Bistrifluoromethanesulfonimide: Chemistry’s Unsung Backbone
Tracing the Roots of Bistrifluoromethanesulfonimide
Watching chemistry unfold in the lab, I’ve seen how certain compounds quietly carry much of the scientific heavy lifting without drawing the fanfare. Bistrifluoromethanesulfonimide, or TFSI as many chemists call it, fits into this mold. The journey of this substance started in the mid-20th century when researchers searched for stronger acids and more stable anions to fuel breakthroughs in organic synthesis and emerging battery chemistries. TFSI emerged from the ambition to create anions that could balance charge in the most demanding settings, far outperforming weaker candidates like perchlorates or nitrates. Decades of research and rising demand for potent ionic liquids carved a place for this molecule in both old-school labs and renewable energy workshops.
Bistrifluoromethanesulfonimide at a Glance
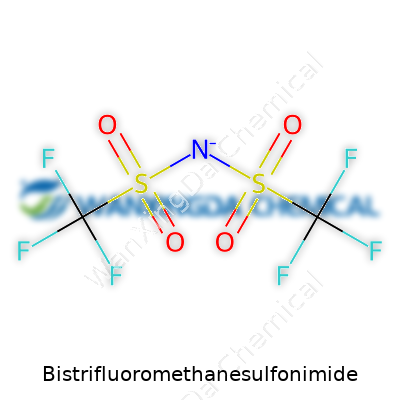
This compound stands out with striking resilience and flexibility in the hands of a chemist. TFSI’s structure sports two strongly electron-withdrawing trifluoromethanesulfonyl groups bonded to a nitrogen, producing an anion with startling stability and an uncanny ability to dissolve in many solvents. Each property feeds into its growing reputation. You don’t see it making headlines, yet it brings out the best in high-end lithium batteries, advanced catalysts, and nonaqueous electrolytes because it doesn’t play games with water or oxygen sensitivity. Businesses in high-tech and energy fields keep coming back to TFSI-driven solutions because the stuff simply works where others fold.
The Nitty-Gritty: Physical and Chemical Traits
From the first handful of TFSI salt I handled, the strong, almost biting smell stood out just as much as the powdery-white crystal look. High melting point, low volatility, and jaw-dropping thermal stability let you heat it up without fretting over nasty decomposition. TFSI anions barely flinch in the presence of strong acids or bases, and since they’re non-coordinating, they don’t interfere with sensitive metal catalysts. These simple facts translated into less downtime cleaning up unexpected side products. And once you see how solubility remains consistent across polar and nonpolar solvents, the practical side becomes clear—TFSI fills the gaps that others leave wide open, allowing tailored syntheses, separation chemistries, and electrochemical systems to thrive.
Behind the Label: Technical Details and Identification
The chemical’s widespread use doesn’t leave much room for ambiguity. You’ll hear chemists call it TFSI or bistriflimide more often than its full name—bis(trifluoromethanesulfonyl)imide—and for good reason. The name’s a mouthful, but the meaning is essential. Chemical identities are more than academic; getting the right label prevents costly errors in industrial settings. TFSI shows up in research publications, patent filings, and supply chain paperwork under dozens of similar monikers, and keeping track of them minimizes confusion. Knowing exactly which variant researchers reference helps keep teams on the same page and processes running smoothly.
How Chemists Get It Done: Preparing Bistrifluoromethanesulfonimide
You don’t need to be a specialist to appreciate that making TFSI requires careful, deliberate handling of powerful reagents. Chemists often start with trifluoromethanesulfonyl chloride, which reacts with ammonia to punch out the central N-H bond. While the process sounds simple enough, it usually involves precise temperature control, inert gas atmospheres, and stepwise purification to strip away remnants of harsh starting materials. I remember spending hours purifying the product to guarantee it hadn’t picked up traces of water or acid, which could ruin downstream testing. No operator who’s committed to performance shortcuts the process, since purity determines whether batteries meet their lifetime targets or research catalysts deliver repeatable results.
Tinkering and Tweaking: Chemical Reactions and Modifications
This molecule isn’t just a finished product; chemists constantly modify it or toss it into reactions where its unique characteristics shine. You can swap out the hydrogen on its nitrogen for various metals or organic groups, forming salts and even ionic liquids with broad application. The stability of the TFSI anion makes it a gold standard in situations that burn up lesser molecules. In the lab, it withstands strong oxidizers and reducers, keeping its cool while pushing reactions forward. Newer research looks at tuning the electron-withdrawing effects or introducing chirality to unlock tailored selectivity in asymmetric synthesis or specialized energy storage.
Staying Safe and Meeting Standards
As with any advanced chemical, operational discipline pays big dividends. TFSI brings its own challenges: inhalation hazards, risk of skin exposure irritation, and possible eye damage get flagged in every standard operating procedure. Lab veterans like me quickly learn that gloves and eyewear aren’t negotiable. Storage demands dryness and sealed containers to maintain long shelf life. At industrial scale, air handling, spill containment, employee training, and regulatory compliance move from afterthoughts to essentials. The fact is, any lapse costs time, money, and safety. Countries with strict chemical control laws keep TFSI’s manufacture and transport under tight watch, reducing environmental and health risks while enabling continued innovation.
From Batteries to Biochemistry: Application Horizons
TFSI’s legacy reaches everywhere phones, electric vehicles, and grid batteries ask more from their electrolytes. Lithium salts derived from TFSI let modern batteries crank out more cycles, tolerate higher voltages, and resist overheating—shared pain points in today’s energy conversation. TFSI-based ionic liquids show up in supercapacitors, advanced fuel cells, and separation methods for metals and rare earth elements. I’ve seen it pop up in enzyme research as well, where its stability permits testing under conditions that destroy less robust molecules. In pharmaceuticals and catalysis, TFSI continues to grease the wheels of green chemistry by supporting reactions that otherwise waste mountains of solvents or fail entirely.
Research and Development: Shaping Tomorrow
TFSI’s rapid march into the future comes down to its unique mix of characteristics and adaptability. Current research looks at ways to further cut costs by simplifying the synthesis and reducing the environmental footprint—big goals with the green tech boom in full swing. Team efforts push toward replacing hazardous reagents during production and speeding up cleanup. Some research groups tinker with fluorinated variants, aiming to combine TFSI’s best qualities with easier biodegradability or even greater temperature resilience. As demand for safe and high-performance batteries grows, so does investment in TFSI’s next-gen relatives, aiming to exceed what classical lithium-ion chemistries offer today.
What Toxicity Research Tells Us
Scientists never cast a blind eye toward potential harm. TFSI doesn’t present the acute dangers of mercury or lead, but chronic exposure stories and animal studies remind everyone to avoid complacency. Inhalation can cause respiratory irritation. Skin contact brings rashes or mild burns, especially with the pure acid form. Longer-term studies search for signs of bioaccumulation or ecological damage, especially since the sulfur-fluorine bonds don’t break down easily in nature. Manufacturers respond by beefing up containment measures, switching production steps to closed systems, and refining wastewater treatment methods. Public demand for transparency in chemical safety pushes researchers to share toxicity data, shore up safety gaps, and look for greener alternatives when possible.
Looking Ahead: TFSI and the Push for Cleaner Chemistry
The future for TFSI, and the larger family of sulfonimides, doesn’t rest on nostalgia. Ongoing progress in battery science, sustainable materials, and process chemistry keeps refining how we understand and use this compound. Green chemistry principles drive researchers to work toward safer derivatives, improved life-cycle impacts, and smarter recycling of spent batteries and catalysts that once relied on TFSI. Cutting-edge labs share know-how across borders using open data and cooperative studies, proving that chemistry’s future is collaborative—driven as much by transparency as by technical prowess. As climate policy turns sharper and resource scarcity creeps up the agenda, the real test is whether TFSI can keep pace with new demands without sacrificing the robustness and reliability that won it quiet respect in the first place.
What Drives the Need for Bistrifluoromethanesulfonimide?
Stepping into a chemistry lab or any modern battery research facility, the word “bistrifluoromethanesulfonimide” doesn’t exactly roll off the tongue, yet it keeps popping up in places people might not expect. In my time dabbling in different research circles, one recurring lesson shows up time and again: progress often hides behind complicated names. This compound, usually short-handed as TFSI, works quietly behind the scenes in several cutting-edge technological fields. The reason people keep coming back to TFSI boils down to its reliability as an acid in making new materials and as a stable part of advanced batteries.
Bistrifluoromethanesulfonimide and the Push for Better Batteries
Talking about progress in battery tech, especially lithium-ion and next-generation batteries, everyone wants more power with less danger and longer life. TFSI-based salts serve as key building blocks for ionic liquids and electrolytes, which help batteries last longer and tolerate being charged and discharged thousands of times. Those searching for grid storage solutions, electric cars, or thinner, more reliable consumer electronics tend to favor battery chemistries that offer lower risk of thermal runaway. Electrolytes built around TFSI don’t just help with improved stability—they also resist breaking down under harsh charging or high temperature cycles. All that means safer batteries with less downtime.
The Chemistry Classroom and Synthesis
Back in a university chemistry classroom, TFSI started turning up not only as theory, but also in practical lab work. Anyone who’s tried to create new molecules knows the value of a strong, stable acid. Bistrifluoromethanesulfonimide acts as a gold standard “superacid.” Chemists reach for it to make organic and inorganic syntheses friendlier and cleaner. By avoiding corrosive leftovers and offering more predictable reactions, labs spend less time fixing mistakes and more time pushing discoveries further. In organic electronics, for example, the right synthesis pathway means getting better performing materials for printed solar panels or flexible displays.
Looking at the Downsides and the Environmental Piece
Useful doesn’t always mean harmless. In manufacturing, I’ve seen companies scrutinize every ingredient, checking impact on workers, processes, and end-of-life disposal. TFSI brings up questions about how we deal with chemicals that stick around for a long time. Some research flags TFSI as a highly persistent substance, which means governments and environmental groups are starting to pay closer attention. Proper disposal, closed systems in chemical plants, and ongoing green chemistry initiatives matter here. It’s not enough to chase the best performance without thinking about what happens after the fact. Innovation always brings responsibility.
How Should We Approach the Future?
There’s little argument about the importance of bistrifluoromethanesulfonimide for science and manufacturing right now. The call for better, cleaner, more responsible technology points at two things: using TFSI where it brings major progress and supporting safer, greener alternatives as soon as they become practical. Clear labeling, giving workers protective training, and funding research into biodegradable options are all things that encourage not just safer labs, but safer communities. By learning from every discovery—good and bad—the picture gets clearer. Our best way forward comes from balancing technical gains with honest awareness of risks along the supply chain, from factory floor to finished device.
Why Storage Matters for Chemicals Like This
Bistrifluoromethanesulfonimide (often shortened to TFSI or sometimes HNTf2) might not ring familiar to folks outside chemistry circles, but among researchers and lab workers, its name triggers an almost automatic mental checklist. I’ve spent enough time in labs and chemical storerooms to know that handling compounds like TFSI safely isn’t optional. It’s essential to prevent not just property loss but injuries and compromised research, too.
Moisture and Air—The Unwelcome Guests
Every bottle of bistrifluoromethanesulfonimide comes with a story about moisture and air. This acid loves to draw in water vapor—something chemists call hygroscopic behavior. Leave the cap off too long, or ignore a crack in the bottle, and TFSI will soak up ambient moisture in no time. That can turn powdery material into lumps and, worse, cause slow decomposition. I learned this the tough way one summer, walking back into a stifling, humid storeroom only to discover a batch that clumped together like old sugar. No one likes realizing a thousand-dollar bottle just went bad thanks to ambient air.
Finding the Right Spot—Dry, Cool, and Out of the Sun
People ask, “So where do you actually put TFSI?” The answer isn’t as complicated as they expect. It belongs on a chemical shelf, tightly sealed, somewhere cool—think 2–8°C, a typical fridge temp for chemicals. Standard refrigerators for flammable and reactive materials, not ones where you keep lunch. Sunlight can also degrade some sensitive reagents over time, so direct sunlight should never touch the bottle. Exposure to heat not only messes with chemical integrity, it can speed up unwanted reactions.
Safety First—Preventing Accidents Before They Begin
Chemical safety isn’t just about individual habit; it starts from the minute a shipment arrives. TFSI comes with hazard warnings about strong acidity. Spills cause burns. I used to think gloves would always be enough. Once, during a rush to weigh out some for a synthesis, I skipped goggles. A droplet splattered and burned a hole in my sleeve. Lesson learned: goggles every time, plus gloves and a lab coat.
Chemicals like TFSI don’t belong near incompatible materials. Alkali metals and strong bases—keep them at arm’s length. I never trust a mixed shelf, no matter how pressed for storage space. Separate acids and bases. If a bottle cracks or breaks, clean-up involves neutralization and breathing protection, not just paper towels.
Label Everything—Memory Isn’t Enough
Permanent, legible labeling—it seems obvious, until the faded, smudged tape on an old bottle forces you to guess. Regulations today back this up, and for good reason. During audits or emergencies, you don’t want doubts about that jar's actual contents.
Taking Responsibility—A Culture of Togetherness
Shortcuts rarely save time in chemical storage. I’ve seen one person’s quick fix become tomorrow’s mess for someone else. Good habits—closing every lid, double-checking seals, staying on top of expiration dates—catch on when everyone cares. Communication and teamwork create safer spaces and better science. TFSI lasts much longer when we respect its quirks and keep the storeroom ready for action.
Bistrifluoromethanesulfonimide's name alone raises a red flag for anyone who spends time in a lab or cares about what ends up in the workplace air. With its growing use in lithium batteries, electrochemistry, and specialized organic reactions, curiosity about toxicity and hazards keeps turning up among chemists and safety officers alike.
How the Hazards Show Up
This chemical, often called “TFSI” for short, doesn’t get used in consumer products. Still, its footprint is spreading quietly through research labs, pilot plants, and, sometimes, big industrial settings. What worries most people isn’t whether TFSI will get on your skin, but what happens if it ends up in water or air, or if you’re not paying attention on the bench. When a compound like TFSI packs fluorine into its structure, the mind immediately goes to risks—the kind that can cause irritation, damage to lungs if fumes escape, or persistent pollution because fluorinated chemicals tend to stick around for a long time.
Years in the lab teach you to pay special attention to anything sulfonimide-based. Strong acids, even when locked up in salt form, still deserve respect. TFSI itself gets used partly because it’s a superacid in its protonated form, and because its salts dissolve and conduct so well. That also means it can break down under harsh conditions and give off byproducts that hurt the respiratory tract or skin. Gloves and goggles aren’t just for show with this stuff; accidental exposure can burn, and inhalation of dust or mist can inflame the lungs. Fluorinated dust may bring longer-term health questions because of how persistent these molecules can be.
Checking the Toxic Track Record
Looking at available research, TFSI comes with a record full of gaps. Not many long-term animal studies show what happens with ongoing low-level exposure. The best data available points toward skin and eye irritation, and acute exposure producing discomfort or harm, depending on concentration and form. Because of its structure, TFSI isn’t likely to explode or catch fire, but it doesn’t need to for people to take it seriously. Public chemical hazard databases specifically note the need for protection against splashing, inhalation, and disposal that keeps it out of water systems.
Another real concern comes from environmental persistence. Like many fluorinated chemicals, TFSI resists natural break-down. This means spills or improper disposal can linger in water or soil for years, moving into the food chain or harming aquatic life. Europe and the US haven’t rolled out strong regulations on TFSI yet, perhaps because it isn’t nearly as high-volume as some notorious perfluorinated compounds, but the science points to caution as the smarter approach.
A Way Forward in Labs and Industry
Whenever I train a new chemist, I never skip the conversation about how even niche chemicals can show their teeth years later. TFSI may sound like a technical curiosity, but history shows what happens with compounds we don’t fully understand. Smart lab practice means using fume hoods, wearing good gloves, and making sure spill kits are ready. Safety sheets do their job, but hands-on training matters more.
Industry might not face a crisis with TFSI yet. Still, waste tracking and management updates can close the door before it opens on contamination. Choosing substitutes whenever possible, sealing containers, and running containment checks make a difference to both workers and the wider world outside the plant’s fence.
The takeaway doesn’t call for alarm, but for honest respect. In science, overlooking small hazards creates big regrets. TFSI keeps proving that good habits and open information beat both ignorance and bravado every time.
The Role of Chemistry in Modern Innovation
At the heart of many breakthroughs in chemistry sits the ability to fine-tune substances for specific scientific needs. One compound that stands out for its versatility and frequent use in research labs is bistrifluoromethanesulfonimide, sometimes simply called TFSI. Scientists chasing higher-performing batteries and cutting-edge catalysts often reach for this molecule. In my years of visiting labs and talking with chemists, TFSI comes up a lot, cementing its reputation as a game-changer.
What Sets Bistrifluoromethanesulfonimide Apart?
Bistrifluoromethanesulfonimide carries the chemical formula C2F6HN2O4S2. Each TFSI molecule holds two trifluoromethanesulfonyl groups, strung together by a nitrogen atom. Weighing it out, TFSI clocks in with a molecular weight of about 280.15 g/mol. This might seem like trivia until you dig into the real-world uses this specific molecular structure unlocks.
The point to remember about TFSI is how its structure offers stability and boosts performance in tough chemical environments. Electrolytes for lithium-ion batteries, for example, benefit from TFSI-based salts due to their ability to resist heat and decomposition. Cheaper salts often fall short during cycles of charging and discharging, leaving TFSI as a favorite among researchers looking for long-term durability.
Environmental and Health Considerations
Nobody works in chemistry without thinking about impact. TFSI, with its perfluorinated chains, sparks discussion regarding persistence in the environment. Many in the industry look at fluorinated compounds and ask about their breakdown products and how long they last if spilled or disposed of improperly. There’s no avoiding the worry: if something hangs around for decades in soil and water, risks for wildlife and eventually people go up.
Regulators have started putting pressure on labs and chemical manufacturers to keep a close eye on how much of these strong, stable compounds get out in the world. Countries within the European Union, as well as states in the US, have started laying down tough laws for perfluorinated substances. Safety officers and environmental engineers in the field have stressed in interviews that tracking and collecting waste needs everyone’s attention from bench chemist to factory operator.
Moving Toward Safer Solutions
This isn’t just an academic point; real progress demands action. Some scientists are working to design alternatives that break down more easily but hold onto the features that made TFSI valuable in the first place. Solid research funding could spark real change here. Academic-industrial partnerships have a shot at developing new molecules offering stability and performance—minus the environmental sting. If government grant makers support teams looking at greener electrolytes or safer catalysts, safer next-generation materials will arrive faster.
Handling TFSI in a responsible way provides a blueprint for future chemical innovation. Careful study, regulation, and willingness to experiment with new ideas together drive both safety and progress. With attention on the details, the world can keep the best of what chemistry brings while respecting the planet and each other.
Why This Compound Demands Respect
Bistrifluoromethanesulfonimide sounds like a mouthful but in the lab, its reputation carries even more weight. Its role as a strong acid attracts chemists working with high-performance electrolytes and catalyst systems. But with power comes responsibility — this compound pushes you to ditch any shortcuts and follow practical experience, not just paperwork.
Personal Experience Trumps Complacency
When handling any corrosive or extremely reactive material, memories of singed gloves and that one near-miss with a splash remind me to never treat familiarity as a safety net. Bistrifluoromethanesulfonimide isn’t interested in giving warnings before it bites; just a broken glove or a hurried transfer invites chemical burns or toxic inhalation. A good lab coat isn’t just a formality. It keeps ruined clothing — and skin — out of the story.
The fumes from compounds like this float downhill, so proper ventilation sets the baseline. Chemical fume hoods pull hazardous vapors away, saving your lungs and any investments in long-term health. Respirators gather dust for many, but for those expecting splashes or spills, they never regret having them nearby.
Mistakes rarely announce themselves in advance. I always check containers and labels twice, because in a busy lab every minute saved by skipping a step can double as the minute lost fixing the mess. Glass breaks, lids jam, gloves tear. Having spill kits set up and everyone on the same page helps. You can’t overtrain for real emergencies.
Disposal: Deal with the Devil, Not Just the Waste
Pouring anything this reactive down the drain belongs to those stories people regret telling. Waste treatment, at both small and industrial scale, isn’t built to neutralize strong acids combined with fluorinated groups. Sodium bicarbonate and similar neutralizing agents only go so far. Reaction byproducts can stay toxic and mobile.
Holding containers used for disposal in clearly labeled, chemically resistant drums puts everyone at less risk. Temptation always lurks to tuck them away in a corner, but central, supervised waste stations make life easier both for compliance and for the person cleaning up. Sharing a workspace with someone careless serves as a lesson: one person’s shortcut is everyone’s hazard.
If the rules seem excessive, count the legal costs and lost hours that come from regulatory breaches. Waste haulers expect you to separate, seal, and document everything. Labs owe clear records and labels, both for internal tracking and because external handlers need to know what they are dealing with. Skipping steps never pays off in this game.
Doing Better — Solutions Rooted in Practice
Simple habits, like changing gloves after handling and logging every use, protect people and equipment. Regularly reviewing safety data — not just the pages written by manufacturers but also local protocols and incident reports — builds a culture where silent risks rarely stay hidden. Working clean, keeping phone distractions out, and running mock spill drills help everyone stay sharp.
Modern labs have more options now: closed-transfer systems cut down on open handling, and chemical-resistant plastics outlast the old glass bottles that always seemed to chip. Encouraging a culture where anyone can speak up about unsafe habits brings problems out before accidents strike.
Treating Bistrifluoromethanesulfonimide as just another lab supply misses the mark. Every time someone thinks they’re too busy for full protection or proper waste management is another chance for both people and the environment to lose. Safety wins only when everyone takes on the job personally, not just as another item on a checklist.