink the environmental footprint. Industry eyes more efficient production routes, lower energy costs, and smarter processes for recovery and recycling. Digital monitoring now tracks real-time exposure, making safety practices proactive instead of reactive. For all its plainness, benzonitrile finds relevance each time a new generation asks how to make chemistry cleaner, safer, and smarter. Its versatility makes it a classroom staple, an industry workhorse, and a quiet driver of progress behind the scenes.
Beneath the Surface of Everyday Chemistry
Benzonitrile often flies under the radar in the conversation about industrial chemicals. Most folks outside the lab never give it a thought, but this simple clear liquid carries a lot of weight in modern manufacturing and research. People like me, who have spent years navigating the complicated territory between chemistry and its real-world effects, notice the remarkable ways certain chemicals shape lives far from the lab bench.
How Benzonitrile Fuels Innovation
Benzonitrile starts with a benzene ring and a nitrile group. On paper, it looks about as exciting as a line of algebra, but don’t let the simplicity fool you. Its main draw comes from being a steppingstone in organic synthesis. Many pharmaceuticals and agrochemicals require chemical reactions where only a handful of starting molecules get the job done right, and benzonitrile is one of them. Drug chemists lean on it to build more elaborate structures, creating antibiotics and other medicines that improve daily living and survival.
It doesn’t stop at medicine. I learned early in my career that dyes, resins, and specialty plastics run through processes where benzonitrile acts as a solvent or intermediate. Its chemical stability and polar nature help dissolve tough reactants or create conditions for making pigments and plastics with just the right color or durability. Anyone who enjoys vivid prints on shirts or sharp images in specialty coatings owes a small tip of the hat to benzonitrile’s behind-the-scenes role.
Risks and Real-World Challenges
Of course, nothing in chemistry comes without downsides. Benzonitrile releases nasty fumes and can cause harm if not handled with care. In factory settings, proper ventilation and protective gear matter a lot. I’ve seen teams scramble after small spills, making it clear there is no room for casualness around this compound. Air monitors, chemical handling training, and regular inspections help but only if companies take them seriously.
Environmental concerns also pop up. Benzonitrile doesn’t break down quickly if it slips into waterways, so factories must keep a close watch on their runoff and emission systems. Strong regulations work as a guardrail, but industry often works a step ahead of the rules, searching for cleaner practices before the law steps in. Switching to closed systems for both storage and transfer keeps both workers and water sources safer than open-air setups from decades past.
Building Trust and Smarter Solutions
As information spreads quicker than ever, the chemistry sector faces scrutiny over transparency and public health. Managing chemicals like benzonitrile calls for clear records, open reporting, and community outreach. When companies talk plainly about what goes into their products and how they protect people and the planet, trust comes more naturally. Even small improvements—better leak detection, training sessions for staff, safer waste methods—add up to a safer industry.
No single chemical solves the world’s problems, but the way we use and manage substances like benzonitrile speaks volumes about progress. The lesson from years of hands-on work: practical chemistry isn’t just about formulas, but about real accountability and a steady drive to do better, one reaction at a time.
Rolling Up Sleeves for Chemical Safety
Benzonitrile looks like a clear, innocent liquid. Step near it without thinking, and that’s a rookie mistake no one forgets twice. I ended up with headaches and a lingering bitter smell in my memory long before I dug into the dos and don’ts. Plenty of chemists have their own stories, because it takes just a little exposure to prove that proper handling isn’t extra credit—it’s the foundation for working with aromatic nitriles like this.
Why Everyone Ought to Respect Benzonitrile
This compound serves up a double whammy: its vapor is irritating, and any skin splash sticks with you in more ways than one. Once, a misplaced pipette spritzed my glove. Peeling that glove off fast saved me a trip to occupational health. Benzonitrile’s molecular weight and vapor pressure mean it doesn’t just stay politely in the beaker. Leave a flask open, and you’ll end up breathing it. Even short-term exposure can give you headaches, nausea, or dizziness. Chronic exposure, as research shows, can take a toll on your liver and nervous system. There’s no tough-guy routine that gets around chemistry like that.
The Simple Tools That Save the Day
Gloves and goggles sound basic, but they’re the best low-budget insurance for your health. I’ve always grabbed disposable nitrile gloves and never looked back—latex doesn’t block everything, and bare hands are asking for it. Even if it seems unlikely, put on a lab coat. Benzonitrile absorbs right through your skin, and long sleeves keep accidents from turning into emergencies.
Goggles shield more than you think. Even a tiny splash burns, and rubbing your eyes doesn’t help. Some folks favor safety glasses, but they don’t give the same side protection. Once, I watched a friend dash salt solution in her eye while prepping a standard. That stung—Benzonitrile would be worse.
Vent, Don’t Just Crack a Window
A lab with nothing but stale air feels wrong for a reason. Proper fume hoods change the game here. The best labs keep air moving away from your face and into the filter where it belongs. If your workspace offers nothing but a ceiling vent, grab a portable extractor. I’ve worked in old buildings and new, and makeshift fans never handled the vapors the way a decent hood does.
Training Makes the Difference
Plenty of people ignore chemical safety until a spill happens. Training isn’t about ticking boxes. Everyone needs to know what Benzonitrile does, how to react if they slip, and where the nearest eyewash and shower sit. I learned how to rinse and run during a fire drill years ago; no amount of online modules stick like a real walk-through.
Storage and Waste Go Hand-in-Hand
Tight lids and cool, dark storage keep Benzonitrile exactly where it should stay. Leaving bottles near heat or sunlight bumps up the pressure inside, creating risks nobody wants. I found out early that tossing waste down the drain set off alarms with our local water authority. Lab partners who set up strong waste protocols keep everyone out of trouble—it took me a while to appreciate, but shared safety beats maverick shortcuts every time.
Strength in Small Habits
Working with Benzonitrile sharpened my respect for routine—daily glove checks, double-labels on bottles, never skipping the vent. These are habits born from seasoned scientists who taught me that safety isn’t an obstacle; it’s how good work gets done. A few careful steps save lives—literally and figuratively—and leave you with clear lungs, clear eyes, and a clear conscience at the end of the day.
Science in Everyday Life: The Story Behind C7H5N
Walking through a high school chemistry class, you might spot the question scribbled on the whiteboard: “What is the molecular formula of Benzonitrile?” On the surface, the answer seems simple—C7H5N—but behind these seven carbons, five hydrogens, and one nitrogen, there’s a web of knowledge stretching from textbook pages to global innovation.
I remember my first real exposure to organic chemistry. The formulas always looked like secret codes. Nothing about those combinations felt connected to life outside a lab. Take Benzonitrile, for example. Most folks haven’t measured it out or watched it bubble in a beaker. Yet, understanding its composition changes how we see the stuff of the world, giving us the tools to judge and value scientific information, rather than just accept it.
Why a Simple Formula Matters
Benzonitrile’s molecular formula isn’t just trivia. It points to the arrangement of atoms, hinting at reactivity, risk, and utility. For researchers in pharmaceuticals and materials science, that combination means Benzonitrile acts as a useful solvent or building block. The presence of that nitrogen atom attached to the benzene ring gives it versatility in synthesizing everything from dyes to medicines.
Digging deeper, the formula tells a story about safety. Working with Benzonitrile brings health risks like toxicity and flammability. If you don’t know what makes up a compound, you can’t judge where it belongs: under a fume hood, inside a secure bottle, or out of your reach altogether. Safe labs and responsible industries begin with this level of understanding.
Education, Trust, and Making Better Choices
Trust in science grows when people feel equipped to decode jargon. When teachers break down formulas like C7H5N in real-world terms, students bridge science and society. They start asking more interesting questions: What does Benzonitrile do? Where do we see it outside classrooms? Each piece of knowledge builds toward better decisions—both in policy and in personal safety.
Science reporting needs the same transparency. Random strings of elements and numbers don’t mean much on their own. Reporters and educators can lift the curtain by connecting facts to applications and impacts. That kind of clarity respects readers’ curiosity and builds resilience against misinformation or marketing half-truths.
Better Science for a Safer Future
Society benefits when more people can read and use chemical formulas. It makes everyone more prepared, whether they work in a lab or not. Even policies on environmental safety or public health depend on leaders knowing how to ask the right questions about what’s in our products or medicines. Groups like the EPA or CDC set guidelines based on this baseline knowledge—cutting through hype with a foundation of facts.
Next time you see C7H5N, remember that each letter and number carries weight. It signals challenges, opportunities, and the need for sharp thinking. Chemistry isn’t just a subject left behind after graduation—it’s a set of skills for understanding the world and shaping it wisely.
Looking Past the Chemistry Set
Benzonitrile doesn’t get the attention of giant polluters or household toxins, but that doesn’t mean it’s harmless. It shows up in the world of chemical synthesis and pops up as a solvent for research and industry. As someone with hands-on experience on both sides of the academic and commercial fence, I’ve seen workers handling benzonitrile with the same casual attitude reserved for nail polish remover. That attitude can invite trouble.
Health Hazards Behind the Lab Bench
Spill some benzonitrile and a sharp, bitter almond smell hits you. The reason? Your nose is picking up something your body doesn’t want. Breathing in those vapors, even at low levels, irritates the respiratory tract. Direct contact burns skin and eyes—fast. I’ve seen a student try to wipe a splash off their hand and wince as redness and pain flared up. There’s more lurking beneath the surface. Swallowing or inhaling a decent amount can lead to dizziness, nausea, and headaches. Animal studies, not always good news for us, suggest that higher exposures could damage internal organs and even spark nerve problems.
The root cause: benzonitrile’s chemical relatives, including cyanide, are notorious for interfering with how the body uses oxygen. No one wants to discover the limits of their own metabolism on a busy Tuesday. Regulations from the likes of OSHA and the EU didn’t come out of nowhere. They drew lines at workplace exposures because the stuff can do real damage if safety rules become an afterthought.
Environmental Invisibility Doesn’t Mean Inaction
Benzonitrile slides under the radar compared to some chemicals with higher environmental profiles. That trick doesn’t mean the coast is clear. If benzonitrile spills into the soil, water, or air, it does break down eventually—but only at its own pace, with enough persistence to hang around longer than people expect. Small aquatic life feels the impact first. Fish and invertebrates, the equivalent of canaries for our waterways, are sensitive to benzonitrile in concentrations not so different from what might leak out of a warehouse or lab.
I’ve watched researchers pour waste solvents into collection drums at the end of the day. One forgotten drum, a leaking seal, and you have a small ecosystem at risk. Missing that story is easy because the effects aren’t always dramatic. No fish floating belly-up, just a slow, invisible squeeze on life.
Avoiding Trouble at the Source
Focus lands on smarter storage, better training, and upgrades in waste handling. Twice a year safety refreshers and regular lab walk-throughs help keep mistakes rare. Companies that put real money into containment and worker education see fewer accidents, saving both dollars and headaches. Shift the culture from "get the job done" to "get the job done safely," and the difference turns up in the logs and in the air.
There’s space here for greener chemistry. Some research groups aim to replace benzonitrile altogether. Less toxic alternatives cut the risk without killing innovation. Still, wherever it’s used, respect and precision matter more. I've learned that “it’s just a solvent” never stays true for long after someone forgets the hazards are real—and often avoidable.
Modern Labs, Old Hazards
Handling chemicals changes the way you look at a lab shelf forever. Benzonitrile is one of those bottles that calls for a bit of respect—a clear, slightly almond-scented compound, much loved and much feared in countless organic syntheses. My memories of grad school are filled with lessons learned the hard way: one splash, one misplaced lid, and suddenly a regular Thursday morphs into a story for future undergrads about what not to do. That sharp, bitter almond note burns into your senses as a reminder—some risks aren’t always visible.
Benzonitrile Isn't Just Another Liquid
Most people won’t ever have a reason to think about benzonitrile unless they’re in a chemistry field. It doesn’t get the same headlines as its more notorious chemical cousins, but its dangers land people in trouble just as quickly. Besides being toxic to breathe and irritating to the skin and eyes, benzonitrile reacts with strong oxidizers—a mistake triggers dangerous byproducts. Reports from environmental protection agencies confirm that accidental releases of aromatic nitriles like benzonitrile have caused contamination at several waste sites, leading to years-long cleanups.
Straightforward Storage Makes All the Difference
A real lesson I picked up from the lab supervisor: don’t stash a risky chemical near the acids, bases, or oxidizers just for convenience. Always keep benzonitrile in a tight, clearly labeled bottle—usually glass, with a solid seal. Flammable storage cabinets with internal temperature controls and clear segregation rules are not luxury extras; they’re essential for safety. Too many fires in research centers have shown that chemicals stored together without thought can lead to expensive, life-threatening disasters.
The Human Risk
Long-term exposure creeps up unnoticed. Chronic contact with small spills, mishaps while transferring, or even workplace air that hasn’t been tested properly—stories like these don’t fill textbooks, but colleagues in the industry share them in whispers and cautionary tales. Real-world cases link prolonged exposure to neurological symptoms and liver issues, and regulatory bodies like OSHA emphasize the need for well-maintained fume hoods and personal protective gear. Handling the bottle with bare hands or working without decent airflow shouldn’t happen, but shortcuts find their way into busy labs.
Safe Disposal: Beyond Pouring Down the Drain
After work, the temptation sometimes hits to just pour leftover liquid into the nearest sink and call it a day. That’s not just lazy; it's illegal and environmentally reckless. Benzonitrile in city water supplies or local wildlands creates headaches for both cleanup crews and communities. Proper chemical waste containers, equipped with absorbents and clearly separated by hazard class, fit every responsible lab’s daily routine. Professional chemical disposal services process these containers using methods that break down toxic components safely. In my own experience, directors who invest in staff training and outside audits see lower incident rates and happier health inspectors.
Better Habits, Fewer Stories
People in chemistry circles swap war stories about spills, close calls, and lessons that stick for life. Treating benzonitrile with caution isn’t just about following rules—it’s about keeping yourself and your community safe, reducing risk, and making sure accident stories remain lessons, not headlines. Investment in training and proper protocols protects people, the environment, and the reputation of scientific work everywhere.