Calcium Trifluoromethanesulphonate: Driving Change in Chemical Sciences
Charting the Early History
Diving into the history of calcium trifluoromethanesulphonate always reminds me that chemistry is a story about constant retooling and discovery, not just by institutions, but by the collective curiosity of researchers around the world. This compound, once a lab curiosity, came into focus in the late 20th century. Back then, research had a strong tilt toward exploring new ionic solutions—materials able to transfer charge effectively. Chemists started to take a harder look at salts containing the triflate group, especially after finding they allowed for high ionic conductivity and chemical stability. Early academic work unlocked the path to this calcium salt mostly through efforts to push the limits of electrolytes for batteries and new catalysts. Over several decades, the role of calcium trifluoromethanesulphonate evolved from laboratory sample to a highly relevant agent in synthesis and energy storage. Glancing back on these years, an obvious pattern emerges: innovation isn’t always about grand breakthroughs, but about gradual improvements and the willingness to ask, “What if we used something nobody's tried yet?”
Inside the Compound
Mentioning calcium trifluoromethanesulphonate to colleagues usually draws quick recognition. Chemists link its structure with reliability—its robust nature opens possibilities across research and design. The molecular picture shows a calcium ion paired with two triflate anions, creating a salt that stands tough even when the surrounding conditions become extreme. It arrives as a white powder, easy to spot in any lab inventory. The magic of this salt lies in its ability to dissolve in organic solvents and water, which gives it flexibility for both laboratory research and industrial use. Unlike many other calcium salts, this compound resists breakdown and doesn’t react with moisture, making shelf storage straightforward. Its low toxicity (when basic protocols are followed) runs counter to those older calcium reagents that sparked more caution. The combination of solubility, thermal stability, and mild reactivity means it will often come off the shelf during experimental setups or scale-ups.
How Chemists Identify Calcium Trifluoromethanesulphonate
Products bearing the name calcium triflate, calcium trifluoromethanesulphonate, or even simply “calcium triflate salt” don’t vary in their core identity. What sets one apart from another comes down to factors like the grade—lab, technical, or ultra-pure—and the way it’s labeled. Most suppliers print the molecular formula Ca(CF3SO3)2 right on the bottle and provide batch-specific analysis. For research-grade salts, checking for metal content, water content, and presence of residual organics helps avoid headaches during later use. These aren’t just bureaucratic hoops; any impurity could throw off results in sensitive experiments. Scientists who’ve seen a project derailed by tainted reagents know you can’t shortcut quality control. On a practical front, clear labeling and robust documentation help researchers replicate results, enabling real progress in both academia and industry.
Making Calcium Trifluoromethanesulphonate
Synthesizing this salt relies on straightforward, repeatable procedures. Researchers favor reacting calcium carbonate or hydroxide with trifluoromethanesulfonic acid under controlled temperature conditions. The transformation yields calcium triflate, water (or carbon dioxide in the case of carbonate), and usually delivers a high yield with simple filtration and drying. From an industrial lens, this method scales without unpredictable side reactions, keeping costs reasonable. Rigorous control over acidity and stoichiometry ensures minimal leftovers, which lowers purification demands. Labs committed to green chemistry recommend minimizing waste and recycling solvents whenever possible, a mindset that’s become part of responsible practice across the chemical industry. I’ve watched more senior chemists stress over purity during salt preparation, especially when the next steps involve reactions sensitive to metals or trace water. Their attention to detail can make all the difference between a breakthrough and wasted time.
What Calcium Trifluoromethanesulphonate Can Do
If someone gave me one minute to explain the importance of this compound, I’d point straight to its work as a non-coordinating salt. Its main draw comes from reliability in polar environments, especially in lithium-ion batteries and advanced organic synthesis. Those in materials science rely on it as a stable calcium ion source, delivering high ionic mobility in both polymer and liquid electrolytes. Electrochemical studies often turn to it because it moves calcium ions efficiently and doesn’t interfere with electrochemical reactions, unlike other anions that tend to coordinate the metal center and slow down the entire process. In organic synthesis, the presence of triflate anions boosts reaction rates by stabilizing charged intermediates. Chemists can exploit these features during catalytic cycles, promoting specific rearrangements, or even when exploring new C-H activation techniques. The pharmaceutical sector scouts it for mild Lewis acid catalysis, taking inspiration from examples published in peer-reviewed literature detailing facile esterifications or unique rearrangement strategies. By linking these real-world applications back to benchwork, you see how theory meets reality.
Reactivity and Pathways for Innovation
This salt doesn’t just sit comfortably in a bottle. It actively joins in chemical transformations, bringing to the table unique pathways for catalytic and synthetic upgrades. In the lab, researchers have observed its ability to exchange its calcium ion with others in metathesis reactions, creating new salts or promoting ion exchange for purification steps. On the modification front, the triflate group itself resists nucleophilic attack, anchoring the molecule in host environments and making it an appealing candidate as a support for immobilized catalysts. Combining calcium triflate with polymer electrolytes produces hybrid materials with high conductivity, supporting the next wave of battery prototypes. Chemical engineers often see value in tuning the surrounding environment—adjusting solvents, temperature, or counter-ions—to leverage the unique balance of Lewis acidity and non-coordinating behavior that this compound displays. This adaptable nature explains why it continues to feature in patents and journal articles pushing the boundaries of green catalysis and sustainable technology.
Staying Safe with Operational Standards
Any researcher who’s spent long hours handling chemicals will insist that safety starts with real habits, not just reading the Material Safety Data Sheet. Even though calcium trifluoromethanesulphonate carries low acute toxicity, gloves and goggles still matter in every handling scenario, mostly to avoid irritation from both the triflate and any acidic leftovers. Respecting ventilation guidelines protects from airborne dust, especially on a production scale. Labs that prioritize in-house training see fewer incidents—teaching protocols early and repeating hands-on training until instincts take over. Waste management policy counts too. Used salts and contaminated containers go to hazardous disposal, keeping workplaces free of environmental contamination and regulatory trouble. Industry standards stress documentation, so every transfer, measurement, and disposal action leaves an auditable trail. This helps trace errors and proves compliance with both workplace and environmental regulations—real-life examples show that following standards isn’t just academic, but a way to keep people safe and operations running smoothly.
Pushing Research Boundaries
What gets me excited about this compound is how researchers keep expanding its horizons. At major research conferences, talk keeps circling back to how calcium trifluoromethanesulphonate can shape future battery architectures. Already, lab studies reveal that calcium-based systems could store more theoretical charge than traditional lithium cells if the electrolyte delivers both stability and ion mobility—boxes that calcium triflate helps check. Synthetic chemists push the salt outside the comfort zone, investigating new ways to use it in C-H activation or to mediate hard-to-control rearrangements and coupling reactions. There’s steady interest in making composite materials with this salt embedded in polymers, aiming for next-gen sensors or flexible supercapacitors. These advancements spring from years of observation, failed reactions, and sometimes accidents that lead to new insights. Having access to reliable, pure salts enables these teams to compare notes across continents—one result in Europe sparking an idea in Japan, a failed batch in the US prompting a safer process in Germany.
Toxicity: Looking at Both Challenge and Promise
Working with calcium trifluoromethanesulphonate brings up necessary questions about both environmental and occupational hazards. Toxicology studies to date show that the compound doesn’t present chronic toxicity at exposure levels typical in research or manufacturing. Animal studies reveal only mild irritation in large doses, far below what would surface in normal circumstances. That said, the triflate group’s persistence raises flags about long-term accumulation in water or soil, especially if disposal isn’t tightly regulated. Because environmental chemistry catches issues decades after introduction, ongoing monitoring matters. Firms that maintain strong waste-handling programs provide the best defense against future regulation or liability. I’ve heard stories from colleagues working in the environmental monitoring field who discovered surprising chemical residues years after compounds were introduced—prompting them to campaign for stricter containment, not just for toxic hazards, but for poorly understood environmental interactions. Smart practice means treating benign-seeming compounds with the same respect as those highlighted for acute risks.
Tomorrow’s Outlook: From Specialty Markets to Everyday Tools
It becomes clear looking at both current and emerging applications that calcium trifluoromethanesulphonate’s future stretches far beyond laboratory experiments. Energy storage continues to capture everyone’s attention, with new prototypes hinting at robust, low-cost battery options running on abundant calcium where the right electrolyte can tip the scales. Green chemistry groups highlight its catalytic efficiency and environmental stability, nudging chemical manufacturing toward greener pathways. The compound also finds use in materials development for electronics, coatings, and advanced composites, where properties like high thermal stability and strong dielectric constant open up product engineering possibilities. Some visionaries predict a wave of consumer-level applications as scalable, greener battery technology takes off. Investment in recycling and material recovery programs means that future production can avoid the lifecycle risks seen in other sectors. Progress depends—like always—on making connections between bench science and real-world need, keeping eyes open to unintended outcomes and future-proofing practices to keep both people and ecosystems safe. Research labs, startups, and regulatory agencies will need to keep pace with the possibilities and pitfalls, drawing on past lessons as they shape technologies of tomorrow.
In research labs and industrial workspaces, specialty chemicals like calcium trifluoromethansulphonate get a lot less spotlight than they deserve. This compound, known by some chemists as calcium triflate, plays a crucial behind-the-scenes role, especially in areas where efficiency and purity matter more than packaging or public fame. Not many outside research or industrial circles hear much about it, yet anyone who cares about the backbone of modern technology handles the ripple effects of such silent partners. Let’s dig into what makes it a unique and sometimes irreplaceable tool.
Driving Performance in Organic Synthesis
I started noticing how calcium trifluoromethansulphonate keeps surfacing in synthetic chemistry projects. Ask a seasoned chemist about making carbon-carbon bonds more easily, and you’ll hear about the need for catalysts that can speed things up and cut down on waste. Calcium triflate serves as a Lewis acid, pulling electrons in a gentle but effective way, which leads to cleaner reactions and purer products. Nobody enjoys cleaning up a heavy-metal mess or wasting precious time debugging sloppy chemistry. Using this compound, the process often becomes less toxic and more reliable. Green chemistry matters now more than ever as labs shift away from heavy metals to pursue safer protocols.
Everyday Electronics Need Stable Electrolytes
Modern gadgets rely on energy storage, especially with portable devices and electric vehicles everywhere. Calcium trifluoromethansulphonate jumps into action as an electrolyte salt. Traditional lithium and sodium-based batteries hog the spotlight, but the hunt for better, safer batteries keeps the door wide open for calcium-based options. Here, calcium triflate offers stability and high solubility in organic solvents. Big players in battery research have noticed it. Electrolytes dictate how well a battery charges and discharges, ultimately influencing how long a phone or car holds a charge, and how safe it remains during use. If a new salt helps batteries last longer and stay cooler, that gains attention beyond the lab.
Industrial Chemistry Sees a Reliable Builder
I’ve seen manufacturers wise up to the benefits of using less aggressive chemicals in their workflows. Acylations and alkylations—those foundational steps in drug and material synthesis—lean on acids that work smoothly. Instead of turning to corrosive or environmentally risky reagents, more producers now select calcium trifluoromethansulphonate, which delivers the catalyst’s kick without bringing along the danger. A strong, water-stable acid can simplify both production and waste handling. That’s not just a cost benefit, but a win for workplace health and community safety.
Facing the Hurdles
No discussion is fair without looking squarely at practical challenges. Calcium trifluoromethansulphonate isn’t widely made at industrial scale yet. Prices stay high, and not every lab can swallow that extra expense. While it offers less toxicity compared to heavy metal salts, it also raises classic chemical handling headaches—storage, disposal, and cost of scale. Manufacturing it still calls for good controls and respect for regulations.
Looking Forward: Expanding Safe Innovation
Brighter minds will keep hunting for electrolytes and catalysts that leave less footprint but deliver more punch in everyday tools, consumables, and electronics. Investments in smarter production methods, resource sharing, and open data may help lower costs and increase adoption. Teams working outside traditional chemical industries—like battery startups or researchers in environmental tech—could help steer more attention toward these less-celebrated chemicals. By choosing smarter building blocks, the next generation of products stands to be safer, more sustainable, and just a bit more efficient.
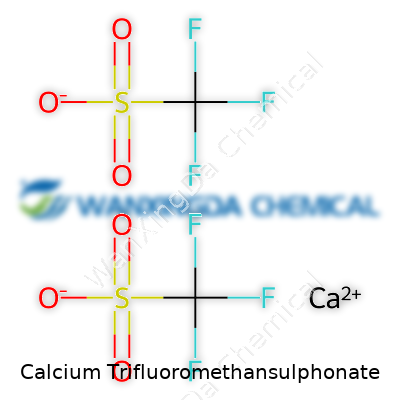
Breaking Down the Formula
Lots of chemicals come with winding, tongue-twisting names, but strip away the jargon and you can pick out the pieces. Calcium trifluoromethansulphonate has a formula that reads Ca(CF3SO3)2. On paper, it looks like a bunch of letters and numbers. In practice, those components tell a story about function and structure. Take the “trifluoromethansulphonate” group—more often called triflate by chemists. Each Ca2+ ion pairs with two of these negatively charged triflate anions, forming a salt that’s surprisingly versatile.
What Makes Triflate Salts Useful?
My time working in a synthetic chemistry lab gave me an up-close look at how some salts get all the attention. Triflates, especially in the calcium form, have a unique place because of their chemical behavior. They dissolve well in plenty of organic solvents, earning their rank as an alternative to more traditional salts in tough or “moisture-sensitive” reactions. You’ll find calcium triflate showing up in organic synthesis, batteries, and even as a catalyst ingredient.
The reason these salts matter sits in the triflate group itself—CF3SO3−. This group brings both bulkiness and the stability gained from all those fluorine atoms. It doesn’t mess with the reaction as much as some other anions do. This helps speed up or direct chemical changes in a lab setting. If you run a reaction and need your metal ion to do the work, but not the anion, a triflate can make life much easier.
Safety Still Matters
Some folks in the chemical business get tunnel vision trying to wring every drop of performance out of their salts and catalysts. Practical experience tells a different story. Handling any salt, especially those with fluorinated compounds, means knowing your risks: inhalation, ingestion, long-term exposure, even waste disposal. Triflates tend to be less hazardous than many other fluoride sources, but nobody should ignore basic safety. Use gloves, work in a well-ventilated place, and never forget the risk of accidental spillage or unwanted reactions with other chemicals.
Addressing Supply and Innovation
Reliable access to specialty salts like calcium trifluoromethansulphonate has become a growing challenge. High demand from industries using these chemicals in electronics, electrochemistry, and even environmentally friendly energy applications puts pressure on suppliers and researchers. Building up recycling pathways in chemical manufacturing could help avoid supply bottlenecks and cut costs. Development of greener synthesis methods for producing triflate salts would bring benefits across the board—less waste, more efficient reactions, and a smaller environmental footprint.
Building Future Applications
Seeing firsthand how a chemical’s formula connects to day-to-day usage still grounds my perspective—this is not just theory, it’s what shapes new technology. Calcium trifluoromethansulphonate’s unique properties let it pop up in everything from specialized catalysts to energy storage research. Getting familiar with formulas, behavior in solution, and interaction with other materials will push both science and industry forward.
Examining Lab Chemicals Without the Fluff
People who work in research, manufacturing, or teaching labs bump into all kinds of substances—some with names that barely fit on the label. Calcium trifluoromethanesulphonate pops up as a reagent in some chemical syntheses. Most folks haven’t heard of it outside of niche chemistry. That doesn’t mean its safety deserves less scrutiny. If you spend enough time handling chemicals, the question moves from “what’s it for?” to “do I need to worry about this?”
Understanding the Hazards
If you take a look at the data sheets for calcium trifluoromethanesulphonate, you’ll spot a familiar warning: avoid breathing the dust, prevent skin and eye contact, wash up thoroughly after use. Some sources list it as an irritant. Not a surprise—many sulfonate salts and fluorinated chemicals in the lab share similar traits. Even so, none of that makes it “just another salt.” Repeated exposure to fine powders, even from materials labeled as low-to-moderate risk, adds up. Anyone who has mixed or weighed dry chemicals knows dust finds a way to linger, especially if air conditioning or static electricity gets involved.
Personal Experience in the Lab
I’ve handled plenty of powdered chemicals under a fume hood, and wearing gloves and goggles becomes second nature. Years ago, a rushed student knocked a jar off the bench. The cloud that escaped took two days to clear out, with everyone hacking and red-eyed for hours. No one remembers the compound’s name, but everyone remembers not wanting to be in that room. Even chemicals without labels like “acutely toxic” can still cause trouble. Skin dries out, eyes water, throats scratch—signals that the body would rather be anywhere else. Respect starts at the bench, long before reaching the Material Safety Data Sheet.
Industry Guidance
Organizations like OSHA and NIH offer simple guidelines: use eye protection, glove up, keep things clean, and don’t touch your face. Calcium trifluoromethanesulphonate isn’t known for life-threatening effects at the scale seen in academic work. Still, it’s got that fluorine count, which hints at more stubborn chemistry. Alongside the direct irritation, poorly-ventilated spaces or messy benches only magnify the risk.
Better Practices, Fewer Headaches
Instead of trusting stocked lab gloves and disposable goggles are enough, build habits. Swap damaged gloves the moment you spot a tear—don’t leave them for the next person. Weigh powders inside fume hoods with the sash pulled as low as possible. Label containers clearly, even for lesser-known chemicals. Spills should mean immediate attention, not a shrug. Encourage everyone, veterans and newbies alike, to speak up if cleanup slips or dust escapes.
The Responsibility We Share
Working with chemical salts like calcium trifluoromethanesulphonate calls for humility in the face of the unknown. Safety rules stick around for a reason. They’re written with the mistakes and mishaps of many before us. Even the most mundane-seeming bottle on the shelf can cause lingering issues if handled carelessly. Staying curious about what we work with, and sharing knowledge gained from experience, builds a safer environment. That goes further than any label or chemical property.
The Overlooked Details Behind Chemical Storage
It’s easy to skip the discussion about chemical storage when daily lab life swallows up so much attention. Most folks remember to tighten the cap and keep things off the edge of a bench, but there’s more to keeping something like calcium trifluoromethansulphonate safe than just a closed container. I’ve seen what happens when storage gets sloppy—labels fade, containers sweat, and sensitive materials lose their punch. Spending time on good habits makes a difference.
Understanding the Nature of the Compound
Calcium trifluoromethansulphonate has a character that sets it apart from table salt or common solvents. With its popularity rising, both in industry and some research settings, word is getting around that moisture and exposure to air spell trouble for this compound. It tends to pull in water from the environment, and that changes its chemical structure, which then throws off lab results or product formulations.
From The Storage Room: Practical Wisdom
In my own lab days, finding material ruined by a little carelessness could stop a project cold. After enough years spent among jars, jars, and more jars, I noticed a couple of practices that lowered losses and cut down on unnecessary headaches later on. First, switching from plastic tubs to glass bottles with secure screw tops helped shut out both air and stray humidity. I also learned early not to tuck sensitive chemicals next to acids, strong oxidizers, or bases. Even a whiff of fumes from those neighbors creeps in and triggers a reaction given enough time.
Keeping Stability: Common-Sense Measures
Calcium trifluoromethansulphonate does its best in a cool, dry, and shady spot. Sunlight might look nice streaming through a window, but it’s not much help for stability. Once moisture gets in, things clump, react, or leave residues that are tough to clean out of equipment. Having shelves away from sinks and avoiding busy corners of the bench with lots of accidental knocks and spills turns out to be well worth the effort.
Labeling and Date Checks: Small Steps, Big Gains
More than once, a quick date check on a label dodged wasted time and money. Chemicals change with age and exposure, especially the ones that fuss over air and water. I started penciling in the date received, the date the container opened, and it paid off every time I went hunting for what might be past its prime. Fresh eyes catch what a rushed day misses. I always pushed to keep backup labels in the drawer and a marker handy, because faded printing doesn't help when you need it.
Looking Ahead: Room for Improvement
Even with careful routines, there’s always a way to tighten things up. Regular training refreshers keep the newest folks from missing crucial steps. Digital inventory systems lend a hand, automatically reminding staff when something’s probably due for a check or replacement. Getting feedback from everyone who uses these chemicals makes a difference too—not every detail gets noticed from behind a desk.
Thoroughness Isn’t Red Tape—It’s Protection
Folks who see chemical storage as a chore often miss the broader view. Each little safeguard pulls its weight, both for safety and the wallet. Calcium trifluoromethansulphonate isn’t the only compound that rewards careful attention, but it’s a good example that small storage tweaks prevent bigger headaches. Sticking to practices that keep air and water away feels like a burden right up until the moment everything works as it should, every time.
Hidden Backbone in Electronics
You might not spot calcium trifluoromethansulphonate on a factory shelf, but it plays a crucial part in industries that shape modern life. Across the world, engineers turn to this compound for its electrical properties. In the lab, I’ve watched as developers rely on it to boost the performance of batteries—especially those that need strong, stable electrolytes. Lithium-ion and lithium-metal batteries, for instance, use salts like this to support higher energy densities. The impact? Lighter laptops and electric cars that go farther than ever before.
Push for Greener Chemistry
Manufacturing sometimes gets tough on the environment. Calcium trifluoromethansulphonate stands out for its relative stability, which cuts down on hazardous byproducts during production processes. Its chemical structure helps it resist breakdown even at higher temperatures. In the world of sustainable technology, this matters. Companies searching for ways to cut emissions or replace toxic substances have started paying more attention to this calcium salt. It’s far from a perfect solution, but for certain polymerizations or as a catalyst, it gets the job done with fewer unwanted surprises.
Helping Polymers Reach New Heights
Anyone who’s worked with plastics knows that even small tweaks in chemistry can change how products behave. Plastic manufacturers add calcium trifluoromethansulphonate to tweak polymer structures. It supports cleaner reactions and boosts the final product’s resistance to heat or moisture. Cable insulation, specialty coatings, and components for smart gadgets all see benefits from this approach. In my own research group, swapping in this compound kept our materials stronger in humidity and less prone to cracking.
Cleaner Water Treatment Processes
Water treatment sometimes calls for more than just filtration. Here, calcium trifluoromethansulphonate finds use as a flocculant or a supporting chemical in advanced filtration systems. Its properties help separate out tiny contaminants more effectively. I’ve seen municipal systems turn to specialty additives like this to get ahead of ever-tougher water quality rules. While it’s not a magic bullet for every pollutant, it does provide one more tool for engineers trying to deliver safer drinking water.
Next Steps: Innovation and Responsibility
Looking down the line, the growing popularity of cleaner energy and careful resource management will push chemists to get even more from calcium trifluoromethansulphonate. The push for lead-free and less toxic compounds in electronics keeps this compound in the spotlight. We’re seeing a shift—more transparent supply chains, rigorous testing for leaching, and partnerships between labs and manufacturers. In my view, this approach builds real trust. Trust is just as important as performance specs. Industry shouldn’t just focus on what a compound can do, but also on how it’s sourced, handled, and recycled at the end of a product’s life.